19 February 2021: Original Paper
Short- and Long-Term Outcomes of Different Reperfusion Sequences in Liver Transplantation
Ann-Kathrin Eichelmann1BCEF*, Thomas Vogel1ABE, Ann-Kathrin Fuchs1BC, Philipp Houben1DE, Shadi Katou1BDF, Felix Becker1CD, Hartmut H. Schmidt2AD, Christian Wilms2DF, Andreas Pascher1BD, Jens G. Brockmann1ACDEDOI: 10.12659/AOT.926847
Ann Transplant 2021; 26:e926847
Abstract
BACKGROUND: Although most centers perform primary portal vein reperfusion (PV) in orthotopic liver transplantation (OLT) for historical reasons, there is so far no sound evidence as to whether this technique is superior. The present study evaluated the long-term outcome of 3 different reperfusion sequences: PV vs primary arterial (A) vs simultaneous reperfusion (SIM).
MATERIAL AND METHODS: All patients at our center who underwent OLT (who received a primary, whole-organ liver graft) from 2006 to 2007 were evaluated for analysis.
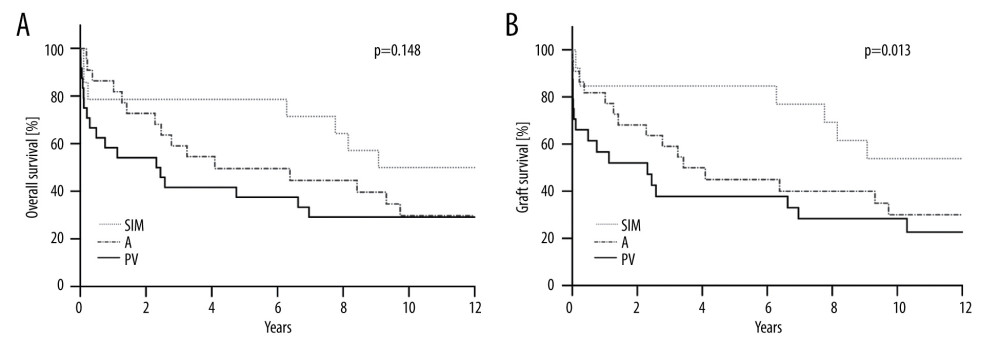
RESULTS: A total of 61 patients were found eligible (PV: 25, A: 22, SIM: 14). Twenty-one patients (35%) were still alive after the follow-up period of 12 years. Despite poorer starting conditions such as higher recipient age (59 y (SIM) vs 55 y (A) vs 50 y (PV), P=0.01) and donor age (56 y (SIM) vs 51 y (PV) vs 50 y (A), n.s.), higher MELD scores (22 vs 19 (PV) vs 17 (A), n.s.), as well as a higher number of marginal donor organs (79% (SIM) vs 36% (A/PV), P=0.02), SIM-recipients demonstrated superior outcomes. Overall survival was 8.1 y (SIM), 4.8 y (PV), and 5.9 y (A, n.s.)). None of the SIM-recipients underwent re-transplantation, while the rate was 32% in the PV-group. The 8.1 y graft survival in SIM-recipients was significantly longer than in the other 2 groups, which were 3.3 y (PV) and 5.5 y (A, P=0.013).
CONCLUSIONS: Although SIM-reperfused recipients were the oldest and received grafts of inferior quality, these recipients showed superior results in terms of overall patient and graft survival. Multicentric randomized controlled trials with larger study populations are required to confirm this finding.
Keywords: Liver Transplantation, Primary Graft Dysfunction, Carcinoma, Hepatocellular, End stage liver disease, Graft Survival, Liver Neoplasms, Reperfusion, Severity of Illness Index
Background
Since its first performance in 1963 by Thomas Starzl, orthotopic liver transplantation (OLT) has undergone remarkable progress. Advances in surgical procedure such as the piggy-back techniques or temporary portocaval bypass, perioperative management, more effective preservation solutions, and immunosuppressive therapy improved outcome. Lately, introduction of machine perfusion has become part of clinical routine [1]. Although all these achievements have improved 1-year patient and graft survival, none has shown benefits for graft attrition following year 1. Furthermore, there is still no consensus on the preferable reperfusion technique, and the question of how to reperfuse livers best is still controversial [2–6]. However, reperfusion sequence is of critical importance and several techniques have been proposed: retrograde vena cava reperfusion (RVC), anterograde reperfusion either via primary portal venous reperfusion (PV), primary arterial reperfusion (A), and simultaneous reperfusion (SIM). Since it is well known that ischemia-reperfusion injury represents one of the key factors affecting graft function, especially with regard to the fate of the biliary tract system, which is highly susceptible to oxygen deprivation, it is of great interest to identify the ideal reperfusion technique in order to decrease the incidence and magnitude of reperfusion syndrome and ischemia-reperfusion injury. Among the different reperfusion sequences, PV-reperfusion is the most widely used approach, as demonstrated by a survey on preferred reperfusion techniques among European transplantation centers. The survey revealed that PV is being performed in 46%, SIM-reperfusion in 39%, retrograde reperfusion in 11%, and initial hepatic artery reperfusion in 4% [2]. Reasons for this approach are thought to be the advantages of larger reperfusion volume resulting in a quicker graft rewarming than in primary arterial reperfusion, as well as a quicker decompression of the inferior venous and portal venous outflow, especially in cases of hemodynamic instability. However, PV-reperfusion is associated with an increased risk of warm ischemic damage to the biliary system, which is predominantly supplied by the hepatic artery. This disadvantage can be avoided by performing primary arterial reperfusion; however, this reperfusion technique results in slower graft rewarming because of the small reperfusion volume. Improvement of hemodynamic management and the introduction of a portocaval shunt have given rise to the SIM technique, in which the graft is simultaneously reperfused by the portal vein and the hepatic artery at the same time, at the cost of a slightly longer warm ischemic time [7,8]. Understandably, this technique results in a prolonged anhepatic phase and prolonged period of warm ischemia. It remains unclear so far whether these circumstances impact postoperative (long-term) graft function, attrition, and/or overall survival rates [4]. Since no evidence of clinical effects and long-term outcome of different reperfusion techniques exists, this retrospective study addresses outcome of 3 reperfusion sequences (PV vs A vs SIM), with special emphasis on long-term outcomes.
Material and Methods
STUDY POPULATION AND DATA COLLECTION:
All adult patients (>18 years) undergoing OLT in 2006 and 2007 at our center were retrospectively evaluated. Of 73 patients, 12 patients were excluded from further analysis because of combined transplantation (i.e., liver/intestine or liver/pancreas) or re-transplantation, resulting in 61 patients. In 7 patients, combined liver-kidney transplantation was performed. Each patient received a primary, ABO-compatible, whole-liver graft from a brain-dead donor. Donor and recipient characteristics are presented in Tables 1 and 2.
All procedures performed in this study were in accordance with the ethics standards of the Institutional and National Research Committee (Ethikkommission Münster, 2018-327-f-S) and with the 1964 Helsinki Declaration and its later amendments or comparable ethics standards. Moreover, informed consent was obtained from all participants included in the study. Demographic data of donors, including calculation of donor risk index (=DRI, according to Feng [9] and ET-DRI [10]) and recipients as well as operating data, were collected from patient charts, in-house transplant data files, and the Eurotransplant Network Information System.
OPERATIVE TECHNIQUES AND POSTOPERATIVE COURSE:
All transplantations were performed by 2 senior surgeons. After backtable preparation, the portal vein and hepatic artery were perfused via gravity perfusion (portal vein) and pressure perfusion (hepatic artery) with pressure perfusion. Additionally, bile ducts were flushed until the backflow was clear. OLT was carried out using piggy-back technique with anastomosis of the suprahepatic vena cava to the common ostium made from the recipients in 59 of 61 patients. In the remaining 2 patients, venous outflow was established by a side-to-side caval anastomosis (Belghiti, n=1) or by classical end-to-end reconstruction (n=1). A temporary portocaval shunt was used in 30 patients. After the anastomosis portal vein and/or the hepatic artery, the graft was reperfused (both surgeons performed either PV, A-, or SIM-reperfusion). Marginal grafts were preferred to be reperfused simultaneously. Thereafter, an end-end bile duct anastomosis was performed in 59 of 61 patients. Two patients received a biliodigestive anastomosis. Following surgery, standard immunosuppression consisted of FK-506, mycophenolate mofetil, and prednisolone without induction therapy as per local protocol. Steroids were tapered from 500 mg preoperatively to 5 mg within 4 weeks. Trough FK-506 levels were 6–8 ng/ml. Mycophenolate mofetil was started after the thrombocyte count was >70 000 cells/μl without trough level monitoring, aiming at 1000 mg BID. Biopsies were only performed on demand. In case of biopsy-proven rejection, a pulse of steroid was the first therapeutic measure performed.
OUTCOME MEASURES:
The outcome measures were total operating time, cold and warm ischemia times, necessity for transfusions, length of hospital stay, length of intensive care unit stay, and occurrence of complications, and laboratory data were analyzed. The follow-up time was 12 years. We analyzed the following long-term outcomes: overall patient and graft survival, incidence of biliary complications, necessity and number of endoscopic retrograde cholangiopancreatography (ERCP), necessity of re-transplantation, and rejection, and biochemical parameters, including liver enzymes and total bilirubin, were also analyzed.
STATISTICAL ANALYSIS:
All data are presented as means with standard deviation or median with range, unless otherwise stated. Statistical analysis was performed with PRISM 8 for macOS (GraphPad Software, Inc. 2019) by using one-way ANOVA followed by a Holm-Sidak test for multiple comparisons and Fisher’s exact test for categorical variables. Kaplan-Meier method with log-rank tests was used for assessment of overall patient and graft survival. Graft survival was defined as graft failure and/or patient death. Thirty-day mortality was excluded for calculation of overall hospital and intensive care unit stay. For correlation analysis between DRI and graft survival, the calculated DRI score was classified into 4 risk groups (low risk (=I): DRI <1.2, moderate risk: 1.21> DRI <1.4, high-risk: 1.41> DRI <2, and very high-risk (=IV): DRI >2.1) as described previously [11] and was correlated with Kaplan-Meier graft survival. A P value <0.05 was considered to be statistically significant.
Results
RECIPIENTS:
PV-reperfusion was performed in 25, A-reperfusion in 22, and SIM-reperfusion in 14 recipients, resulting in a total of 61 patients. Mean recipient age at time of transplantation was 55 years. SIM-reperfused patients were significantly older (59 vs 55 (A) vs 49 PV, P=0.01). Additionally, the SIM-group had a higher pretransplant MELD (model for end-stage liver disease) score (22 vs 19 (A) vs 17 (PV)). The proportion of patients who underwent OLT because of hepatocellular carcinoma (HCC) was similar in all 3 groups: 8 (32%, PV) vs 8 (36%, A) vs 4 (29%, SIM). Table 1 gives an overview of recipient baseline clinical data.
DONOR DATA:
Table 2 gives an overview of the donor details, which were comparable between groups. Although it failed to reach statistical significance, donors in the SIM-group were the oldest (56 y) vs 51 y (PV) vs 50 y (A), P=0.37). According to the extended donor criteria (ECD) classification [12,13], 80% of the SIM-reperfused grafts were classified as marginal organs (>2 criteria), while only 36% of the donors in the other 2 groups met > 2 criteria (PV: 9 donors, A: 8 donors, P=0.02). However, no difference was observed regarding the DRI calculated according to Feng or the ET-DRI. There were 53 donors (93%) classified as high-risk or very high-risk donors.
OPERATIVE AND EARLY POSTOPERATIVE DATA:
Warm ischemia time was longer in the SIM-group (48 min vs 36 min (PV) and 29 (A) min,
Overall median hospital stay was similar between groups: 30 (PV) vs 30 (A) vs 28 days (SIM), P=0.99. Additionally, no difference was observed regarding length of stay in the intensive or intermediate care unit. With 7 deceased patients (28%), 60-day mortality was highest in the PV-group (0% (A) vs 14% (SIM), P=0.03). These 7 patients died because of septic complications (n=3), multi-organ failure (n=3), and acute heart failure (n=1). Moreover, one-fifth of patients in this group underwent early re-transplantation because of primary non-function (PNF, n=3) or early allograft dysfunction (EAD, n=2) within 30 days after initial OLT. None of the SIM-reperfused patients required re-transplantation, while the rate was 9% in the A-group (PNF: n=1, EAD: n=1; P=0.15). The incidence of postoperative complications such as bleeding or biliary complications were similar between groups (Table 3). Regarding postoperative peak values of liver enzymes, patients in the A-group showed the lowest AST or ALT peak values, while the values were the highest in the PV-group (GOT: P=0.01; GPT: P=0.03). There was no difference between groups in bilirubin or gGT peak values.
TWELVE-YEAR POST-LIVER TRANSPLANT MORBIDITY AND MORTALITY:
One-third of the study population was still alive at 12-year follow-up (n=21, 35%, Table 4). Although SIM-reperfused recipients were the oldest, overall mortality rates were the lowest in this group (50% vs 68% (A) vs 72% (PV). Mean overall survival for all recipients was 5.9 years. SIM-reperfused patients showed a tendency for longer survival compared to survival rates of the other groups (8.1 y vs 4.8 y (PV) vs 5.9 y (A), (Figure 1) according to Kaplan-Meier analysis (log-rank P=0.148). Re-transplantation was performed in 10 patients (16%). None of the SIM-reperfused recipients underwent re-transplantation in the 12 years after OLT, while re-transplantation was necessary in one-third of patients in the PV-group (5 patients within 30 days, as mentioned above, n=1 after 47 days, and n=1 after 7 years). Two patients in this group underwent 2 re-transplantations. Because of the re-transplantation rate of 0%, graft survival was equivalent to overall survival for SIM-reperfused patients, with 8.1 years, which was significantly longer compared to the PV- (3.3 y) and A-group (5.5 y, P=0.03) (Figure 1, Kaplan-Meier, P=0.013). In our study population, neither the DRI according to Feng nor the ET-DRI were correlated with graft survival.
We did not observe any statistically significant difference in incidence of biliary strictures, in need to perform an ERCP or in terms of absolute numbers of ERCPs. However, the proportion of patients that developed a biliary stricture was highest in the PV-group (28%). Furthermore, in this group, more patients needed rejection treatment compared to the other groups (32% vs 5% (A) vs 8% (SIM), although this also failed to reach statistical significance (
Discussion
In the era of organ shortage leading to utilization of ECD livers, strategies to improve outcome in OLT are of importance. Although OLT is a highly standardized procedure, there is so far no consensus on the best reperfusion technique. Within the ET region, portal vein reperfusion was recently identified as the most frequently used reperfusion technique. Interestingly, most centers based their decision solely on personal and institutional experience [2]. This might be explained by the fact that evidence is lacking in the literature, and, aggravating the situation, conflicting observations have been made in different studies. Therefore, we sought to investigate the clinical effect and outcome of 3 different reperfusion techniques with emphasis on 12-year long-term outcome. Our data show that despite higher age, less favorable MELD status, and higher number of marginal organs, SIM-reperfused patients demonstrated superior outcomes in terms of graft survival. Remarkably, none of these patients underwent re-transplantation, while re-transplantation was performed in one-third of recipients in the PV-group.
Concerns that prolonged warm ischemia time in SIM-reperfusion might have adverse effects on long-term outcome were not supported by the present results in our study population. Our results showed superior outcomes in terms of overall and graft survival compared to the PV- and A-groups. This observation may be attributed to attenuated reperfusion injury following simultaneous reperfusion compared to other reperfusion techniques, as demonstrated in an animal model. Despite a prolongation of warm ischemia of 10 min, simultaneous reperfusion caused the least reperfusion injury as demonstrated by histopathological findings and serum parameters and resulted in superior primary transplant function. On the contrary, arterial reperfusion showed the worst overall outcome among the 3 reperfusion techniques analyzed in 24 pigs [14]. Our observations regarding peak transaminases in the early postoperative course demonstrated significantly higher peak values in the PV-group compared to the other 2 groups, suggesting more reperfusion injury with PV-reperfusion. Analysis of peak transaminase following different reperfusion sequences in clinical setting is sparely described in the literature. A recently published network meta-analysis by Yao et al including 7 randomized controlled trials with 550 patients, showing that the lowest liver dysfunction rate (defined as occurrence of >1 enzymological aberration of the following: bilirubin > 10 mg/dl, INR >1.6 and ALT or AST >2000 U/ml within 7 days following OLT) was associated with the shortest ICU stay in patients with RVC [15]. Other studies that compared peak levels of liver enzymes on days 1–90 following OLT found no differences following either sequential or SIM-reperfusion [7,8,16].
Contradictory results can also be found regarding patient and graft survival. Polak et al analyzing outcome of 71 PV-reperfused recipients compared to 31 SIM-reperfused recipients did not observe advantages regarding overall 1-year survival among reperfusion techniques [16]. The meta-analysis by Yao et al identified RVC and SIM-reperfusion as superior techniques compared to the PV- or A-reperfusion technique. Regarding long-term outcome (1-year graft and patient survival), the RVC reperfusion technique resulted in highest graft survival followed by SIM-reperfusion technique, while it was the opposite for patient survival [15]. However, the meta-analysis only included one trial from one center that compared retrograde vs anterograde reperfusion (SIM) in 131 recipients [17]. Secondly, the authors of the meta-analysis themselves reported that the RVC reperfusion technique is an approach that is still under debate and “not widely accepted” because of prolonged ischemic time and biliary damage [15]. Therefore, the question of which reperfusion technique is superior regarding graft and patient survival has not yet been fully answered; however, data suggest that, in line with our findings, SIM-reperfusion is superior compared to PV- and A-reperfusion.
When evaluating outcomes after OLT, a point that has to be considered is occurrence of biliary complications, since the rationale for performing SIM-reperfusion is to decrease the risk of ischemic injury to the bile ducts, hypothesizing that biliary complications occur less frequently when compared to PV-reperfusion. In line with findings for graft and patient survival following different reperfusion sequences, as described above, data on the incidence of biliary complications also remain controversial, despite existing meta-analyses. While some authors found no difference in the rate of non-anastomotic biliary strictures or other complications between initial portal reperfusion vs SIM-reperfusion [16,18], others authors have found that SIM-reperfusion is superior to sequential reperfusion techniques [4,8,15,19–21]. The meta-analysis Wang et al, including 6 studies with a total of 467 patients, found a significantly lower rate of ischemic-type biliary lesions in the SIM-group vs the sequential reperfusion group (
In summary, the results from 4 meta-analyses addressing outcomes following OLT for different reperfusion sequences are available in the literature (3 of them only very recently published), illustrating the great importance of the subject for clinical routine. The authors of these meta-analyses observed either no difference regarding patient and graft survival between different revascularization approaches [18,20] or considered SIM-reperfusion to be superior to sequential PV- or A-reperfusion [4,15]. Therefore, evidence suggests that SIM-reperfusion is at least equivalent to, if not better than, the widely used PV-reperfusion technique, which should thus be critically reviewed. Furthermore, the results do not support concern of impaired outcomes because of prolonged warm ischemia time if SIM-reperfusion cannot be maintained. It is worth noting that most studies included in the meta-analyses report a long-term outcome of 1 year or even less. The maximum follow-up period of all studies was 2 years, described by Massarollo et al [19]. To the best of our knowledge, no data have been published for 12-year outcomes following OLT. In our study, we did not observe a difference in incidence of biliary strictures or in need to perform an ERCP as indicated by absolute numbers of ERCPs in the 12-year follow-up period. However, the proportion of patients that developed a biliary stricture was highest in the PV-group (28% vs 21% in the SIM-group).
There are some limitations of the current study that need consideration. First, the current study was a retrospective, single-center cohort study with all the inherent limitations, as well as a limited number of patients. Second, recipients were not randomized into the 3 reperfusion groups prior to surgery. These limitations might be countered by strengths such as performance of surgery by the same 2 senior surgeons, a comparable group size, and the long-term follow-up. Moreover, despite lacking randomization, marginal grafts were preferred to reperfuse simultaneously. This approach is reflected by the fact that 80% of the SIM-reperfused grafts met >2 ECD criteria, and in our study population, the calculated DRI, in contrast to findings from other studies [20–23], did not correlate with 12-year graft survival: despite a slightly higher ET-DRI calculated for the SIM-group compared to the other 2 groups, graft survival of these recipients was significantly prolonged. It has to be considered that for an appropriate prospective randomization, large study arms consisting of 300 patients are required, which is difficult to implement in a clinical setting. It also has to be mentioned that none of the PV-reperfused recipients had a portocaval shunt performed. However, results of a meta-analysis suggest that performance of a portocaval shunt does not impact primary graft function [24]; the adverse outcome of PV-reperfused recipients is thus unlikely to be solely due to the absence of a portocaval shunt.
Conclusions
Our data support the superiority of SIM-reperfusion over PV and A-reperfusion. Despite disadvantageous conditions such as higher (donor and recipient) age, slightly higher MELD scores, and higher number of marginal organs, these patients demonstrated superior 12-year outcomes in terms of overall and graft survival. Moreover, in contrast to the PV- and A-reperfusion groups, there was no need for re-transplantation in the follow-up period of 12 years. To confirm these findings, further multicenter randomized controlled trials with larger study populations are mandatory.
References
1. Bodzin AS, Baker TB, Liver transplantation today: Where we are now and where we are going: Liver Transpl, 2018; 24(10); 1470-75
2. Manzini G, Kremer M, Houben P, Reperfusion of liver graft during transplantation: Techniques used in transplant centres within Eurotransplant and meta-analysis of the literature: Transpl Int, 2013; 26(5); 508-16
3. Gurusamy KS, Naik P, Abu-Amara M, Techniques of flushing and reperfusion for liver transplantation: Cochrane Database Syst Rev, 2012(3); CD007512
4. Wang JZ, Liu Y, Wang JL, Sequential vs simultaneous revascularization in patients undergoing liver transplantation: A meta-analysis: World J Gastroenterol, 2015; 21(22); 7036-46
5. Polak WG, Porte RJ, The sequence of revascularization in liver transplantation: It does make a difference: Liver Transpl, 2006; 12(11); 1566-70
6. Bartlett A, Rela M, Heaton N, Reperfusion of the liver allograft with blue blood: is it still the royal perfusate?: Am J Transplant, 2007; 7(7); 1689-91
7. Baccarani U, Rossetto A, Lorenzin D, Protection of the intrahepatic biliary tree by contemporaneous portal and arterial reperfusion: Results of a prospective randomized pilot study: Updates Surg, 2012; 64(3); 173-77
8. Adani GL, Rossetto A, Lorenzin D, Sequential versus contemporaneous portal and arterial reperfusion during liver transplantation: Transplant Proc, 2011; 43(4); 1107-9
9. Feng S, Goodrich NP, Bragg-Gresham JL, Characteristics associated with liver graft failure. The concept of a donor risk index: Am J Transplant, 2006; 6(4); 783-90
10. Braat AE, Blok JJ, Putter H, The Eurotransplant donor risk index in liver transplantation: ET-DRI: Am J Transplant, 2012; 12(10); 2789-96
11. Schoening W, Helbig M, Buescher N, Eurotransplant donor-risk-index and recipient factors: Influence on long-term outcome after liver transplantation – A large single-center experience: Clin Transplant, 2016; 30(5); 508-17
12. Cameron AM, Ghobrial RM, Yersiz H, Optimal utilization of donor grafts with extended criteria: A single-center experience in over 1000 liver transplants: Ann Surg, 2006; 243(6); 748-53 discussion 753–55
13. Busuttil RW, Tanaka K, The utility of marginal donors in liver transplantation: Liver Transpl, 2003; 9(7); 651-63
14. Brockmann JG, August C, Wolters HH, Sequence of reperfusion influences ischemia/reperfusion injury and primary graft function following porcine liver transplantation: Liver Transpl, 2005; 11(10); 1214-22
15. Yao Y, Wu P, Guo T, Identifying the superior reperfusion technique in liver transplantation: A network meta-analysis: Gastroenterol Res Pract, 2019; 2019 9034263
16. Polak WG, Miyamoto S, Nemes BA, Sequential and simultaneous revascularization in adult orthotopic piggyback liver transplantation: Liver Transpl, 2005; 11(8); 934-40
17. Heidenhain C, Heise M, Jonas S, Retrograde reperfusion via vena cava lowers the risk of initial nonfunction but increases the risk of ischemic-type biliary lesions in liver transplantation – a randomized clinical trial: Transpl Int, 2006; 19(9); 738-48
18. Bekheit M, Catanzano M, Shand S, The role of graft reperfusion sequence in the development of non-anastomotic biliary strictures following orthotopic liver transplantation: A meta-analysis: Hepatobiliary Pancreat Dis Int, 2019; 18(1); 4-11
19. Massarollo PC, Mies S, Raia S, Simultaneous arterial and portal revascularization in liver transplantation: Transplant Proc, 1998; 30(6); 2883-84
20. Domagala P, Takagi K, Porte RJ, Polak WG, Order of liver graft revascularization in deceased liver transplantation: A systematic review and meta-analysis: Surgery, 2019; 166(3); 237-46
21. Sankary HN, McChesney L, Frye E, A simple modification in operative technique can reduce the incidence of nonanastomotic biliary strictures after orthotopic liver transplantation: Hepatology, 1995; 21(1); 63-69
22. Blok JJ, Putter H, Metselaar HJ, Identification and validation of the predictive capacity of risk factors and models in liver transplantation over time: Transplant Direct, 2018; 4(9); e382
23. de Boer JD, Putter H, Blok JJ, Predictive capacity of risk models in liver transplantation: Transplant Direct, 2019; 5(6); e457
24. Pratschke S, Rauch A, Albertsmeier M, Temporary intraoperative porto-caval shunts in piggy-back liver transplantation reduce intraoperative blood loss and improve postoperative transaminases and renal function. A meta-analysis: World J Surg, 2016; 40(12); 2988-98
Tables
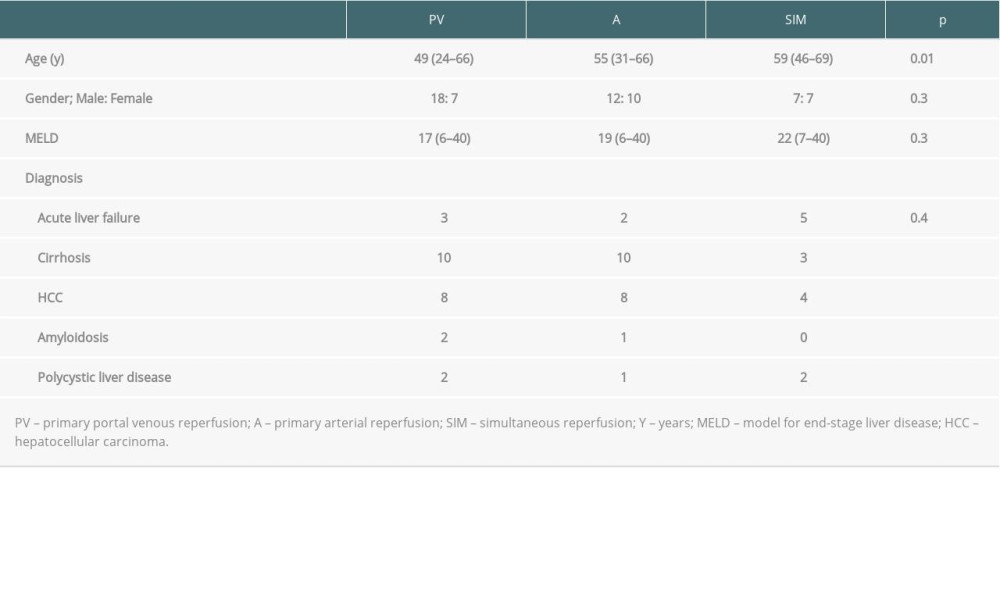 Table 1. Recipient data.
Table 1. Recipient data.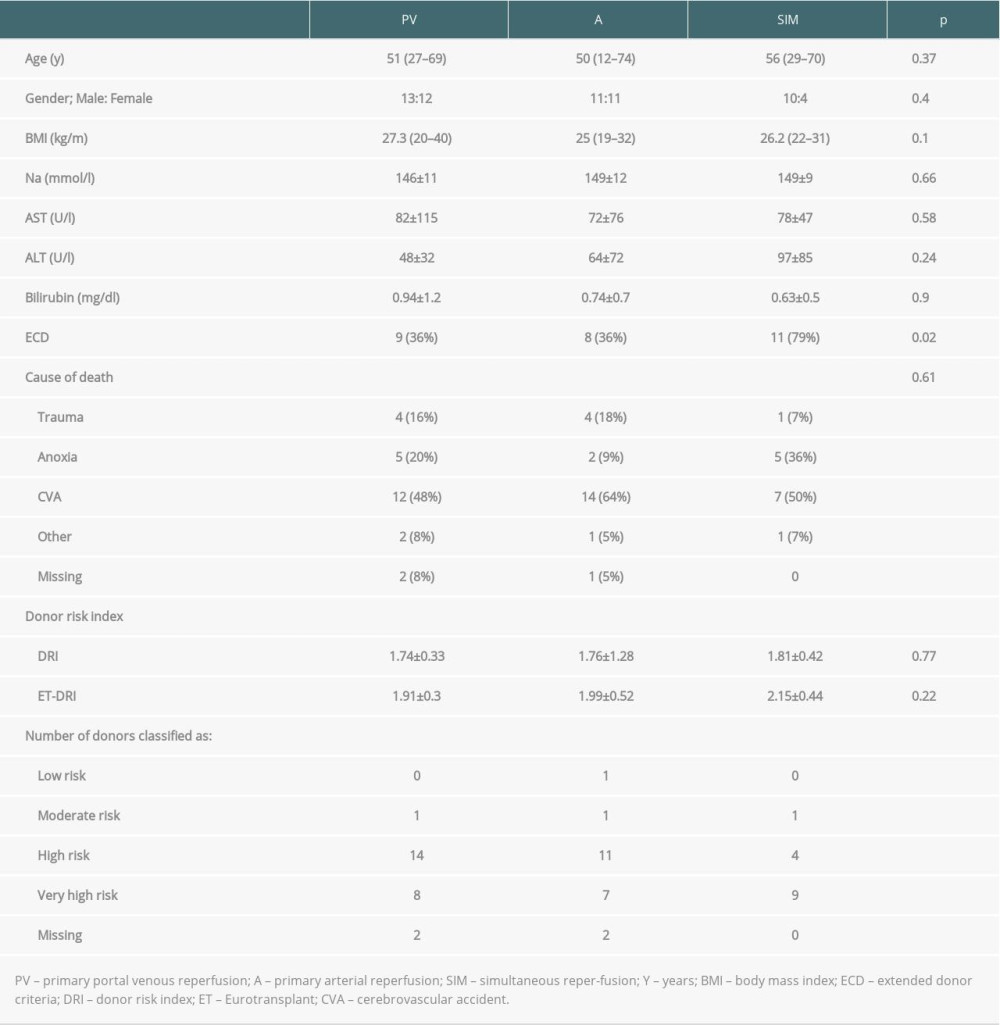 Table 2. Donor data.
Table 2. Donor data.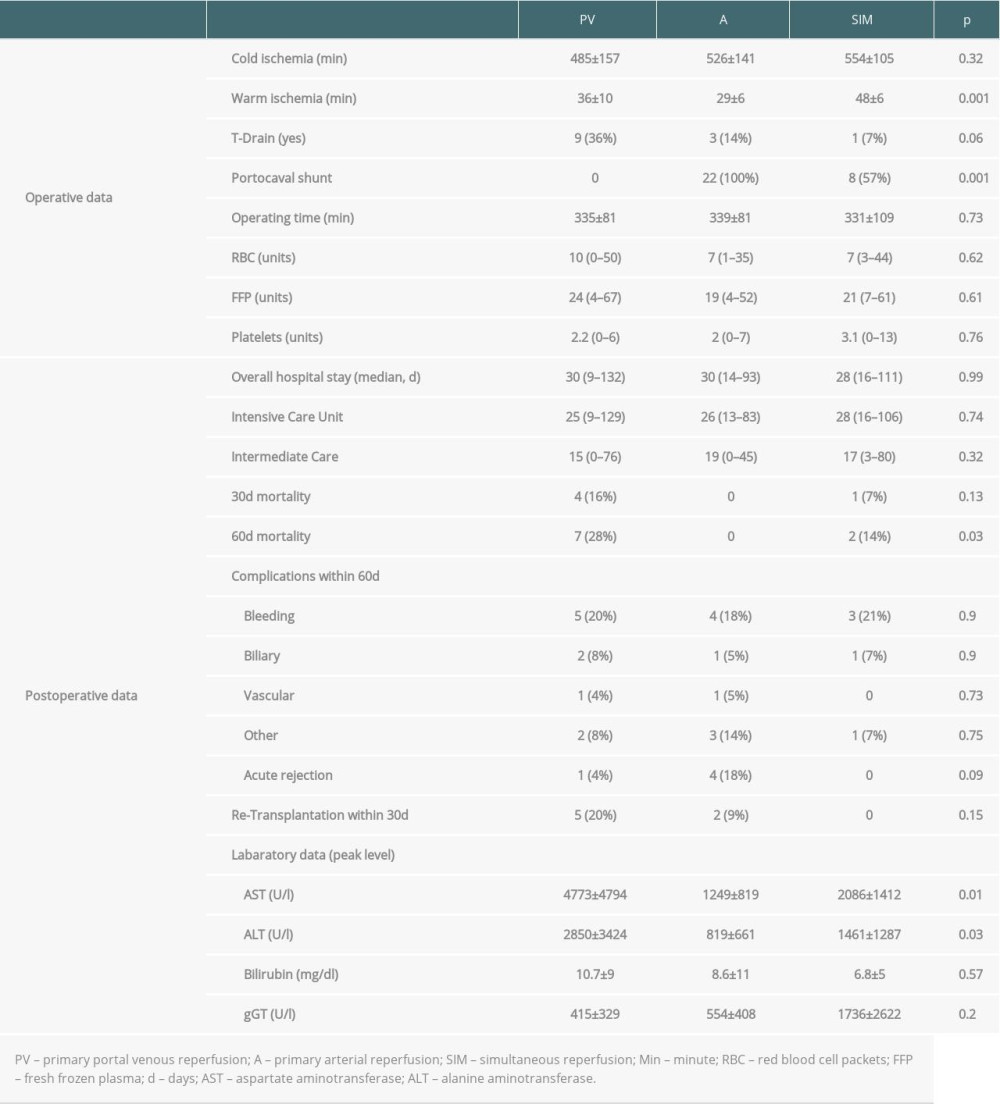 Table 3. Operative and (early) postoperative data.
Table 3. Operative and (early) postoperative data.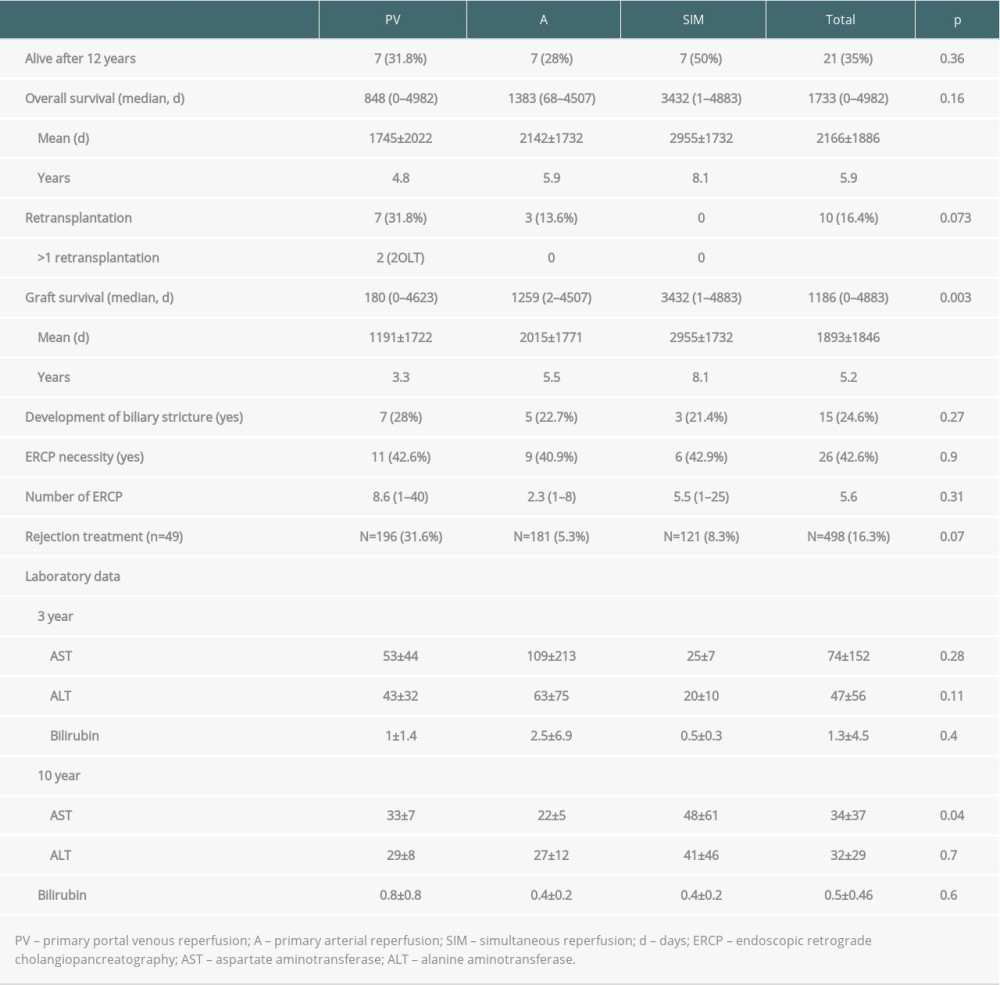 Table 4. 12-year post-liver transplant morbidity and mortality.
Table 4. 12-year post-liver transplant morbidity and mortality. Table 1. Recipient data.
Table 1. Recipient data. Table 2. Donor data.
Table 2. Donor data. Table 3. Operative and (early) postoperative data.
Table 3. Operative and (early) postoperative data. Table 4. 12-year post-liver transplant morbidity and mortality.
Table 4. 12-year post-liver transplant morbidity and mortality. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860