25 May 2021: Original Paper
Results from an International Survey of Donor and Recipient Eligibility for Solid Organ Pancreas Transplantation
Jonathan E.H. Ling12ABCDEF*, Kevan R. Polkinghorne123ACDE, John Kanellis12ADEDOI: 10.12659/AOT.930787
Ann Transplant 2021; 26:e930787
Abstract
BACKGROUND: Current solid organ pancreas transplantation protocols have differing donor criteria for donor pancreas acceptance and recipient eligibility criteria for transplant workup. We quantified this variation and compared current Australia and New Zealand (ANZ) solid pancreas transplant eligibility criteria with current international practice.
MATERIAL AND METHODS: A survey of donor and recipient eligibility criteria for solid pancreas transplantation was disseminated to 85 transplant units in 23 countries. Responses were grouped by regions (ANZ, North America, Eurotransplant, Europe, United Kingdom) and analyzed for significant differences between regions and for ANZ compared to all other regions.
RESULTS: Responding UK pancreas transplant units reported the highest mean donor upper age limit (61 years old) and the highest mean donation after cardiac death donor (DCD) age limit (55 years old). All responding UK and USA units utilized DCD pancreas donors and accepted suitable type 2 diabetes (T2DM) recipients for pancreas transplantation; however, this was less common among responding European or Eurotransplant units. ANZ mean standard and DCD pancreas donor upper age limits (47 and 35 years old, respectively) were lower compared to all other regions (54 years old and 48 years old, respectively).
CONCLUSIONS: Pancreas donor age limits, DCD pancreas donor utilization, and transplanting T2DM recipients differ between responding pancreas transplant units. ANZ units have more conservative donor upper age limits compared to other responding units. Increased utilization of DCD pancreas donors and T2DM recipients while standardizing pancreas donor age limits might increase donor numbers and improve access to solid pancreas transplantation both locally and abroad.
Keywords: Donor Selection, Eligibility Determination, Pancreas Transplantation, Resource Allocation, Transplantation, Australia, Diabetes Mellitus, Type 2, Graft Survival, New Zealand, Tissue Donors, Tissue and Organ Procurement
Background
Solid organ pancreas transplantation, usually performed in combination with a kidney transplant when renal failure is present, is the definitive treatment for medically suitable patients with type 1 diabetes [1]. Pancreas transplant recipients have improved mortality and quality of life compared to patients on the waiting list or who cannot be transplanted [1–3]. In Australia and New Zealand, annual solid pancreas transplantation and pancreas donations have increased over the last decade [4–8], while in other regions such as the United States of America (USA) and the United Kingdom (UK), transplant numbers have decreased or plateaued during the same period [7,9]. However, the total number of solid pancreas transplants performed per capita annually in Australia/New Zealand is much lower than in the USA or the UK (2 per million population vs 2.8 in the UK and 3.1 in USA in 2018) [4,5,7,8].
National pancreas transplant protocols guide donor organ acceptance as well as recipient assessment for transplant eligibility [10–14]. However, differences exist in donor and recipient criteria determining organ acceptance and eligibility for entering transplant workup, respectively, between pancreas transplant protocols for different jurisdictions (Table 1). Transplant volume can influence these differences, as demonstrated by a USA registry study reporting that low-volume pancreas transplant centers were accepting more ‘favorable’ pancreas donors with lower Pancreas Donor Risk Index (PDRI) [15] scores compared to high-volume centers [16]. Furthermore, varying eligibility criteria between regions might result in a pancreas transplant candidate or donor being considered high-risk in one jurisdiction but acceptable in another. We aimed to quantify these differences by region and to determine whether these differences were correlated with the transplant volume by region. We also compared Australia and New Zealand (ANZ) solid pancreas transplant criteria to those of similar units worldwide. We hypothesized that donor and recipient eligibility criteria for solid pancreas transplantation significantly differ between units as well as between countries and that local criteria are more conservative than those employed elsewhere.
Material and Methods
SURVEY DEVELOPMENT:
A survey on donor and recipient eligibility criteria for pancreas transplantation was disseminated to pancreas transplant units worldwide. The survey format was chosen due to its ability to rapidly obtain information from a targeted population at a low cost [17]. With guidance from the existing literature [18–20], Microsoft Excel and Qualtrics were used to design the survey, consisting of 4 domains (Unit demographics, Recipient criteria, Donor criteria, and Other criteria) (Supplementary Table 1).
Unit demographics requested included the number of solid pancreas transplants performed between 1 January 2017 and 31 December 2017 stratified by transplant type: whether simultaneous pancreas-kidney (SPK) transplant, pancreas after kidney (PAK) transplant, or pancreas transplant alone (PTA). Recipient criteria questions included upper age limit, body mass index (BMI) limits, whether type 2 diabetic (T2DM) recipients were accepted, criteria regarding type 1 diabetes severity, smoking history, alcohol intake, recreational drug use, and any criteria regarding severity of peripheral vascular disease or cardiovascular disease. Donor criteria questions also included age and BMI limits, whether donation after cardiac death (DCD) donors were accepted, and whether specific criteria on age, BMI, and ischemic time existed for DCD donors, as well as any use of donor risk indices to guide donor pancreas acceptance. Other criteria questions included if any logistical factors would impede pancreas transplantation. Questions regarding immunological matching criteria were excluded, as their use depended on existing infrastructure and funding between various regions. Units could link national/regional protocols influencing unit-specific protocols. The survey (designed to be completed within 30 minutes) was piloted internally on 2 occasions by 2 nephrologists with experience in pancreas-kidney transplantation with no changes made after the piloting stage. The project was approved by the Monash Health Human Research Ethics Committee as a quality and service improvement project without ethical concerns.
Purposive sampling [21] and snowball sampling [22] were employed to identify the intended cohort. Contact details for 134 pancreas transplant units worldwide were identified via directory-searching by country (ie, the Scientific Registry of Transplant Recipients directory of pancreas transplant units in the United States of America [USA]), as well as personal correspondence with unit directors abroad via direct contact with email and telephone. Due to the number of pancreas transplant units in the USA (n=143), only units that had performed more than 10 solid pancreas transplants in 2017 were contacted (n=26). However, contact details were incorrect or outdated for 49 units and further attempts to obtain contact details for these units were unsuccessful. In total, the survey was distributed via email to the unit directors of 85 pancreas transplant units worldwide: Australia, Austria, Belgium, Canada, the Czech Republic, France, Germany, Hungary, Ireland, Israel, Italy, the Netherlands, New Zealand, Norway, Poland, Portugal, Romania, Russia, Spain, Sweden, Switzerland, Turkey, the United Kingdom (UK), and the USA. An email reminder was sent 1 month after the initial invitation to participate.
DATA ANALYSIS:
Units were de-identified and grouped by regions to allow basic statistical analysis. Differences between continuous donor and recipient variables by region were analyzed via one-way analysis of variance with Bonferroni correction, while Fisher’s exact test was used for categorical variables with binary outcomes. We aggregated the mean transplants by number of units per region to derive the transplant volume for each region. This represented the differences in transplant number by region. We then analyzed the data for associations between donor and recipient eligibility criteria and transplant volume (as defined above) via linear regression analysis. Differences in eligibility criteria between countries was examined for units in the Eurotransplant (6 units), the rest of Europe (5 units), UK (4 units), North America (3 units), and ANZ (3 units) based on existing pancreas transplant guidelines (Supplementary Tables 2, 3).
Results from Australia and New Zealand were also analyzed as a single region against all other regions.
Results
SURVEY RESPONSE:
From 85 pancreas transplant units across 23 countries, 21 units (24.7%) in 15 countries responded: Australia, Austria, Belgium, Canada, Germany, Hungary, Israel, Italy, the Netherlands, Norway, New Zealand, Spain, Sweden, UK, and USA. All 3 Australia/New Zealand units responded, as did 2 of 11 USA units, 4 of 7 UK units, and 1 of 7 Canadian units. Units were divided into 5 regions: North America (Canada and USA), UK, Europe (Israel, Italy, Norway, Spain, Sweden), Eurotransplant (Austria, Belgium, Germany, Hungary, the Netherlands), and ANZ (Australia and New Zealand) (Table 1).
Transplant volume as defined above was the highest in North America (45 transplants per unit) in 2017, followed by UK (25), ANZ (17), Eurotransplant (9), and Europe (7). SPK was the most common transplant type performed, followed by PTA and PAK (Table 1). Six units linked to national pancreas protocols (4 from UK and 2 from Eurotransplant). Transplant volume was only significantly different between North America, Eurotransplant, and Europe regions (45 vs 9.2 vs 8 transplants/unit, respectively) (Table 2). When analyzed by transplant type, North American units performed more PTA transplants per unit compared to other regions (P<0.05). No difference was seen in SPK or PAK transplants by region.
DONOR CRITERIA:
All responding units reported a donor upper age limit to guide pancreas acceptance; the commonest being 55 years (6/21 units, 29%). Other reported upper age limits were 45 years (5 units), 50 years (5 units), 60 years (4 units), and 65 years (1 unit). UK units reported the highest mean donor upper age limit (61.3±2.5 years) compared to North America (50±8.7 years), Eurotransplant (51.7±4.1 years), and ANZ (46.7±2.9 years). Donor upper age limits significantly differed (P<0.05) between UK and all regions apart from Europe (Table 2).
Seventeen of 21 (81%) responding units reported a donor upper BMI limit; the commonest being 30 kg/m2 (10 of 17 units, 59%). Other donor upper BMI limits ranged from 32 kg/m2 (1 unit) and 33 kg/m2 (1 unit) to 35 kg/m2 (3 units). Two units did not specify a donor upper BMI limit despite stating donor BMI was part of the unit protocol. No significant difference was observed between regions for the mean donor upper BMI limit (Table 2).
Twelve of 21 units (57%) accepted DCD pancreas donors. All responding units from North America and the UK accepted suitable DCD donors, in contrast to Europe (2 of 6 units) and Eurotransplant (1 of 5 units). Within ANZ, only Australian units accepted DCD donors for solid pancreas transplantation. Acceptance of DCD donors significantly differed by region (P=0.047) (Table 2).
Within units utilizing DCD pancreas donors, the DCD donor upper age limit ranged from 35 years (2 units), 40 years (3 units), 45 years (2 units), and 50 years (3 units) to 55 years (2 units) (Table 2). Between regions, UK units reported the highest mean DCD donor upper age limit (55±4.1 years) compared to North America (43.3±2.9 years, p=0.017), Eurotransplant (40 years, p=0.008), and ANZ (35 years, P=0.001) (Table 2). DCD donor upper age limit was not significantly different between UK and Europe (45 years, P=0.23) (Table 2).
All units accepting DCD donors reported a DCD donor upper BMI limit of 30 kg/m2. The commonest time limits for DCD donor warm ischemic time (WIT) and cold ischemic time (CIT) were 30 minutes (8 of 12 units, 67%) and 12 hours (5 of 12 units, 42%), respectively. Other WIT limits ranged from 60 minutes (2 units), while 2 other units did not specify a maximum time limit. Other CIT limits ranged from 8 hours (1 unit), 10 hours (2 units), 11 hours (1 unit), and 18 hours (1 unit) to 20 hours (1 unit). Only 1 unit utilizing DCD donors did not report a CIT time limit. For units accepting DCD donors, 9 of 12 units (75%) stated that the retrieval of other organs from the same donor would not affect donor pancreas acceptance. No significant differences were observed between regions for DCD donor BMI/WIT/CIT limits (Table 2).
Only 4 of 21 units used a risk index (Pre-procurement Pancreas Suitability Score) to guide donor pancreas acceptance [23]. Finally, no association was observed between transplant volume by region and limits for donor age/BMI, or DCD donor age/WIT/CIT (Table 3).
RECIPIENT CRITERIA:
Seventeen of 21 units (81%) reported a recipient upper age limit for entering solid pancreas transplant workup; the commonest being 60 years (7 of 17 units, 41%). Other recipient upper age limits reported were 50 years (2 units), 55 years (6 units) and 65 years (1 unit). Regarding recipient BMI, 10 units reported upper BMI cut-offs of 30 kg/m2 and 10 other units reported upper BMI cut-offs between 30 to 35 kg/m2 (1 unit did not specify a limit). No responding units used donor-recipient age-matching protocols. No significant difference was observed between regions for upper limits of recipient age or BMI (Table 2). Similarly, no association was observed between transplant volume by region with recipient age or BMI limits (Table 3).
Acceptance of T2DM recipients for solid pancreas transplantation differed significantly between region (P=0.028) (Table 2). Eleven units (52.3%) performed solid pancreas transplantation in T2DM patients (all responding USA and UK units, 2 units each from Europe and Eurotransplant regions). The commonest reported endocrine criteria guiding acceptance of T2DM patients for pancreas transplant workup was insulin dependence (9 of 11 units, 81%). Six units further defined daily insulin limits, of which the commonest cut-off for failing eligibility was ≥1 unit/kg/day. Six of 11 units (55%) performed C-peptide testing, but only 2 units defined C-peptide cut-offs (<0.01 and <5 ng/ml respectively). Four of 11 units (36%) included BMI assessment as part of their endocrine criteria; however, only 2 units further defined an upper BMI limit (≤30 kg/m2).
The commonest endocrine criteria guiding acceptance of a type 1 diabetic patient for transplant workup was C-peptide levels (ranging from absent levels to <0.8 ng/ml) (9 units), followed by quantification of insulin burden (5 units), autoantibody levels, and measures of hypoglycemic severity such as hypoglycemic unawareness (3 units). Regarding patient referral for pancreas transplant workup, 16 of 21 units (76%) received referrals before patients had entered chronic kidney disease stage V (eGFR ≤15 ml/min).
Current smoking status was considered as a relative contraindication for solid pancreas transplant workup by 86% of units. All units considered excessive alcohol intake and ongoing illicit drug use as relative contraindications for transplant workup. Abstinence before transplant workup for patients with illicit drug use was required by 71% of units, but only 50% of units considered abstinence necessary in current smokers and by 38% of units for patients with excessive alcohol intake. Referral to drug and alcohol services or psychologists was indicated by 62% of units for illicit drug use, 56% of units for smoking, and 43% of units for excessive alcohol abuse.
Eighteen of 21 units (86%) reported criteria governing acceptable cardiac and vascular comorbidities to guide acceptance for transplant workup. Specifically, 11 units reported a stratified approach using cardiac stress testing, with positive results proceeding to coronary angiogram and intervention as necessary. Left ventricular ejection fraction lower limits of 40% (2 units) and 30% (1 unit) were described as relative contraindications. Four units cited “non-improvable coronary artery disease” as an absolute contraindication. Three units reported that they would accept cardiology recommendations regarding solid pancreas transplant candidacy.
Regarding vascular criteria, 5 units reported limitations based on degree of iliac artery stenosis not amenable to revascularization as contraindications for pancreas transplant workup (up to 50% in 2 units and 60% in 3 units). As an absolute contraindication, 5 units cited “the absence of clinically significant peripheral vascular disease allowing anastomosis”, while 2 units cited “non-improvable peripheral vascular disease”. Three units reported they would accept a vascular surgeon’s recommendations regarding transplant candidacy. Two units reported that any history of peripheral vascular disease requiring revascularization would be an absolute contraindication for solid pancreas transplantation.
OTHER CRITERIA:
Eight of 21 units (38%) stated that geographical factors would affect decisions to proceed with pancreas transplantation. Of these, 6 units (75%) stated associated concerns regarding CIT. One unit reported ‘multiple factors’ without further elaboration. Another unit specified an important factor was ensuring the surgeons explanting the donor organs were from the same transplant hospital. Fifteen of 21 units (71%) stated that logistical factors would not affect the decision to proceed with pancreas transplantation. Logistical factors affected transplantation decisions in 5 units (unavailability of vascular surgeons in 4 units and unavailability of intensive care beds post-operatively in 1 unit). One unit reported multiple factors (transport costs, donor team availability) would influence their decision to proceed with solid pancreas transplantation.
ANZ AGAINST ALL REGIONS:
The mean DCD donor upper age limit was lower in ANZ than in all other regions (35 years vs 47.5±7.2 years respectively, P=0.001) (Table 4). The mean donor upper age limit was also lower in ANZ than in all other regions (46.7±2.9 years vs 53.9±5.8 years respectively, P=0.052). The difference between the mean recipient upper age limit also trended toward significance (58.3±4.1 for ANZ vs 52.5±3.5, P=0.075). Other donor and recipient characteristics were not significantly different between ANZ and all other regions.
VARIATION BETWEEN UNITS WITHIN REGIONS:
Responding UK, Eurotransplant and ANZ units reported recipient and donor pancreas age limits consistent with their national protocols [10,11,13,14]. Varying recipient and donor upper age limits were reported for the USA units (55–65 years and 45–65 years, respectively), consistent with the lack of formal recipient and donor age limits within the USA pancreas protocol [12]. Reported recipient and donor BMI limits were consistent between units (30–35 kg/m2).
All responding UK and USA units accepted suitable T2DM recipients for transplantation, while only a minority of responding Eurotransplant (2 of 5), other European units (2 of 6), and none of the ANZ units did so. All responding UK and USA units also accepted DCD pancreas donors, but this was less common among responding Eurotransplant (1 of 5), other European (2 of 6), and ANZ (2 of 3) units. DCD pancreas donor age ranged from 50 to 60 years old in responding UK units and from 40 to 50 years old in Eurotransplant units. Comparatively, responding units within ANZ and USA had more uniform DCD donor upper age limits (35 years and 45 years old, respectively).
VARIATION BETWEEN NATIONAL PANCREAS PROTOCOLS:
Transplant eligibility criteria for solid pancreas recipients and donor pancreas acceptance for the Eurotransplant [10], UK [13], and the USA [12] were compared with the Transplantation Society of Australia and New Zealand (TSANZ) criteria [14] (Supplementary Tables 2, 3). The Eurotransplant, UK, and USA do not have formal recipient upper age limits for solid pancreas transplantation. Eurotransplant and the USA do not have a recipient BMI upper limit, while this is 30 kg/m2 for the UK. Regarding diabetes classification, the Eurotransplant and UK criteria stipulates autoantibody and C-peptide testing in selecting suitable pancreas transplant candidates, while USA criteria broadly define suitable recipients as those demonstrating pancreatic exocrine insufficiency. Only UK criteria contain relative and absolute contraindications for pancreas transplantation based on recipient comorbidities as well as addressing other risk-taking behavior such as drug or alcohol abuse. In comparison, the TSANZ pancreas recipient criteria state an upper age limit of 50 years, a BMI limit of under 35 kg/m2, and also suggests C-peptide testing to delineate the presence of low or absent insulin production. TSANZ criteria also comment on the risk of significant cardiovascular and peripheral vascular disease and risk-taking behavior in the form of smoking and illicit drug use.
Regarding donor criteria for solid pancreas acceptance, the upper donor age limit is 50 years old from Eurotransplant criteria (up to 60 years old in the Netherlands), while the UK criteria accept donation after brain death (DBD) donors under 66 years old and DCD donors under 56 years old for solid pancreas transplantation. Donor BMI criteria range from 25 kg/m2 (UK) to 30 kg/m2 (Eurotransplant). The USA does not have formal donor pancreas age or BMI limits but allocate donors above 50 years old and above 30 kg/m2 differently to younger and lighter donors. In comparison, TSANZ donor pancreas criteria stipulate a 45-year age limit, a donor weight between 25 to 100 kg, and specific DCD donor pancreas criteria (Supplementary Table 2).
Discussion
This is the first study to report differences in donor and recipient criteria for solid pancreas transplant eligibility between units from various countries with comparisons to current criteria in Australia and New Zealand. The main differences between regions were pancreas donor and DCD donor upper age limits and the acceptance of DCD pancreas donors and T2DM recipients for solid pancreas transplantation. There are a few reasons for these differences.
Firstly, differences between local pancreas allocation protocols contribute to the differences in unit responses observed (Supplementary Tables 2, 3). For instance, Eurotransplant guidelines defines pancreas donors >50 years old and with BMI ≥30 kg/m2 as so-called extended-criteria donors [10], with the Netherlands extending the DBD donor upper age limit to 60 years old. UK criteria also have a donor upper age limit of up to 60 years for DBD donors. However, ANZ guidelines would not accept pancreas donors >45 years old [14]. Also, the presence of unique mechanisms such as donor-recipient age-matching, which is built into the UK pancreas allocation scheme [11], may have contributed to the high mean donor (and DCD donor) age limit reported by responding UK units, allowing selected older recipients to receive older donor pancreata.
Secondly, individual pancreas units are likely to assess transplant candidates on a case-by-case basis, using existing guidelines as relative rather than absolute contraindications for transplantation. Units reported slightly higher recipient and donor upper age limits and upper BMI limits than their corresponding national pancreas transplant guidelines (Supplementary Tables 2, 3). For instance, Eurotransplant units reported a donor upper age limit of 45 to 55 years old despite the Eurotransplant guidelines recommending a donor upper age limit of 50 years old (with donors over 50 considered as meeting extended criteria) [10]. Our unit has previously transplanted patients up to 55 years old, although solid pancreas transplants in patients above 50 years of age are relatively rare. Transplant caseload in individual centers and available expertise may also influence these differences [16], although no significant associations between transplant volume by region and donor/recipient age and BMI were noted in our results.
The varying acceptance of T2DM patients for solid pancreas transplantation is due to the difficulty in classifying diabetes type (and assessing transplant benefit) [24,28,29]. Solid pancreas transplantation has been avoided in the setting of insulin resistance, leading to the use of C-peptide testing as a surrogate for insulin synthesis [25]. High C-peptide levels are not usually be observed in type 1 diabetic patients (with little to no insulin production) [25]. However, retrospective reviews of solid pancreas recipients with diabetes classified by C-peptide levels showed no significant difference in graft survival by diabetes type at 5–10 years, although sample sizes were generally small [26–28]. Furthermore, when diabetes classification was based on a combination of autoantibodies, C-peptide measurements, and absence of ketoacidosis, some type 1 diabetic patients still had detectable C-peptide levels, while some T2DM patients had low C-peptide levels <0.8 ng/ml [29], which was used in other studies to classify type 1 diabetic patients [27,28]. Therefore, C-peptide should be used in combination with clinical and biochemical criteria, as C-peptide alone may not correctly classify diabetes type [30] or predict transplant outcomes [26,27]. This correlates with our findings, in which the top reported criteria for assessing T2DM recipients for solid pancreas transplantation included C-peptide levels, insulin dependence, and recipient BMI.
Despite previous findings [24,31], there is evidence that including older donors and recipients results in non-inferior solid pancreas transplant outcomes [32,33]. A retrospective study showed non-inferior outcomes at 5 years after transplant between solid pancreas recipients from different age groups (from <30 to ≥60 years old) [34]. Similarly, a Canadian study found no difference in pancreas/kidney or patient survival between recipients ≥55 years and recipients <55 years old [35]. The Extended Pancreas Donor Program study showed similar 1-year pancreas survival between a cohort transplanted with extended criteria organs (donors aged 50–60 years) and a standard criteria donor cohort [36]. This implies that carefully selected older recipients (≥55 years old) and donors (>50 years old) have non-inferior solid pancreas transplant outcomes compared to standard criteria donors and recipients. Also, a recent systematic review reported non-inferior outcomes up to 10 years with DCD donors (compared to DBD) for solid pancreas transplantation [37].
Despite the differences in the criteria presented above, some similarities exist among pancreas transplanting units. Recipient and donor BMI limits were uniform within most responding pancreas transplant units. C-peptide and autoantibody testing were commonly used measures for identifying suitable type 1 diabetic candidates for transplantation throughout responding units. Smoking, excessive alcohol intake, and illicit drug use were relative contraindications for pancreas transplantation; however, most units did not require total abstinence from such risk-taking behavior to determine eligibility for solid pancreas transplantation (with the exception of illicit drug use). Most units did not use donor risk indices to guide donor pancreas acceptance.
A limitation of the present study was the relatively low response rate. Anticipating this, we contacted department heads directly via telephone and email to introduce the survey prior to sending the initial survey invitation, followed by a reminder 1 month later. The survey response rate, while low, is similar to that of other survey-based organ transplant research [38,39]. Another limitation was that data consisted of pre-specified and free-text responses, which varied in details. For example, responses to cardiac and vascular criteria such as ‘stable coronary artery disease’ or ‘absence of clinically relevant peripheral vasculopathy’ limited our analysis. Also, responses pertaining to extended donor criteria were also excluded from the analysis, as the definition of an extended criteria donor differs between units, making comparison difficult. These limitations are due to the survey format; however, this was a low-cost method to contact a large cross-section of the international pancreas transplant community relatively quickly. We also acknowledge that the variability in pancreas transplant eligibility criteria worldwide involves more factors than those included in this survey, such as human leukocyte antigen (HLA) matching, which is included in USA and UK pancreas allocation protocols [11–13], as well as institutional factors such as availability of donor retrieval services. However, capturing such data was beyond the scope of our project.
Given the response rate, we acknowledge the results may not be fully representative of worldwide practice. However, the existing variation demonstrated warrants further research into determining optimal pancreas donor and recipient eligibility criteria for solid pancreas transplantation. Certainly, despite the differences in eligibility criteria, pancreas transplant outcomes are fairly similar between jurisdictions. UK data from 2018/2019 reported 1- and 5-year pancreas graft survival at 90% and 81%, respectively, similar to ANZ data from 2018 showing 1- and 5-year pancreas graft survival of 90% and 85%, respectively [9,40]. One-year and 5-year patient survival was 98% and 89%, respectively, from UK data and 96% and 92%, respectively, from ANZ data during the same period [9,40]. Similarly, 1- and 5-year patient survival for SPK transplants was 97.6% and 91.9%, respectively from USA data [7]. Standardizing eligibility criteria would potentially reduce non-utilization of donor organs as well as increasing wait-listing of recipients previously felt to be too high-risk for transplantation. Indeed, in ANZ, donor age is currently the second most common reason for donor pancreas non-retrieval (21% of non-retrieved pancreata in 2018), following the donor being ‘not medically suitable’ [4]. However, such changes in donor/recipient eligibility criteria need to consider other factors, such as the current waiting time and mortality on the solid pancreas waiting list (as an alternative to transplanting with older donors), and ensuring that other donor factors are accounted for such that the overall pancreas donor quality remains acceptable.
Conclusions
Solid pancreas transplant units in the UK and USA more commonly utilize DCD donors and transplant suitable T2DM recipients compared to European and ANZ units. DCD and standard donor upper age limits for solid pancreas transplantation were significantly higher in the UK compared to the other regions represented in this study. ANZ solid pancreas transplant units reported lower DCD and standard donor upper age limits compared to other regions. Transplanting T2DM patients remains controversial due to the phenotypic overlap and current limitations in differentiating between diabetes types. Given the current variation in pancreas donor and recipient eligibility worldwide, more research into optimal criteria defining eligible solid pancreas donors and recipients (both locally and abroad) is needed to minimize non-utilization of potentially usable donors and to increase the number of wait-listed recipients. This also would better inform potential future changes in the ANZ solid pancreas transplant protocol to reflect worldwide practice.
Tables
Table 1. Responding units by region with number of SOPTx by transplant type performed within the 2017 calendar year.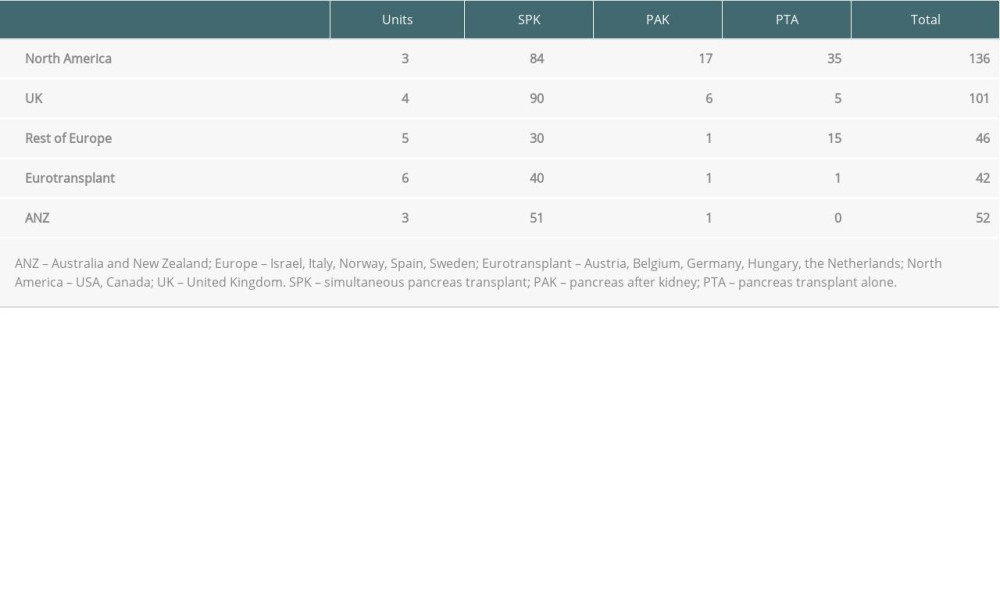 Table 2. Donor and recipient characteristics between all regions.
Table 2. Donor and recipient characteristics between all regions.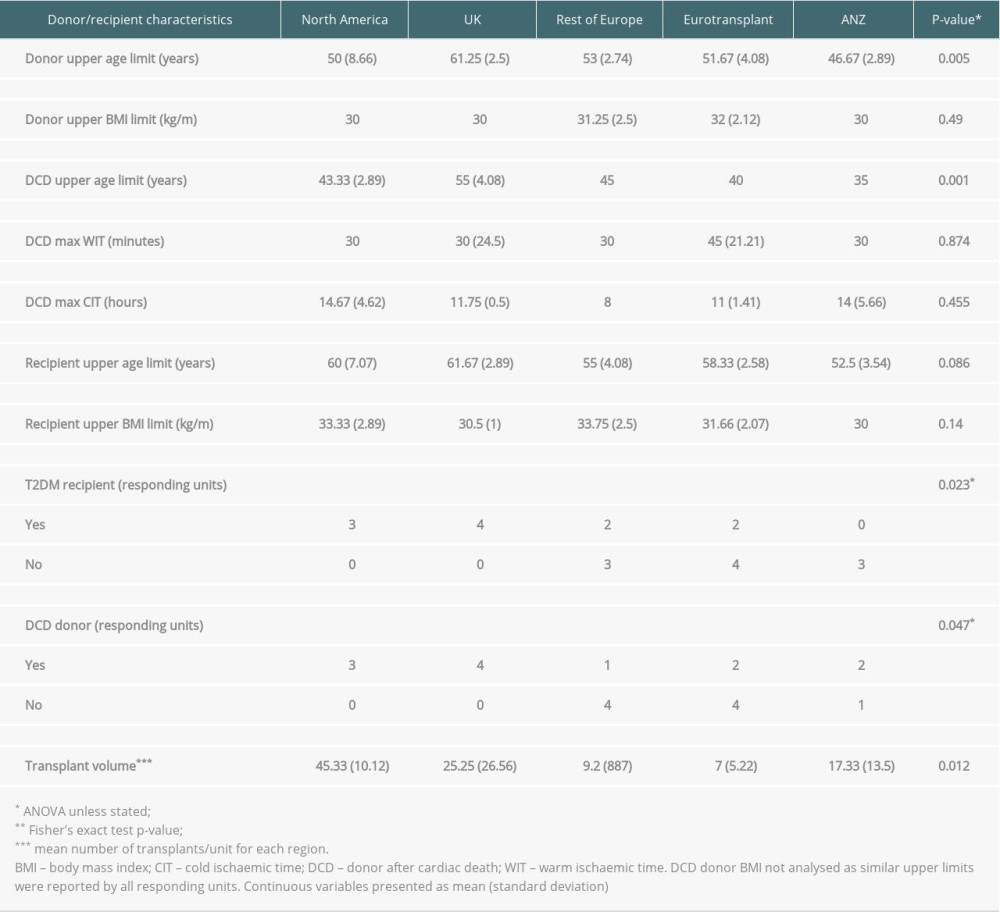 Table 3. Donor and recipient characteristics by transplant volume via linear regression analysis.
Table 3. Donor and recipient characteristics by transplant volume via linear regression analysis.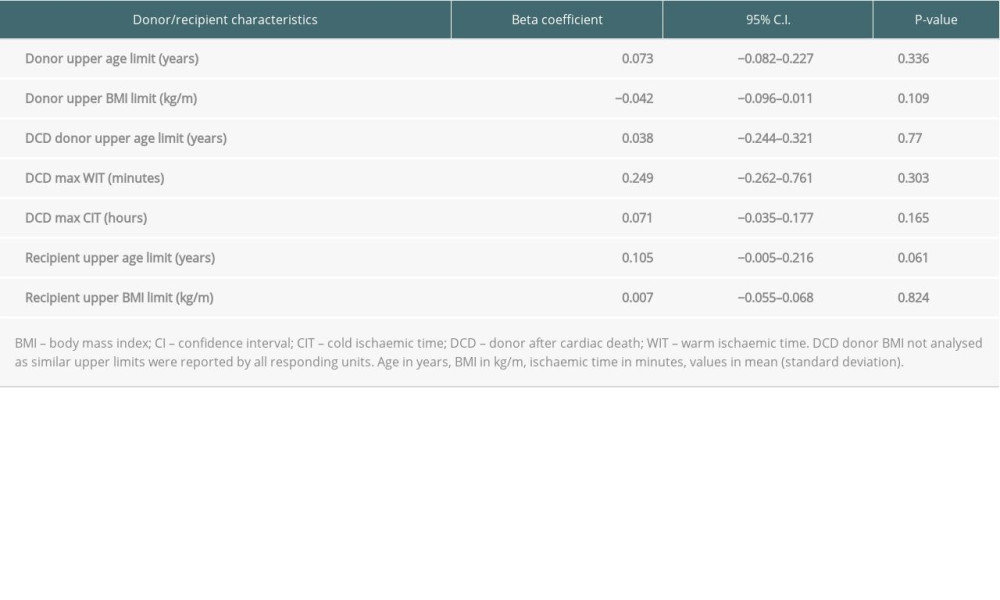 Table 4. Comparison of donor and recipient characteristics between ANZ and all other responding units.
Table 4. Comparison of donor and recipient characteristics between ANZ and all other responding units.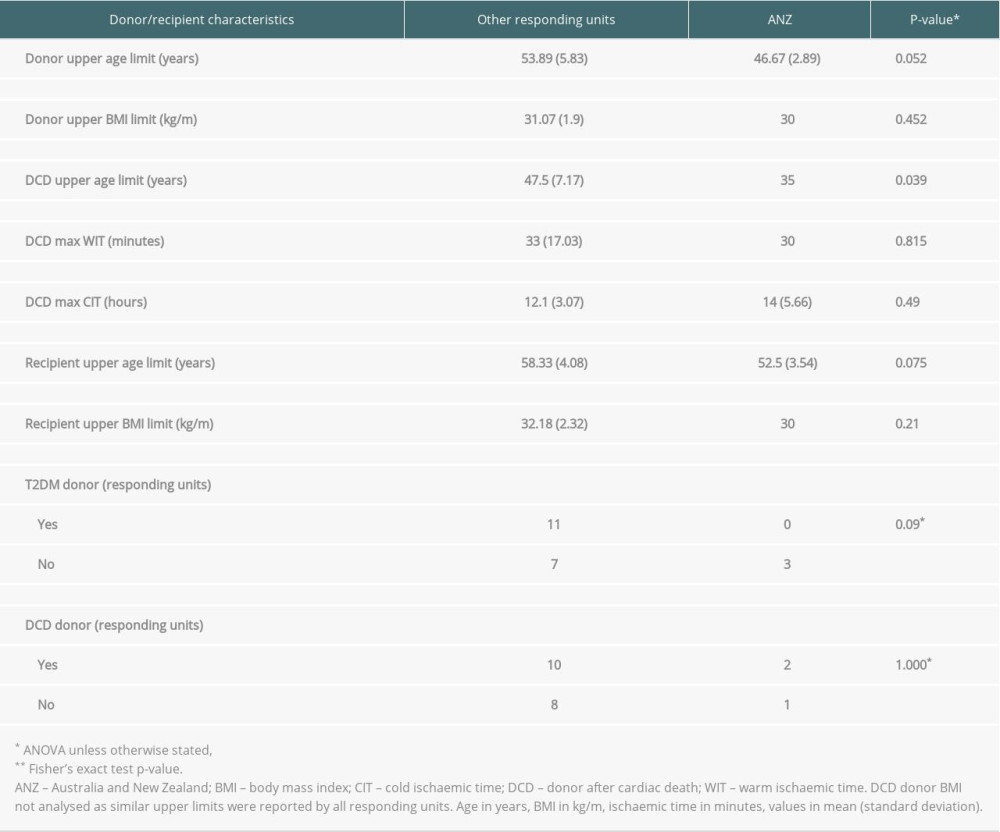 Supplementary Table 1. Survey questions.
Supplementary Table 1. Survey questions. Supplementary Table 2. Donor criteria for pancreas acceptance for solid pancreas transplantation according to current national pancreas protocols.
Supplementary Table 2. Donor criteria for pancreas acceptance for solid pancreas transplantation according to current national pancreas protocols.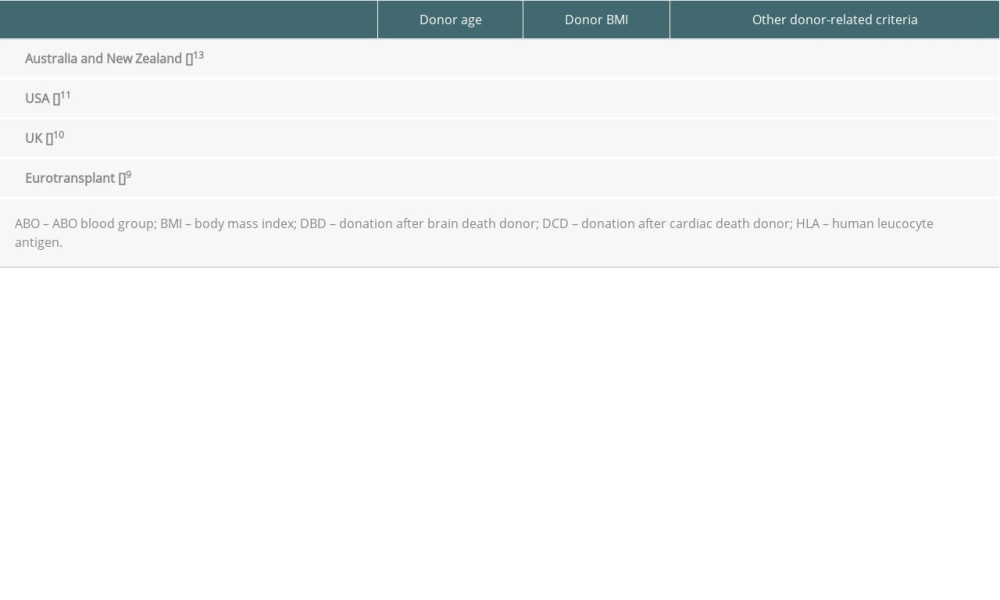 Supplementary Table 3. Recipient eligibility criteria for solid pancreas transplantation by current national pancreas protocols.
Supplementary Table 3. Recipient eligibility criteria for solid pancreas transplantation by current national pancreas protocols.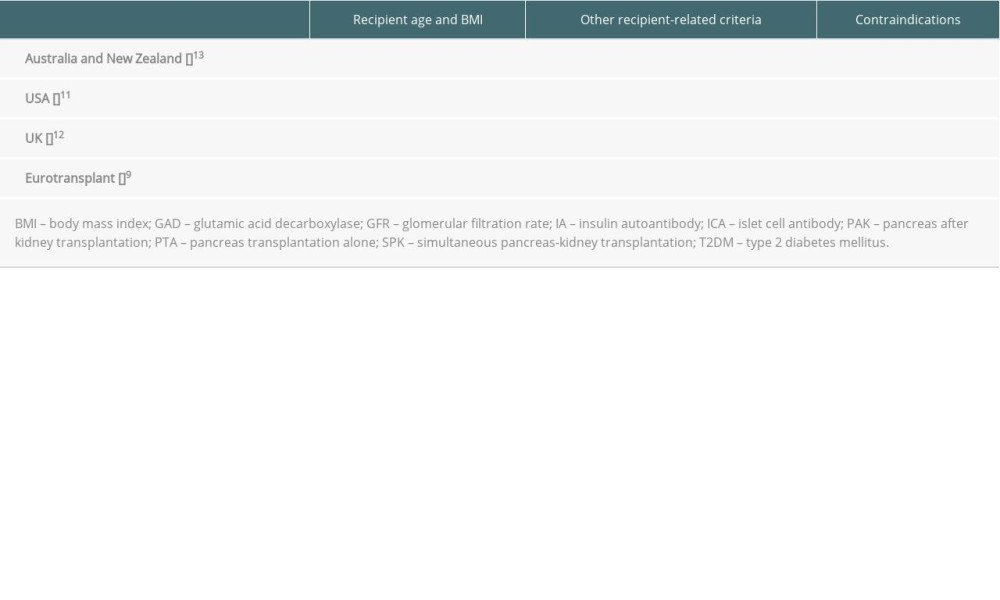
References
1. Mittal S, Gough SC, Pancreas transplantation: A treatment option for people with diabetes: Diabet Med, 2014; 31(5); 512-21
2. Tydén G, Tollemar J, Bolinder J, Combined pancreas and kidney transplantation improves survival in patients with end-stage diabetic nephropathy: Clin Transplant, 2000; 14(5); 505-8
3. Speight J, Reaney MD, Woodcock AJ, Patient-reported outcomes following islet cell or pancreas transplantation (alone or after kidney) in Type 1 diabetes: A systematic review: Diabet Med, 2010; 27(7); 812-22
4. ANZOD, 2018 Annual Report, Section 9: Deceased donor pancreas donation: Australia and New Zealand Dialysis and Transplant Registry, 2018
5. Webster ARP, Mark T, Hedley J, Kelly P, Chapter 2: Pancreas transplant recipients ANZIPTR Report 2018: Australian and New Zealand Islet and Pancreas Transplant Registry, 2018; 14-16
6. OTA, Progressing Australian organ and tissue donation and transplantation to 2020: The 2016–2020 Strategy: Australian Organ and Tissue Donation and Transplantation Authority, 2016
7. Kandaswamy R, Stock PG, Gustafson SK, OPTN/SRTR 2018 annual data report: Pancreas: Am J Transplant, 2020; 20(s1); 131-92
8. Burnett JCC, Annual report on pancreas and islet transplantation: NHS Blood and Transplant, 2018
9. Bunnett JCC, Annual report on pancreas and islet transplantation. Report for 2018/2019: NHS Blood and Transplant, 2019
10. DBJ , ET Pancreas allocation system (EPAS) 5.4: Eurotransplant, 2019
11. NHSBT, POL199/11 Pancreas transplantation: Organ allocation: NHS Blood and Transplant, 2020
12. Policy OPTN, 11: Allocation of pancreas, kidney-pancreas, and islets: Organ Procurement and Transplantation Network, 2020; 253-261
13. , C R. POLICY POL185/6 Pancreas Transplantation: Patient Selection: NHS Blood and Transplant, 2019
14. TSANZ, Clinical guidelines for organ transplantation from deceased donors V1.4: Transplantation Society of Australia and New Zealand, 2020
15. Axelrod DA, Sung RS, Meyer KH, Systematic evaluation of pancreas allograft quality, outcomes and geographic variation in utilization: Am J Transplant, 2010; 10(4); 837-45
16. Mandal AK, Drew N, Lapidus JA, The effect of center volume on pancreas transplant outcomes: Surgery, 2004; 136(2); 225-31
17. Ponto J, Understanding and evaluating survey research: J Adv Pract Oncol, 2015; 6(2); 168-71
18. Kelley K, Clark B, Brown V, Sitzia J, Good practice in the conduct and reporting of survey research: Int J Qual Health Care, 2003; 15(3); 261-66
19. Burns KEA, Kho ME, How to assess a survey report: A guide for readers and peer reviewers: CMAJ, 2015; 187(6); E198-205
20. Bennett C, Khangura S, Brehaut JC, Reporting guidelines for survey research: An analysis of published guidance and reporting practices: PLoS Med, 2010; 8(8); e1001069
21. Palinkas LA, Horwitz SM, Green CA, Purposeful sampling for qualitative data collection and analysis in mixed method implementation research: Adm Policy Ment Health, 2015; 42(5); 533-44
22. Biernacki P, Waldorf D, Snowball sampling: Problems and techniques of chain referral sampling: Sociological Methods & Research, 1981; 10(2); 141-63
23. Vinkers MT, Rahmel AO, Slot MC, How to recognize a suitable pancreas donor: A Eurotransplant study of preprocurement factors: Transplant Proc, 2008; 40(5); 1275-78
24. Fridell JA, Rogers J, Stratta RJ, The pancreas allograft donor: Current status, controversies, and challenges for the future: Clin Transplant, 2010; 24(4); 433-49
25. Leighton E, Sainsbury CA, Jones GC, A practical review of C-peptide testing in diabetes: Diabetes Ther, 2017; 8(3); 475-87
26. Singh RP, Rogers J, Farney AC, Do pretransplant C-peptide levels influence outcomes in simultaneous kidney-pancreas transplantation?: Transplant Proc, 2008; 40(2); 510-512
27. Light JA, Sasaki TM, Currier CB, Barhyte DY, Successful long-term kidney-pancreas transplants regardless of C-peptide status or race: Transplantation, 2001; 71(1); 152-54
28. Light J, Tucker M, Simultaneous pancreas kidney transplants in diabetic patients with end-stage renal disease: the 20-yr experience: Clin Transplant, 2013; 27(3); E256-63
29. Chakkera HA, Bodner JK, Heilman RL, Outcomes after simultaneous pancreas and kidney transplantation and the discriminative ability of the C-peptide measurement pretransplant among type 1 and type 2 diabetes mellitus: Transplant Proc, 2010; 42(7); 2650-52
30. Stratta RJ, Farney AC, Orlando G, Rogers J, Pancreas transplantation for type 2 diabetes mellitus: Who and why?: Current Transplantation Reports, 2015; 2(2); 149-58
31. Fellmer PT, Pascher A, Kahl A, Influence of donor- and recipient-specific factors on the postoperative course after combined pancreas-kidney transplantation: Langenbecks Arch Surg, 2010; 395(1); 19-25
32. Scalea JR, Redfield RR, Arpali E, Pancreas transplantation in older patients is safe, but patient selection is paramount: Transpl Int, 2016; 29(7); 810-18
33. Dayoub JC, Cortese F, Anžič A, The effects of donor age on organ transplants: A review and implications for aging research: Exp Gerontol, 2018; 110; 230-40
34. Shah AP, Mangus RS, Powelson JA, Impact of recipient age on whole organ pancreas transplantation: Clin Transplant, 2013; 27(1); E49-55
35. Laurence JM, Marquez MA, Seal JB, The effect of recipient age on outcome after pancreas transplantation: Transplantation, 2015; 99(2); e13-14
36. Proneth A, Schnitzbauer AA, Schenker P, Extended PANCREAS DONOR PROGRAM – The EXPAND study: A prospective multicenter trial testing the use of pancreas donors older than 50 years: Transplantation, 2018; 102(8); 1330-37
37. Shahrestani S, Webster AC, Lam VW, Outcomes from pancreatic transplantation in donation after cardiac death: A systematic review and meta-analysis: Transplantation, 2017; 101(1); 122-30
38. Godown J, Kirk R, Joong A, Variability in donor selection among pediatric heart transplant providers: Results from an international survey: Pediatr Transplant, 2019; 23(5); e13417
39. Kroeker KI, Bain VG, Shaw-Stiffel T, Adult liver transplant survey: Policies towards eligibility criteria in Canada and the United States 2007: Liver Int, 2008; 28(9); 1250-55
40. Webster ARP, Mark T, Hedley J, Kelly P, Chapter 2: Pancreas transplant recipients, ANZIPTR report 2019: Australia and New zealand islet and pancreas transplant registry, 2019; 15-18
Tables
 Table 1. Responding units by region with number of SOPTx by transplant type performed within the 2017 calendar year.
Table 1. Responding units by region with number of SOPTx by transplant type performed within the 2017 calendar year. Table 2. Donor and recipient characteristics between all regions.
Table 2. Donor and recipient characteristics between all regions. Table 3. Donor and recipient characteristics by transplant volume via linear regression analysis.
Table 3. Donor and recipient characteristics by transplant volume via linear regression analysis. Table 4. Comparison of donor and recipient characteristics between ANZ and all other responding units.
Table 4. Comparison of donor and recipient characteristics between ANZ and all other responding units. Supplementary Table 1. Survey questions.
Supplementary Table 1. Survey questions. Supplementary Table 2. Donor criteria for pancreas acceptance for solid pancreas transplantation according to current national pancreas protocols.
Supplementary Table 2. Donor criteria for pancreas acceptance for solid pancreas transplantation according to current national pancreas protocols. Supplementary Table 3. Recipient eligibility criteria for solid pancreas transplantation by current national pancreas protocols.
Supplementary Table 3. Recipient eligibility criteria for solid pancreas transplantation by current national pancreas protocols. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








