10 May 2022 : Original article
Long-Term Effectiveness, Safety, and Patient-Reported Outcomes of Self-Administered Subcutaneous Hepatitis B Immunoglobulin in Liver Post-Transplant Hepatitis B Prophylaxis: A Prospective Non-Interventional Study
Bruno Roche1BCDE, Artur Bauhofer2ACDEF*, Miguel Ãngel Gomez Bravo3BCDE, Georges Philippe Pageaux4ABCDE, Fabien Zoulim5BDE, Alejandra Otero6BC, Martin Prieto78BC, Carmen Baliellas9BC, Didier Samuel1ACDEDOI: 10.12659/AOT.936162
Ann Transplant 2022; 27:e936162
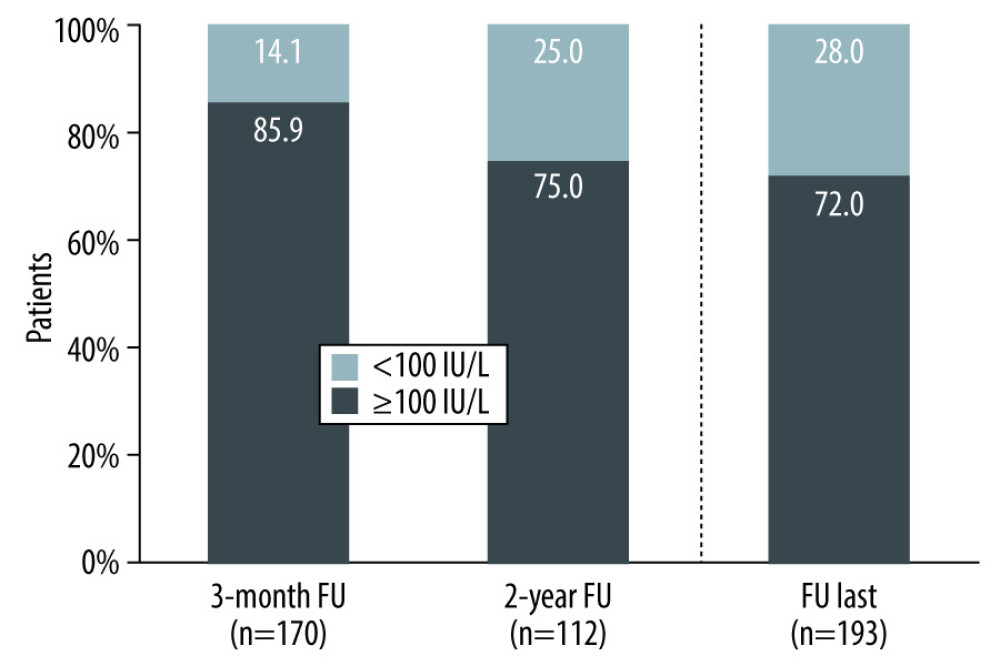
Figure 2 Proportion of patients with serum anti-HBs trough levels at/above or below 100 IU/L under treatment with s.c. HBIg. Percentages are based on patients with quantitative anti-HBs test results available at the respective visits (number in brackets). FU last: last available anti-HBs value documented in a patient (Prepared with Microsoft Office 2016).ADR – adverse drug reaction (adverse event possibly related to treatment with sc HBIg); MedDRA – Medical Dictionary for Regulatory Activities; PT – preferred term.








