27 October 2020: Review Paper
Prognosis and Treatment of Liver Transplant Recipients in the COVID-19 Era: A Literature Review
Ashraf Imam1ADEFG, Sadi A. Abukhalaf1ABCDEF, Hadar Merhav1CDFG, Samir Abu-Gazala1ABDG, Oded Cohen-Arazi1BCEG, Alon Josef Pikarsky1DEFG, Rifaat Safadi2EFG, Abed Khalaileh1AEFG*DOI: 10.12659/AOT.926196
Ann Transplant 2020; 25:e926196
Abstract
ABSTRACT: Liver transplantation during the COVID-19 pandemic is challenging. Both donor and recipient issues can be influenced by the risks attributed to the pandemic. Allocation policy may need to be modified and criteria may be influenced by local infection rates and availability of medical facilities. Modifying immunosuppression (IS) protocols is controversial and is not evidence-based. In this study, we review the published literature on liver transplant recipients who were infected with COVID-19. A literature review was performed using PubMed, ScienceDirect, and WHO databases to identify relevant English-language articles published up to May 20, 2020. Fifteen articles reported 120 liver transplant recipients who were infected with COVID-19. Only 10 papers with 22 patients reported full encounter characteristics. Four papers reported 23, 17, 13, and 6 patients, respectively, but with minimal data. One paper reported the authors’ own 39 patients’ characteristics and demographics. The mean age was 58.2 years with 66% males. The most commonly reported presentations in descending order were fever (91%), cough (36.7%), shortness of breath (SOB) (31.8%), and diarrhea (31.8%). Liver transplant patients infected with COVID-19 were maintained on Tac (79%), mycophenolate (MMF) (48.4%), and Prednisone (29.6%) and were managed by reducing MMF in 14.3% of patients and reducing Tac in 14.3% of patients; 28.6% of patients needed ICU admission, 13.6% of patients had died, and the reported general population COVID-19 mortality rate was 3.4%. The clinical presentation of COVID-19 in liver transplant recipients may be different from the general population, with higher rates of severe disease, complications, and mortality.
Keywords: COVID-19, Liver Transplantation, Organ Transplantation, SARS Virus, Betacoronavirus, COVID-19, Coronavirus Infections, Humans, Infection Control, Liver Diseases, Pandemics, Pneumonia, Viral, SARS-CoV-2
Background
Coronavirus disease 2019 (COVID-19) is caused by the severe acute respiratory syndrome coronavirus type 2 (SARS-CoV2) [1]. The disease was initially confirmed in China and then rapidly spread worldwide with more than 4.8 million infected individuals and over 300 000 deaths worldwide [2]. This disease is especially deadly in elderly patients (over 70 years old) with comorbidities [3]. Most published data regarding COVID-19 and organ transplant recipients are nonspecific and lack quality evidence. Data on demographics, characteristics, and clinical presentations of COVID-19 in organ transplant recipients are scarce. In the present study, we review the published literature regarding liver transplant patients who were infected with COVID-19.
Material and Methods
LITERATURE SEARCH:
A systematic literature review was performed using PubMed and ScienceDirect databases to identify relevant English-language articles published through May 20, 2020. Search terms were COVID-19, coronavirus, severe acute respiratory syndrome coronavirus 2, 2019-nCoV, SARS-CoV-2, SARS-CoV, MERS-CoV, liver transplantation, and liver transplant. All article types were included: case reports, case series, commentaries, and review articles. A search in the database of the COVID-19 Global Research on Coronavirus Disease section of the WHO website through May 20, 2020 was performed using the following criteria: liver transplantation or liver transplant with English language as limit or filter [4]. Additional articles were retrieved by screening the reference lists of the included studies. The search strategy was approved and reviewed by all authors.
ELIGIBILITY CRITERIA AND STUDY SELECTION:
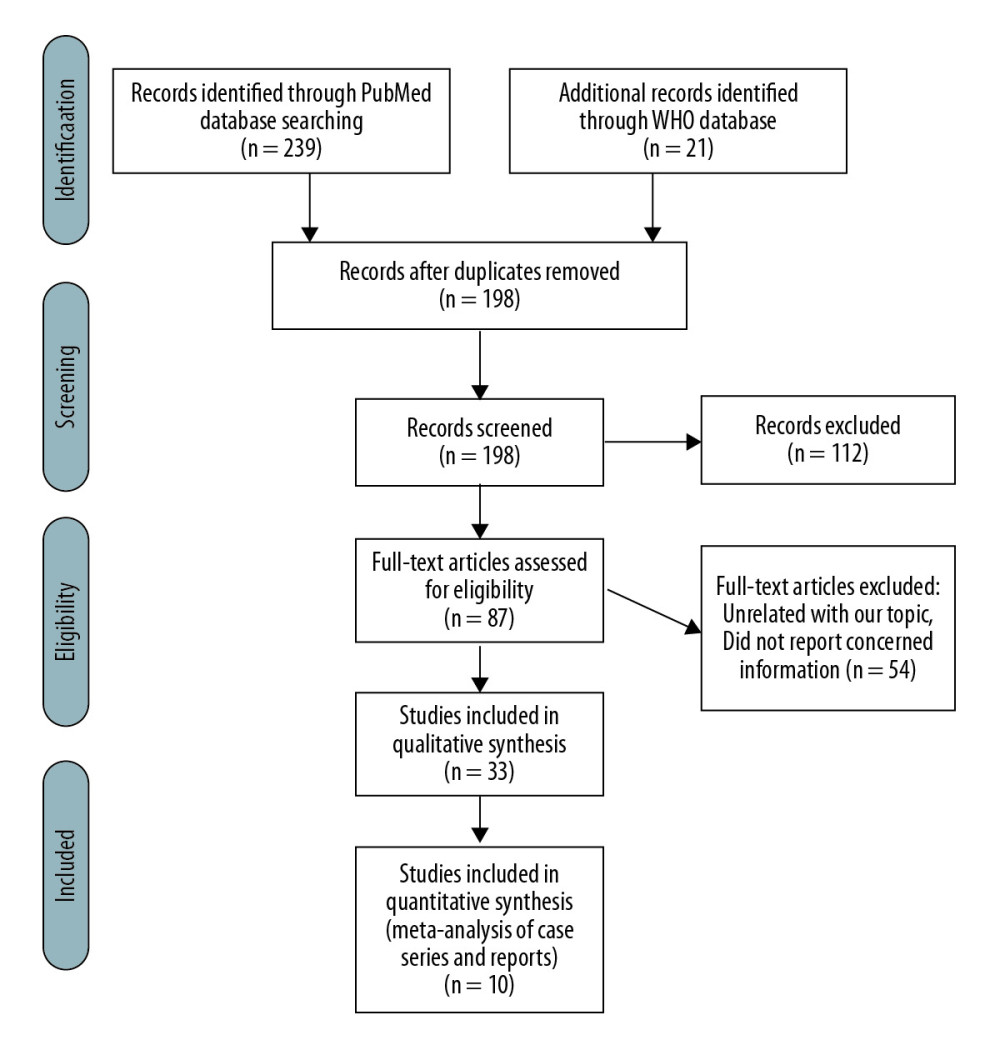
The authors independently reviewed the titles and abstracts for inclusion. Figure 1 displays the flow diagram for this systematic review, based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 [5]. Databases were screened, filtered, and assessed for eligibility. Cases of COVID-19 in liver transplant patients were included in this study. Articles with unrelated topics and/or with missed information were excluded.
RISK OF BIAS:
The National Institutes of Health Quality Assessment Tool for Case Series Studies was used to qualify the reviewed articles [6]. Table 1 shows the results of the 2 reviewers who independently rated the quality of the included studies.
DATA EXTRACTION AND SYNTHESIS:
Data were independently extracted from reports by 2 reviewers. All reported patients’ demographic and clinical characteristics (country, age, sex, time from transplant, comorbidities, clinical presentation and maximum body temperature, initial laboratory values, baseline immunosuppressant medications (ISM), need for intensive care unit (ICU), duration of illness, and outcomes) were extracted, collected, and analyzed. Due to the lack of sufficient data, we did not perform a meta-analysis to assess the association of various patients’ findings with demographic data, disease and patient characteristics, or outcomes. The principal summary measures used were the median, mean, standard deviation, and incidence.
Results
OVERVIEW OF THE INCLUDED STUDIES:
A total of 260 articles were retrieved using the search strategy. After removing duplicate articles, 198 articles were screened, of which 112 articles were excluded due to unrelated content. The remaining 87 articles were assessed for eligibility through full-text screening, then 54 articles were excluded due to unrelated content or lack of relevant information. There were 33 articles included but only 15 articles reported liver transplant recipients infected with COVID-19 (Figure 1). Of these 15 articles, there were 2 case series and 8 case reports that reported a total of 22 liver transplant patients. Four articles reported 23, 17, 13, and 6 patients, respectively, but with minimal data [7–10]. One paper reported the authors’ own 39 patients’ characteristics and demographics [11] and was used in Table 2 for comparison and to extract pooled measurements. The remaining 18 articles were commentaries and review articles. Patients’ characteristics and demographics were included in Tables 3–7.
PATIENTS DEMOGRAPHICS AND CHARACTERISTICS:
Ten articles reported 22 liver transplant patients infected with COVID-19. There were 8 from Italy, 6 from Spain, 4 from China, 3 from the USA, and 1 from France. There were 15 (68.2%) males. The mean age was 55.96 (0.5–79) years. The mean post-transplant period was 8.75 (0.005–26) years, and 22.7% of patients were within the first year after transplantation. The most common reported comorbidities were diabetes mellitus in 22.7%, heart disease in 22.7%, lung disease in 22.7%, HTN in 18.2%, and obesity in 4.6%, and 40.9% of patients had no reported comorbidities.
CLINICAL PRESENTATION:
The most frequently reported clinical presentation was fever; it was reported in 90.9%, with a mean maximum temperature of 39.26°C (±0.54). Other reported clinical symptoms were cough (36.7%), shortness of breath (31.8%), unspecified respiratory symptoms (31.8%), diarrhea (31.8%), and fatigue (27.3%). Less frequently reported symptoms included headache, dizziness, weakness, abdominal discomfort, sleep disorders, chills, rhinitis, confusion, myalgia, and chest pain.
LABORATORY RESULTS:
The initial white blood cell count median was 3.2×103 (±1.56×103). The initial median lymphocyte cell count was 0.67×103 (±0.18×103). Initial leukopenia and lymphopenia were reported in 60% and 100%, respectively. The median initial CRP was 21.5 (±15.6) mg/L, and high CRP (>5 mg/dl) levels were noted in 75%. High ALT (>50 U/l) and AST (>54 U/l) were reported in 71.4% and 60%, respectively.
IMMUNOSUPPRESSION MANAGEMENT:
Patients were treated with different IS regimens. Tacrolimus (Tac) was prescribed in 68.2% of patients and discontinued in 14.3%, reduced in 14.3%, and not changed in 28.6%. MMF was prescribed in 45.5% of patients and discontinued in 7.1% and reduced in 14.3%. The exact MMF management was not reported in the remaining 79% of patients. Pred was prescribed in 18.2% of patients and not changed in 50% of patients; the exact Pred management was not reported in the remaining 50% of patients. Other baseline ISMs were Azathioprine, Everolimus, and Cyclosporin. These medications were held in some patients and not changed in others; 27.2% of patients had no change in ISM and recovered.
Deceased patients were prescribed MMF in 100%, Tac in 33.3%, and EVE in 33.3% and had their ISM held (MMF 66.6%; EVE 100%) or reduced (MMF 33.3%; Tac 33.3%).
COVID-19 DIRECTED MANAGEMENT:
Treatment targeted COVID-19 included hydroxychloroquine (42.9%), antibiotics (35.7%), lopinavir/ritonavir (28.6%), INF-a,b (28.6%), intravenous methylprednisolone (21.4%), intravenous immunoglobulin (14.3%), oseltamivir (14.3%), azithromycin (7.1%), and tocilizumab (7.1%). COVID-19 targeting treatment used in patients who died included antibiotics (33.3%), hydroxychloroquine (33.3%), lopinavir/ritonavir (66.6%), INF-a,b (66.6%), and umifenovir (33.3%).
PATIENTS OUTCOMES:
Intensive care unit (ICU) admission occurred in 28.6% of patients, 72.7% of patients had recovered and were discharged with a median illness duration of 17 days (range, 6–53 days), 13.6% of patients were alive and hospitalized at the time of publication with a median illness duration of 16 days, and 13.6% of patients had died with a median illness duration of 24 days (range, 7–45 days).
CHARACTERISTICS OF PATIENTS WHO DIED:
The mean age of patients who dies was 68 years, 66.6% of patients were older than 65 years, 100% of patients were male, and the mean time from transplant was 8.2 years. Diabetes mellitus, hypertension (HTN), obesity, asthma, and bronchiectasis were reported in these patients; 66.6% had lymphopenia and 66.6% had high CRP serum levels. ARDS and acute renal failure were reported in 100% and 66.6% of patients, respectively.
POOLED RESULTS:
One study reported 39 patients without demographics and characteristics for each patient and these were thus not included in our tabulated results. However, the study was used to draw pooled measures for all reported liver transplant patients infected with COVID-19 in the literature. A total of 61 liver transplant patients with COVID-19 used in the comparison. The age mean was 58.25 years and 66.15% were males. The mean age at transplant was 7.13 years. HTN, DM, heart disease, and obesity were reported in 32.2%, 30.6%, 19%, and 15.1% of patients, respectively. Liver transplant patients infected with COVID-19 were maintained on Tac (79%), MMF (48.4%), and Pred (29.6%).
Discussion
Most published reports on COVID-19 in organ transplant recipients are nonspecific and lack quality evidence. Data on demographics, characteristics, and clinical presentations of COVID-19 in organ transplant recipients is scarce. However, it is well known that among patients with advanced age and medical comorbidities, COVID-19 is frequently severe. Studies have reported that COVID-19 can present differently in immunosuppressed patients, including organ transplant recipients.
Fever in COVID-19 in the general population is reported in 99% of patients [12,13]. Our study has shown that 9% of the liver transplant patients did not have fever on presentation or during their hospitalization. On the other hand, cough, SOB, myalgia, headache, sore throat, and gastrointestinal symptoms were different in incidence from the typical COVID-19 presentation [12–15]. Additionally, this review shows that there are several unreported symptoms that can appear in the COVID-19-positive liver transplant patients, including chest tightness and pain and jaundice [14]. Notably, the unreported chest tightness and pain in other cohorts have an incidence of 9% in this cohort.
COVID-19 causes pneumonia that can be severe enough to be lethal, especially in patients with advanced age or underlying medical comorbidities [16]. Those comorbidities include cardiovascular disease, diabetes mellitus, HTN, chronic lung disease, cancer, chronic kidney disease, and obesity (body mass index ≥30) [3,16]. Liver transplant patients infected with COVID-19 are a fragile and high-risk group due to immunosuppression and common comorbidities. In this review, 60% of patients had comorbidities. These comorbidities predispose these patients to a more severe COVID-19 infection. The attendant IS is an additional risk factor for severe disease. Liver transplant recipients and candidates are in a high-risk group due to the high incidence and prevalence of hypertension, renal failure, diabetes, obesity, and advanced age in this group.
It was previously reported that a progressive decline in lymphocyte count was observed in non-survivors compared to more stable levels in survivors [12]. In the present study, 100% of patients who died had lymphopenia. Given the fact that ISM can induce lymphopenia, many liver transplant patients have a baseline lymphopenia that might further deteriorate and worsen the prognosis [17,18].
Several studies and a report from the Chinese Center for Disease Control and Prevention have classified COVID-19 severity in the general population [19,20] as mild, severe, and critical disease in 81%, 14%, and 5% of patients, respectively. In this report, severe disease defined by SOB and/or hypoxemia was reported in 31.3% of patients.
The WHO reported that recovery time appears to be around 2 weeks for mild infections and 3 to 6 weeks for severe disease [21]. In the present study, we found that 72% of patients recovered clinically from the COVID-19, with a median duration of illness of 17 (6–53) days. Rates of ICU admission were reported to range between 5% and 12% in the general population [22]. In our cohort, 28.6% of patients were admitted to the ICU.
The most recently reported mortality rate of COVID-19 was 3.77% [23]. The mortality rate of liver transplant patients infected with COVID-19 in this cohort was 13.6% (3 out of 22 patients). Despite the small number of patients included in the present study, this high mortality indicates that COVID-19 in liver transplant patients may portend an ominous outcome. Surprisingly, 17.6% of patients with time from transplant longer than 1 year had died, while there were no deaths within the first year of transplantation. This is despite the fact that the first year is fraught with postoperative complications and higher immunosuppression.
It was reported that 81% of COVID-19 patients present with mild disease and can be managed at home, and those with severe and critical COVID-19 disease should be managed with prompt hospitalization while ensuring appropriate infection control and supportive care [19,24,25]. The hospitalized patients should be managed with empiric treatment for bacterial pneumonia in selected patients, prevention of venous thromboembolism, and avoiding nebulized medications. The WHO and CDC recommend against use of systemic glucocorticoids in COVID-19 patients unless there are other indications [24,25]. To date, all suggested medications are under investigation, with no proven efficacy against COVID-19. These medications include Remdesivir, hydroxychloroquine/chloroquine, azithromycin, hydroxychloroquine, convalescent plasma, tocilizumab, favipiravir, interferon beta, and lopinavir/ritonavir. Most of our cohort were hospitalized and could not be managed at home due to the severity of the disease. They were managed with hydroxychloroquine (42.9%), antibiotics (35.7%), lopinavir/ritonavir (28.6%), INF-a,b (28.6%), intravenous methylprednisolone (21.4%), intravenous immunoglobulin (14.3%), oseltamivir (14.3%), azithromycin (7.1%), and tocilizumab (7.1%).
The effect of IS on the progression of COVID-19 is unclear. There are 2 aspects that should not be ignored when dealing with IS and COVID-19. First, it was proved that COVID-19 patients have a high prevalence of lymphopenia [26] but its unclear whether lymphopenia is a risk factor for COVID-19 or is a result of it. Second, it is thought that the severity of COVID-19 may be the result of a hyperinflammatory response (cytokine storm). Thus, the role of immunomodulation may have a positive and not just a negative effect in the treatment of severe cases [27,28].
Interestingly, a recent Italian report by Lorezo D’Antiga concluded that immunocompromised patients do not have an increased risk of developing severe pulmonary disease compared to the general population [29]. Nevertheless, it is evident from this study that liver transplant patients are at an increased risk for serious, critical, and fatal disease when infected with COVID-19.
Conclusions
Liver transplant patients may have an atypical clinical presentation of COVID-19 infection. Fever may be absent while other atypical symptoms may prevail. Therefore, a high index of suspicion for COVID-19 and perhaps even surveillance in this population may help in early diagnosis and prevention of further transmission. The role of IS therapy should be assessed in every case individually.
Tables
Table 1. Quality ratings of included studies according to NIH Quality Assessment Tool for case series studies.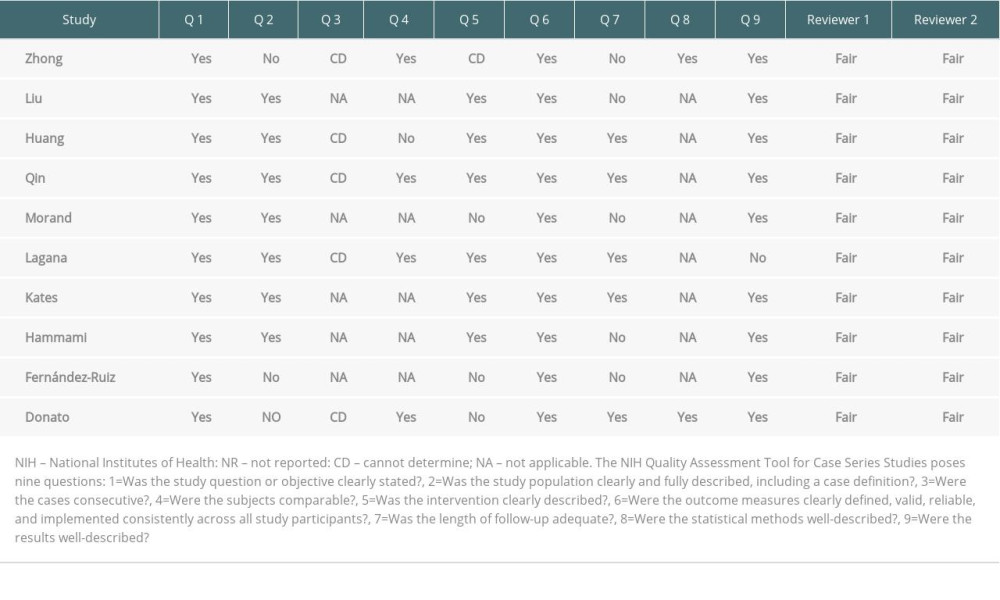 Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies.
Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies.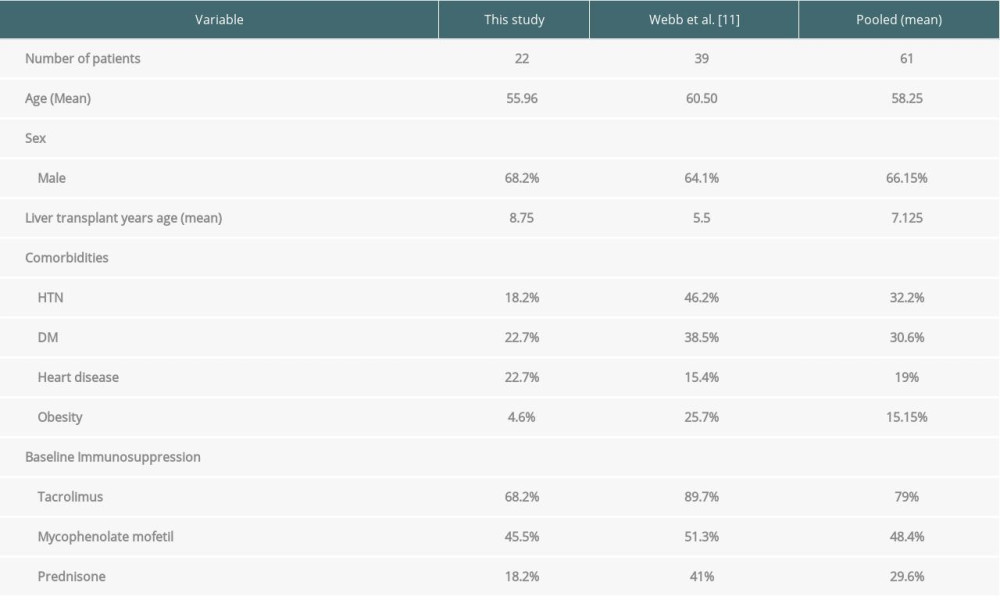 Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19.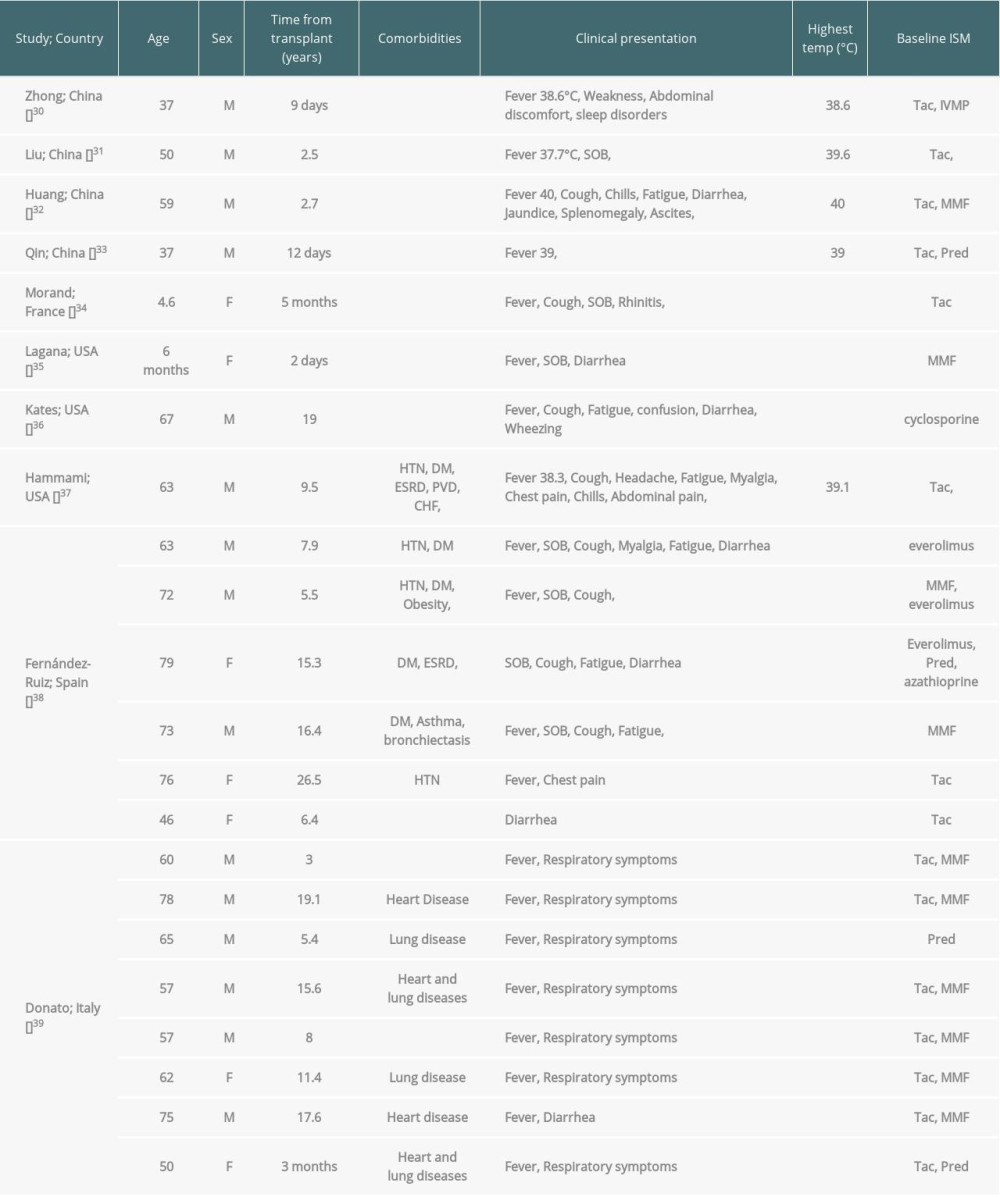 Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19.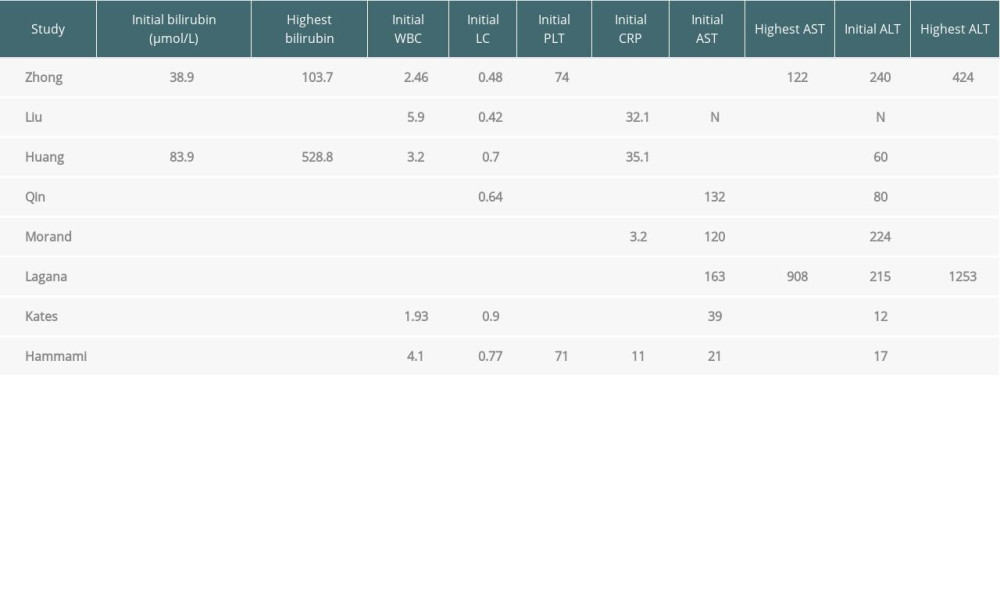 Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19.
Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19.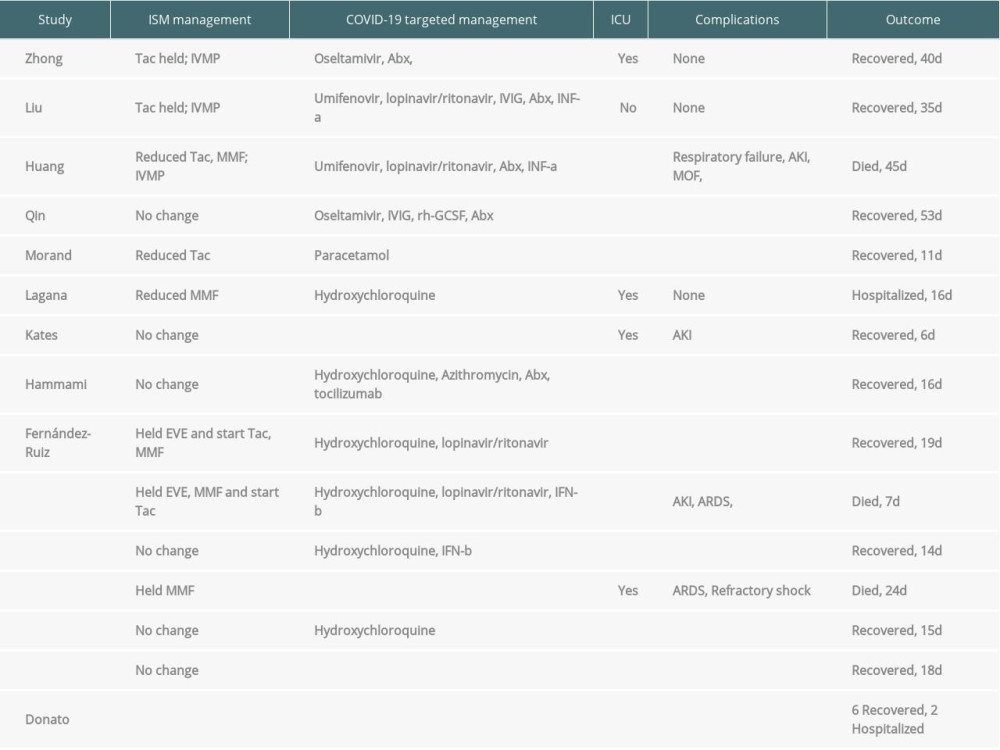 Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19.
Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19.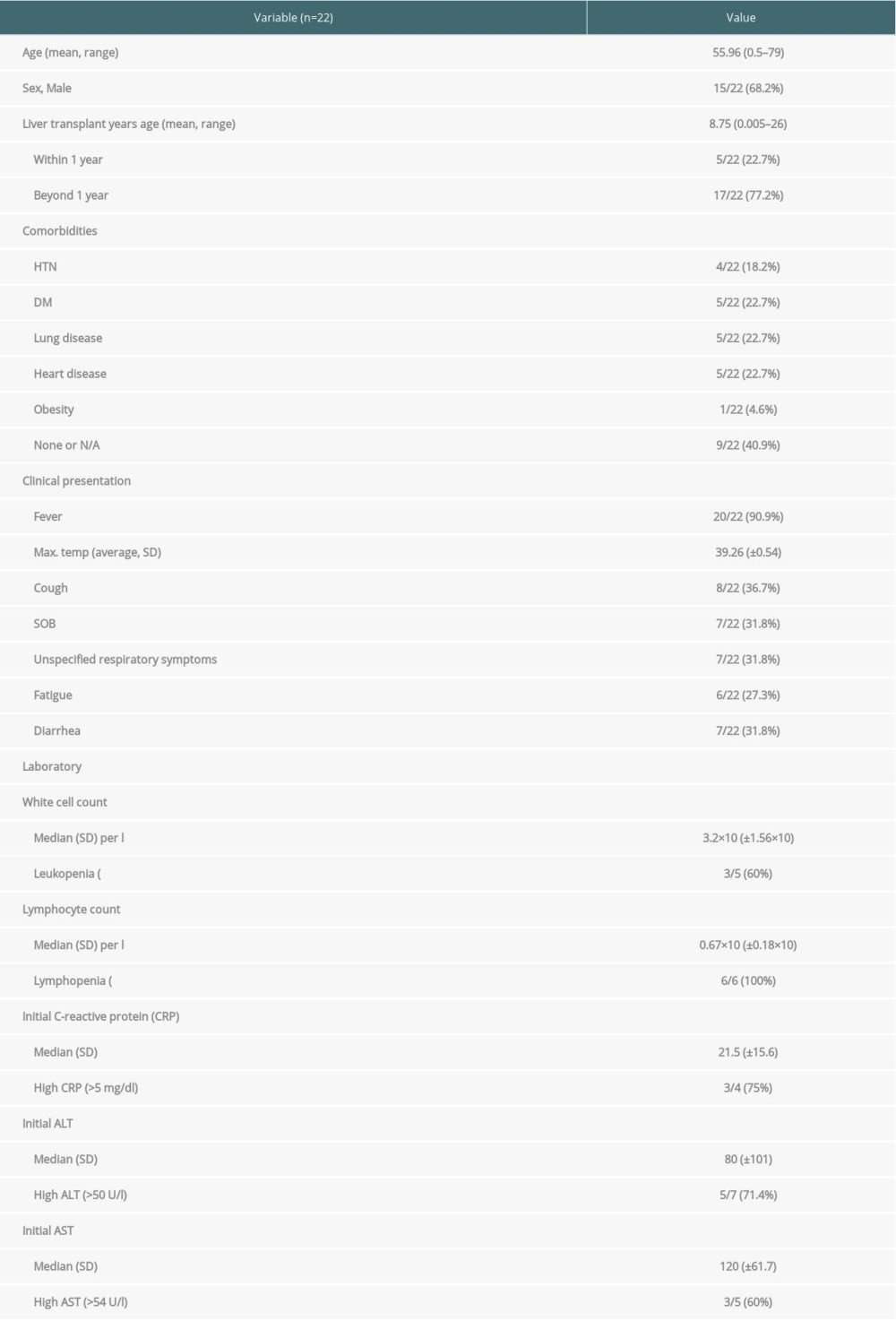 Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19.
Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19.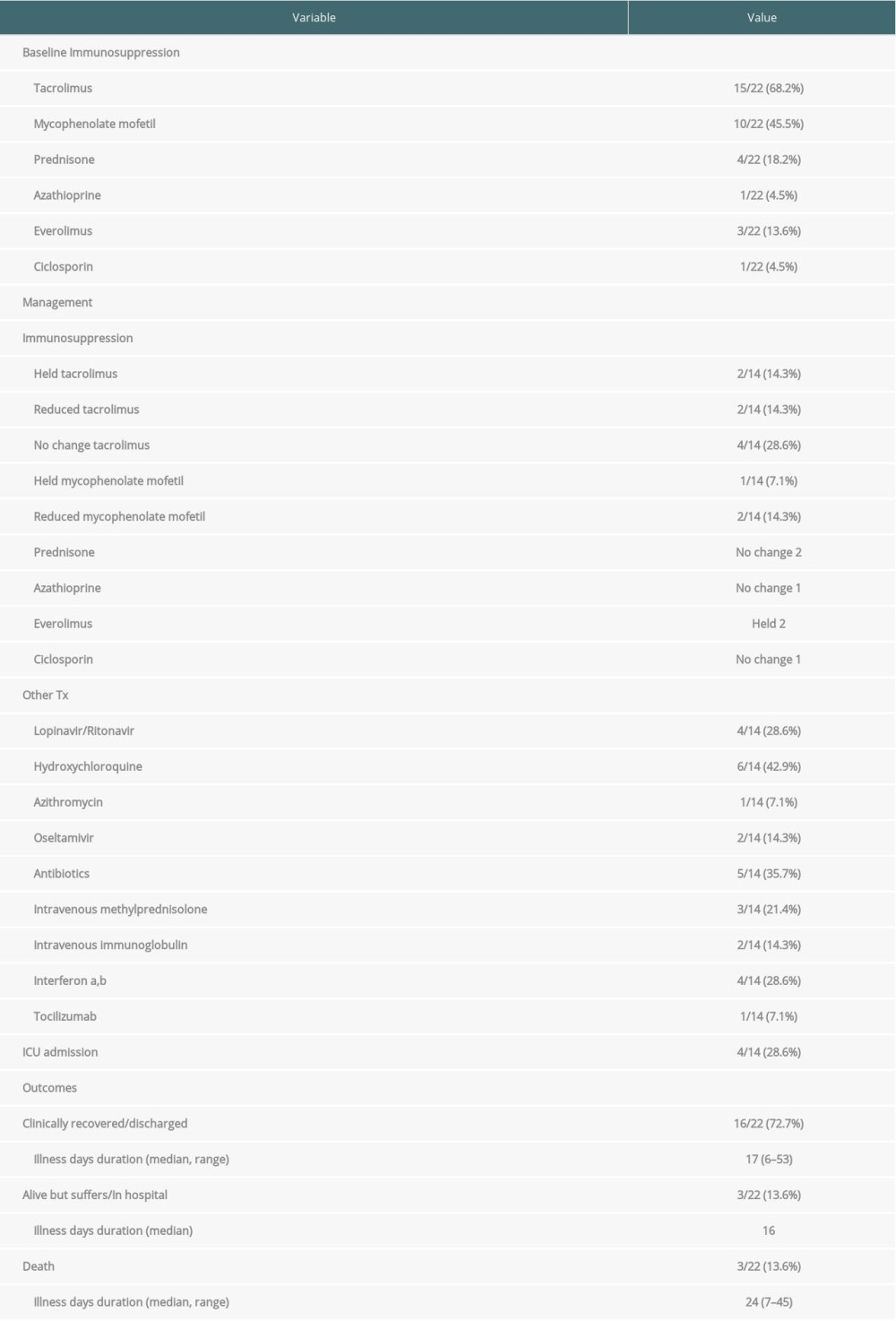
References
1. World Health Organization (WHO): Novel coronavirus (2019 nCoV), 2020 https://www.who.int/emergencies/diseases/novel-coronavirus-2019
2. World Health Organization (WHO): Coronavirus disease 2019 (COVID-19) Situation Report – 88, 2020 https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200417-sitrep-88-covid-191b6cccd94f8b4f219377bff55719a6ed.pdf?sfvrsn=ebe78315_6
3. Zhou F, Yu T, Du R, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study: Lancet, 2020; 395(10229); 1054-62
4. World Health Organization (WHO): COVID-19 global literature on coronavirus disease, 2020 https://search.bvsalud.org/global-literature-on-novel-coronavirus-2019-ncov/
5. Moher D, Liberati A, Tetzlaff J, Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement: PLoS Med, 2009; 6(7); e1000097
6. National Heart Blood Institute: Study quality assessment tools, 2020 https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools
7. Boyarsky BJ, Chiang TPY, Werbel WA, Early impact of COVID-19 on transplant center practices and policies in the United States: Am J Transplant, 2020; 20(7); 1809-18
8. Maggi U, De Carlis L, Yiu D, The impact of the COVID-19 outbreak on Liver Transplantation programmes in Northern Italy: Am J Transplant, 2020; 20(7); 1840-48
9. Pereira MR, Mohan S, Cohen DJ, COVID-19 in Solid Organ Transplant Recipients: Initial Report from the US Epicenter: Am J Transplant, 2020; 20(7); 1800-8
10. Bhoori S, Rossi RE, Citterio D, COVID-19 in long-term liver transplant patients: Preliminary experience from an Italian transplant centre in Lombardy: Lancet Gastroenterol Hepatol, 2020; 5(6); 532-33
11. Webb GJ, Moon AM, Barnes E, Determining risk factors for mortality in liver transplant patients with COVID-19: Lancet Gastroenterol Hepatol, 2020; 5(7); 643-44
12. Wang D, Hu B, Hu C, Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus – infected pneumonia in Wuhan, China: JAMA, 2020; 323(11); 1061-69
13. Huang C, Wang Y, Li X, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China: Lancet, 2020; 395; 497-506
14. World Health Organization (WHO): Report of the WHO-China joint mission on coronavirus disease 2019 (COVID-19), 2020 https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-fnal-report.pdf
15. Cheung KS, Hung IFN, Chan PPY, Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from the Hong Kong cohort and systematic review and meta-analysis: Gastroenterology, 2020; 159(1); 81-95
16. Wu Z, McGoogan JM, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention: JAMA, 2020 [Online ahead of print]
17. Banerjee D, Popoola J, Shah S, COVID-19 infection in kidney transplant recipients: Kidney Int, 2020; 97(6); 1076-82
18. Chen S, Yin Q, Shi H, A familial cluster, including a kidney transplant recipient, of Coronavirus Disease 2019 (COVID-19) in Wuhan, China: Am J Transplant, 2020; 20(7); 1869-74
19. Yang X, Yu Y, Xu J, Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study: Lancet Respir Med, 2020; 8(5); 475-81
20. Kujawski SA, Wong KK, Collins JP, First 12 patients with coronavirus disease 2019 (COVID-19) in the United States: MedRxiv, 2020 20032896
21. World Health Organization: Director-General’s opening remarks at the media briefing on COVID-19 February 24, 2020 https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-themedia-briefng-on-covid-19---24-february-2020
22. Grasselli G, Pesenti A, Cecconi M, Critical care utilization for the COVID-19 outbreak in Lombardy, Italy: Early experience and forecast during an emergency response: JAMA, 2020 [Online ahead of print]
23. Zhang J, Wang X, Jia X, Risk factors for disease severity, unimprovement, and mortality of COVID-19 patients in Wuhan, China: Clin Microbiol Infect, 2020; 26(6); 767-72
24. Centers for Disease Control and Prevention: Interim clinical guidance for management of patients with confirmed 2019 novel coronavirus (2019-nCoV) infection, 2020
25. World Health Organization: Home care for patients with suspected novel coronavirus (nCoV) infection presenting with mild symptoms and management of contacts, 2020
26. Guan W, Ni Z, Hu Y, Clinical characteristics of 2019 novel coronavirus infection in China: MedRxiv, 2020 20020974
27. Ritchie AI, Singanayagam A, Immunosuppression for hyperinflammation in COVID-19: a double-edged sword?: Lancet (London, England), 2020; 95; 1111
28. Booth CM, Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area: JAMA, 2003; 289(12); 2801-9
29. D’Antiga L, Coronaviruses and immunosuppressed patients: The facts during the third epidemic: Liver Transpl, 2020; 26(6); 832-34
30. Zhong Z, Zhang Q, Xia H, Clinical characteristics and immunosuppressant management of coronavirus disease 2019 in solid organ transplant recipients: Am J Transplant, 2020; 20(7); 1916-21
31. Bin L, Wang Y, Zhao Y, Successful treatment of severe COVID-19 pneumonia in a liver transplant recipient: Am J Transplant, 2020; 20(7); 1891-95
32. Huang J-F, Zheng KI, George J, Fatal outcome in a liver transplant recipient with COVID-19: Am J Transplant, 2020; 20(7); 1907-10
33. Qin J, Wang H, Qin X, Perioperative presentation of COVID-19 disease in a liver transplant recipient: Hepatology, 2020 [Online ahead of print]
34. Morand A, Roquelaure B, Colson P, Child with liver transplant recovers from COVID-19 infection. A case report: Arch Pediatr, 2020; 27(5); 275-76
35. Lagana SM, De Michele S, Lee MJ, COVID-19 associated hepatitis complicating recent living donor liver transplantation: Arch Pathol Lab Med, 2020 [Online ahead of print]
36. Kates OS, Fisher CE, Stankiewicz-Karita HC, Earliest cases of coronavirus disease 2019 (COVID-19) identified in solid organ transplant recipients in the United States: Am J Transplant, 2020; 20(7); 1885-90
37. Hammami MB, Garibaldi B, Shah P, Clinical course of COVID-19 in a liver transplant recipient on hemodialysis and response to tocilizumab therapy: A case report: Am J Transplant, 2020; 20(8); 2254-59
38. Fernández-Ruiz M, Andrés A, Loinaz CA, COVID-19 in solid organ transplant recipients: a single-center case series from Spain: Am J Transplant, 2020; 20(7); 1849-58
39. Donato MF, Invernizzi F, Lampertico P, Rossi G, Health status of liver transplanted patients during the coronavirus outbreak in Italy: a large single center experience from Milan: Clin Gastroenterol Hepatol 04 01, 2020; 18 2020
Tables
 Table 1. Quality ratings of included studies according to NIH Quality Assessment Tool for case series studies.
Table 1. Quality ratings of included studies according to NIH Quality Assessment Tool for case series studies. Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies.
Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies. Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19. Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19. Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19.
Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19. Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19.
Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19. Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19.
Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19. Table 1. Quality ratings of included studies according to NIH Quality Assessment Tool for case series studies.
Table 1. Quality ratings of included studies according to NIH Quality Assessment Tool for case series studies. Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies.
Table 2. Clinical characteristics of the 22 reported liver transplant patients infected with COVID-19 compared with previous published studies. Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 3. Characteristics of all available reported liver transplant recipients infected with COVID-19. Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19.
Table 4. Laboratory characteristics of all available reported liver transplant recipients infected with COVID-19. Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19.
Table 5. Management and outcomes of all available reported liver transplant recipients infected with COVID-19. Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19.
Table 6. Clinical characteristics for the 22 reported liver transplant patients infected with COVID-19. Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19.
Table 7. Management and outcomes of the 22 reported liver transplant patients infected with COVID-19. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860