22 November 2022: Original Paper
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipients with Hepatocellular Carcinoma
Gonzalo SapisochinDOI: 10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
Abstract
BACKGROUND: The study objective was to evaluate the effect of everolimus (EVR) in combination with reduced tacrolimus (rTAC) compared with a standard TAC (sTAC) regimen on hepatocellular carcinoma (HCC) recurrence in de novo living-donor liver transplantation recipients (LDLTRs) with primary HCC at liver transplantation through 5 years after transplantation.
MATERIAL AND METHODS: In this multicenter, non-interventional study, LDLTRs with primary HCC, who were previously randomized to either everolimus plus reduced tacrolimus (EVR+rTAC) or standard tacrolimus (sTAC), and who completed the 2-year core H2307 study, were followed up. Data were collected retrospectively (end of core to the start of follow-up study), and prospectively (during the 3-year follow-up study).
RESULTS: Of 117 LDLTRs with HCC at LT in the core H2307 study (EVR+rTAC, N=56; sTAC, N=61), 86 patients (EVR+rTAC, N=41; sTAC, N=45) entered the follow-up study. Overall HCC recurrence was lower but statistically non-significant in the EVR+rTAC group (3.6% vs 11.5% in sTAC; P=0.136) at 5 years after LT. There was no graft loss or chronic rejection. Acute rejection and death were comparable between treatment groups. Higher mean estimated glomerular filtration rate in the EVR+rTAC group (76.8 vs 65.8 mL/min/1.73 m² in sTAC) was maintained up to 5 years. Reported adverse events were numerically lower in the EVR+rTAC group (41.0% vs 53.5% sTAC) but not statistically significant.
CONCLUSIONS: Although statistically not significant, early EVR initiation reduced HCC recurrence, with comparable efficacy and safety, and better long-term renal function, than that of sTAC treatment.
Keywords: Carcinoma, Hepatocellular, Everolimus, Immunosuppressive Agents, Liver Transplantation, Humans, Living Donors, Tacrolimus, Follow-Up Studies, Liver Neoplasms
Background
Liver transplantation (LT) in patients with hepatocellular carcinoma (HCC) is the optimal treatment resulting in cancer resection and total replacement of potentially carcinogenic hepatocytes. However, post-transplant HCC recurrence remains an unsolved critical issue. Reduced host immunity against micro-metastases or
The pivotal H2304 study (NCT00622869) showed comparable efficacy and superior renal function in deceased donor LT (DDLT) recipients, who received EVR with early tacrolimus (TAC) reduction (starting Month 1) compared to standard TAC (sTAC), at Month 12 [5]. In addition, improved renal function over sTAC treatment persisted with EVR treatment until Month 36 [6,7]. A similarly designed randomized controlled study (RCT) (H2307 study, NCT01888432) in the living-donor liver transplantation (LDLT) setting reported comparable efficacy and renal function with no HCC recurrence in the EVR group at Month 12 [8]. Analysis of data pooled from 2 RCTs (NCT00622869 and NCT01888432) further supported the benefit of early EVR-facilitated CNI reduction, resulting in comparable efficacy across 2 treatment regimens at Month 24 [9]. Moreover, the findings of a meta-analysis, including 3666 LT recipients from 42 RCTs with a median follow-up of 36 months, supported the effectiveness of mTOR inhibitors (EVR and SRL) in significantly lowering HCC recurrence rates compared to standard CNI treatment [10]. Other studies, including the Preservation of Renal Function in Liver Transplant Recipients with Certican Therapy (PROTECT) study [11] and the observational Everolimus Liver registry (EVEROLIVER) in France [12], showed superior renal function with EVR, which was maintained long-term in DDLT recipients. However, long-term data are limited in the LDLT setting.
Here, we report the results of a non-interventional, long-term follow-up study (CRAD001H2406) designed to evaluate HCC recurrence, efficacy, renal function, and safety outcomes at 5 years after LT in HCC patients who underwent LDLT in the core study (NCT01888432).
Material and Methods
STUDY DESIGN AND CONDUCT:
The follow-up study was a long-term, multicenter, non-interventional, observational study in LDLT recipients (LDLTRs) with primary HCC at LT who completed the 2-year multicenter, open-label, randomized, controlled core H2307 study and were followed for another 3 years. In the core H2307 study, LDLTRs receiving TAC-based immunosuppression were randomized (1: 1) at 30±5 days after transplantation to EVR plus reduced TAC (EVR+rTAC) or sTAC [8]. The study protocol and amendments were reviewed by the Independent Ethics Committee or the Institutional Review Board for each center. The study was conducted according to International Council for Harmonization (ICH) E6 guideline for Good Clinical Practice (GCP) that has its origin in the Declaration of Helsinki, and the Strengthening of the Reporting of Observational Studies in Epidemiology guidelines. Written informed consent was obtained from each patient as per local regulatory guidance.
Available data at the clinical practice were collected using a flexible visit schedule. Primary data of the eligible patients, who provided consent, from the core H2307 study including CT scan (or other imaging data) for HCC recurrence were collected prospectively at a routine outpatient clinic visit until the end of the follow-up study or withdrawal of consent. Secondary patient data were collected retrospectively between the completion/end of study visit in the core study and the start of the observational follow-up study, including information on patients that died after the end of the core study. A schematic diagram of the study design is presented in Figure 1.
STUDY POPULATION:
The study population was composed of patients who completed the 2-year core study, signed the informed consent, and had primary HCC at the time of LT. Patients who experienced graft loss either during the 2-year core study, or in the 9 months before the start of follow-up study, were excluded.
STUDY MEDICATION:
In the follow-up study, patients continued their randomized treatment of either EVR+rTAC (target trough concentration 3–8 ng/mL for EVR and 3–5 ng/mL for TAC) or sTAC (target trough concentration, 6–10 ng/mL), as assigned in the core study [8]. Any treatment changes during the follow-up period were captured and evaluated.
STUDY ENDPOINTS:
The primary endpoint was HCC recurrence from randomization in core study until Month 60 after transplantation in patients with HCC at the time of LT. Secondary endpoints, including efficacy assessments (incidences of acute and chronic rejections [AR/CR], graft loss [GL], death [D], ‘composite of drop-out’ [death, withdrawal of consent and lost to follow-up], malignancies other than HCC), changes in immunosuppressive regimen, and change in renal function as measured by the estimated glomerular filtration rate [modification of diet in renal diseases-4] (eGFR [MDRD-4] mL/min/1.73 m2) and other formulae (MDRD-6, chronic kidney disease classification-epidemiology collaboration [CKD-EPI], Cockcroft-Gault), were analyzed from randomization in the core study until Months 36, 48, and 60 after transplantation. The incidence of HCC recurrence was analyzed for Months 36 and 48 for secondary endpoints, as well as at Month 60 for the primary endpoint. Safety endpoints were incidences of adverse events (AEs), including infections and renal replacement therapy, and severe AEs (SAEs) and were analyzed from the start of the follow-up study until Month 60 after transplantation.
STATISTICAL ANALYSIS:
The 5-year post-transplant data analysis was based on combined datasets of patients with HCC from the start of the core study and 3 years of retrospective and prospective data from the follow-up study, except for medical history, AEs (other than HCC recurrence and malignancies), and concomitant medications, which were based on follow-up study data only.
The intent-to-treat (ITT) population consisted of all patients who had HCC at the time of transplantation and initiated treatment in the core study. It was used for primary analysis and secondary analyses for rejections/graft loss/death and renal parameters. The ITT population also included data retrospectively collected from patients who died after the end of the core study and before the start of the follow-up study. Informed consent was obtained from next of kin in case of such deceased patients. The as-treated (AT) population consisted of patient groups according to the presence or absence of EVR in the treatment regimen at any time during the prospective period of the follow-up study. The AT population excluded patients who died after the end of the core study and before the start of the follow-up study. The AT population was utilized for analyses of safety data (AEs and concomitant medications) during the follow-up study.
Demographics, patient baseline characteristics, recipient LT information, and HCC history were summarized using frequency distributions in case of categorical variables and descriptive statistics in case of continuous variables. Efficacy parameters, including HCC recurrence, as well as the cumulative rate of HCC or death at Months 24, 36, 48, and 60, were estimated based on the Kaplan-Meier (KM) estimator in the ITT population. Greenwood’s formula was used to derive the variance and the two-sided 95% Z-test based confidence interval (CI). For analyses on incidence of rejection, graft loss, death, and HCC recurrence, patients who discontinued this study without any event were censored at the latest known day to be free of the event. The HCC recurrence rate until Month 60 was also analyzed in subgroups within and beyond Milan criteria at transplantation.
Summary statistics for eGFR (MDRD-4, MDRD-6, CKD-EPI, Cockcroft-Gault), as well as serum creatinine, were captured by visit and analyzed within the ITT population, as well as for the on-treatment data defined as those measured up to the last dose of randomized medication + 2 days. No imputation was performed for eGFR.
Results
PATIENT DISPOSITION:
In the core H2307 study, 117 patients who had HCC were randomized after LT (EVR+rTAC, 56 patients; sTAC, 61 patients), out of which 86 patients (EVR+rTAC, 41; sTAC, 45) entered the follow-up study. Four patients who died after completion of the core study but before start of the follow-up study (1 patient in EVR+rTAC and 3 patients in sTAC) were also included in the 86 patients (Figure 2).
DEMOGRAPHICS AND BASELINE CHARACTERISTICS:
Patient demographics and background characteristics for patients on or before the first dose of study treatment in the core study and who entered the follow-up study were comparable between 2 treatment groups (Table 1). The majority of patients were Asians (91.9%), and 31.4% of patients had type II diabetes at baseline. Most patients had high (≥60) eGFR (MDRD 4) [mL/min/1.73 m2] at baseline (90.7%) as well as at randomization (74.4%) in the core study. More than 75% of patients had MELD score ≤14, and 73.3% of the patients with HCC from the core study who entered the follow-up study were within Milan criteria at the time of transplantation.
IMMUNOSUPPRESSION/PATIENT EXPOSURE:
In the EVR+rTAC group, 39 patients received EVR+rTAC treatment, while only 41 of 43 in the sTAC group received the standard dose of TAC. At Month 60 in the follow-up study, 32 out of 39 patients in the EVR+rTAC treatment arm were still receiving EVR (6 patients switched to sTAC), while 34 of 41 patients in the sTAC arm continued to receive TAC (5 patients switched to the EVR treatment). At Month 60, 82% to 83% of the patients who received either EVR+rTAC or sTAC in core study continued to receive their respective treatment regimen (Table 2).
EFFICACY:
The summary of efficacy outcomes is presented in Table 3. HCC recurrence was observed at a lower numerical rate in the EVR+rTAC group as compared to the sTAC group (1 vs 3; observed difference of −4.2% in KM rate [95% CI: −12.9%, 4.5%]) at Month 60 after LT, in the follow-up study; however, the difference was not statistically significant. HCC recurrence occurred in 1 patient in each treatment group during the follow-up study period. In the EVR+rTAC group, HCC recurrence occurred a few weeks after completion of the core Month 24 visit (ie, during the retrospective period), while in the sTAC group, the recurrence occurred at Month 30 after LT. The lower rate of HCC recurrence for the EVR+rTAC group was consistent over the 60-month period in the ITT population across the core and follow-up studies (Figure 3A, Table 4).
The KM incidence of composite endpoint of AR/CR/GL/death was lower in the EVR+rTAC group versus sTAC group, showing the largest difference of −8.4% (95% CI: −21.8, 4.9%) among all the efficacy endpoints for the ITT population who entered follow-up study. GL or CR was not observed during the follow-up study for either treatment, and the KM incidences of death (EVR+rTAC, 2; sTAC, 4) were comparable between the 2 treatment groups. The KM incidence of HCC recurrence remained the same from Month 24 to Month 60 in the EVR+rTAC group, while the KM incidence of the composite endpoint of AR/CR/GL/death increased in both the treatment groups from Month 24 to Month 60 (EVR+rTAC vs sTAC arm: 2 vs 3 at Month 24; 3 vs 5 Month 48; 3 vs 7 at Month 60). The KM incidence of HCC recurrence was 1 and 2 in the EVR+rTAC group and sTAC group, respectively, at Month 24 (Table 3).
The cumulative rate of either no HCC recurrence or death from any cause (whichever occurred earlier) in the ITT population had a higher KM estimate at Month 60 in the EVR+rTAC group (86.2%) than in the sTAC group (82.5%) (Figure 3B, Table 5). A smaller reduction in the cumulative rate of HCC or death was also observed for the ITT patients (difference=−4.0% with 95% CI: −14.6%, 6.6%). A subgroup analysis for HCC recurrence in ITT population at Month 60 found that 2 out of 42 patients (KM rate%=5.8) in the EVR+rTAC group and 1 out of 40 patients (KM rate%=2.7) in the sTAC group were within Milan criteria, while 6 out of 19 patients (KM rate%=34.3) in the sTAC group were beyond Milan criteria. No HCC recurrence was found in the 14 patients in the EVR+rTAC group that were beyond Milan criteria. The KM incidence of malignancies other than HCC recurrence was also numerically lower in the EVR+rTAC group (12.6%) than in the sTAC group (17.5%), with a difference of −4.8% (−18.5, 8.9) at Month 60; however, the difference was not statistically significant.
Multivariate (MV) Cox regression analysis for overall survival (OS) showed a significant difference for recipient age and Milan criteria, as shown in Table 6; however, there is no significant difference across treatments. OS is defined here as the length of time when recipients survived from death by any cause or HCC recurrence, whichever occurred earlier.
RENAL FUNCTION:
Mean eGFR [MDRD4] remained numerically higher in the EVR+rTAC group as compared to the sTAC group throughout the core study period (Month 24: EVR+rTAC: 84.28 mL/min/1.73 m2, sTAC: 70.18 mL/min/1.73 m2), as well as through the course of H2406 up to Month 60 (EVR+rTAC: 76.83 mL/min/1.73 m2, sTAC: 65.84 mL/min/1.73 m2) after transplantation in the ITT population (Figure 4).
Mean eGFR [MDRD-4] by treatment and time point was also higher in the EVR+rTAC group for most of the eGFR formulae used (MDRD-6, CKD-EPI), except for mean values using Cockcroft-Gault method from Month 42 onward.
SAFETY:
The proportion of patients with AEs was numerically lower in patients receiving EVR+rTAC (16 [41.0%]) compared to those receiving sTAC (23 [53.5%]) at Month 60. Most AEs were mild in nature, with 3 patients (7.7%) in the EVR+rTAC group and 6 patients (14.0%) in the sTAC group that presented with severe AEs. The incidence of infections and infestations was lower in the patients receiving EVR+rTAC (15.4%) as compared to those receiving sTAC (23.3%). Safety events are presented as a MedDRA preferred term (PT) in Table 7. Two deaths occurred in the prospective period of H2406 in the AT population, out of which 1 death in the EVR+rTAC group was due to HCC recurrence (diagnosed at Day 1318 and died on Day 1350), while that in the sTAC group was due to respiratory failure. The investigator did not suspect a relationship of death with treatment in either of the cases. AEs leading to study drug discontinuation were experienced by 1 patient in each treatment group.
Discussion
We present the results of a 5-year follow-up study examining the long-term efficacy and safety of early EVR initiation in patients who underwent LDLT for HCC and completed the 2-year core RCT study. A numerically lower rate of HCC recurrence in EVR+rTAC was observed as compared to those in sTAC at 5 years after LT. Moreover, better renal function was maintained in the EVR+rTAC group than that in the sTAC group throughout 5 years after LDLT. Overall, a balanced safety profile was observed for the EVR regimen, with lower incidence of infections versus sTAC regimen. Although statistically not significant, the 5-year OS (ie, death by any cause or HCC recurrence, whichever occurred earlier) was 86.2% and 82.5% in the EVR+rTAC group and sTAC group, respectively. The recurrence-free survival (RFS) was also numerically higher with EVR+rTAC than with the sTAC regimen (95.6% vs 87.7%) in the whole cohort of the core study. The trend was maintained throughout the 5-year study period, with no HCC recurrence beyond 24 months after LT EVR+rTAC, while 1 HCC recurrence was observed in sTAC at Month 30. MV Cox regression analysis identified exceeding Milan criteria (
HCC is most prevalent in Asia, accounting for >60% of HCC cases reported globally, and is the most common indication of LT, secondary to hepatitis B [15–17]. Therefore, the efficacy of early EVR initiation in lowering HCC recurrence is certainly encouraging for this population. The results of this observational study are also in line with the findings of meta-analyses that showed mTOR inhibitors were better at lowering the HCC recurrence rates after LT compared to that of CNIs [10,18,19]. Also, a recent meta-analysis showed that 5-year OS and 3-year RFS improved with mTOR inhibitor-based compared with mTOR inhibitor-free immunosuppression [20].
The KM incidence of malignancies other than HCC recurrence was also numerically lower in the patients (in ITT) receiving EVR+rTAC (12.6% vs 17.4% in sTAC group; 90% CI: −18.5%, 8.9%) in the core study at 5 years. In ITT patients who entered the follow-up study, no incidence of CR or GL up to 60 months after LT was observed, and the KM incidences of AR and death were comparable (AR: difference=−2.2%, 95% CI: −12.4, 8.0%; death: difference=−4.0%, 95% CI: −14.6, 6.6%) between the treatment arms. In the core study, the non-inferiority of the primary composite efficacy failure endpoint (treated BPAR, GL, or death) was maintained until Month 24, and a lower incidence of tBPAR in patients receiving EVR was also observed [8,21]. The analysis of patient data pooled from 2 RCTs (pivotal H2304 in DDLT setting and 2-year core study in LDLT setting) also found comparable efficacy between the 2 treatment groups [9]. Although tBPAR was not evaluated in the follow-up period, the reduction was highest for the composite endpoint of AR/CR/GL/death in the EVR group versus the sTAC group (difference=−8.4%, 95% CI: −21.8, 4.9%) at 5-year post-LT for ITT patients who entered in the follow-up study. Therefore, early initiation of EVR is effective in both DDLT [6,7] and LDLT [8,21] settings, even in the long term.
Chronic renal failure is another concern associated with prolonged use of CNI in solid-organ recipients, and mTOR inhibitor-facilitated CNI reduction has been effective in preserving renal function. In DDLT settings, several clinical trials, including H2304 (3 years) [7], SIMCER (5 years) [22, 23] and PROTECT (5 years) [11], demonstrated better renal function with EVR-based CNI reduction or CNI-free EVR regimens for up to 5 years of follow-up. The 2-year core study showed significantly higher renal function at 2 years after LT in patients (with HCC at LT) receiving EVR+rTAC versus sTAC, which was maintained up to 5-year after LT [21]. Furthermore, the pooled analysis of the H2304 and H2307 trials demonstrated significantly better renal function in the EVR+rTAC group versus the sTAC group at 24 months of follow-up, particularly in patients with normal/mildly decreased renal function (chronic kidney disease stage 1 or 2) at randomization [9]. The EVEROLIVER registry data also reflected the renal benefit of the EVR-facilitated CNI reduction regimen in improving mean eGFR in patients at up to 5-year follow-up [12]. The findings from this follow-up study in the LDLT setting further demonstrated that early conversion to an EVR-based regimen improves/preserves renal function, both immediately and long-term. A recent retrospective real-world study showed that patients with renal impairment experienced significant improvement in renal function after conversion to mTOR inhibitor [24].
Compared to a very high proportion of patients with AEs in both treatment groups (98.6% in EVR+rTAC vs 96.5% in sTAC) during the core study, a larger reduction in the proportion of patients with AEs in both treatment groups (41.0% vs 53.5%) with greater reduction in the EVR group was reported in the follow-up period [8]. A lower incidence of infections and infestations was also observed in the patients receiving EVR during the follow-up period. Overall, the safety data obtained during the follow-up study period suggest that reducing CNI exposure with early EVR initiation would provide LT recipients long-term safety against exposure to immunosuppressive treatment involving the standard CNI regimen.
As this was a follow-up study of the preceding randomized controlled study (H2307 study, NCT01888432), the statistical power was not sufficient due to the limited number of cases that could be analyzed.
Conclusions
This follow-up study demonstrated the efficacy of early initiation of EVR with reduced TAC in numerically lowering the rate HCC recurrence and graft rejection. Statistically, the efficacy of EVR regimen was comparable with standard TAC regimen. Better long-term renal function was preserved in patients receiving the EVR regimen throughout the course of the study. Importantly, a safety profile comparable to sTAC with less incidence of infections was also achieved with EVR treatment at up to 5-year follow-up in the LDLT setting.
Figures
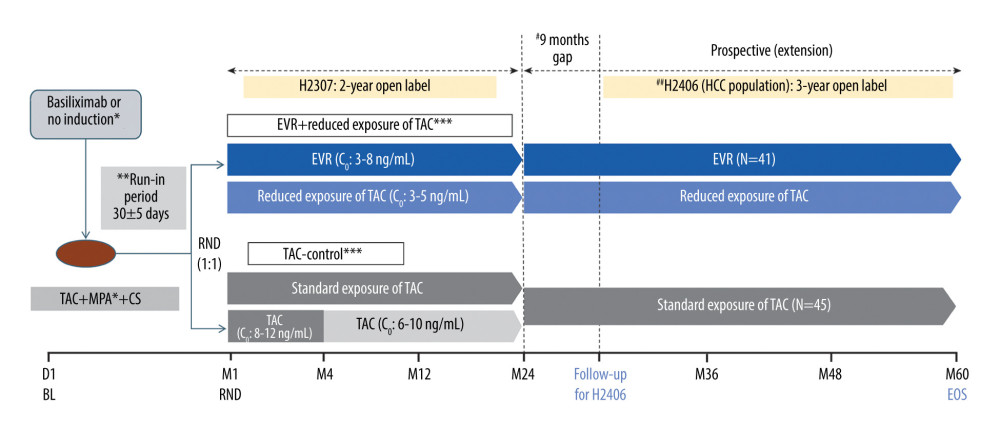 Figure 1. Design of the follow-up study. * Per the center’s choice; ** All patients received TAC (C0: 5–15 ng/mL) during the run-in phase; *** CS in both arms per the local practice; # variable period between the end of study visit in the H2307 study and the start of data collection for study H2406; ## treatment as per the local clinical practice during H2406. C0 – trough level; CS – corticosteroid; EOS – end of study; EVR – everolimus; HCC – hepatocellular carcinoma; LT – liver transplantation; M – month; MPA – mycophenolic acid; RND – randomization; TAC – tacrolimus. Created using Microsoft Office (2016, Microsoft).
Figure 1. Design of the follow-up study. * Per the center’s choice; ** All patients received TAC (C0: 5–15 ng/mL) during the run-in phase; *** CS in both arms per the local practice; # variable period between the end of study visit in the H2307 study and the start of data collection for study H2406; ## treatment as per the local clinical practice during H2406. C0 – trough level; CS – corticosteroid; EOS – end of study; EVR – everolimus; HCC – hepatocellular carcinoma; LT – liver transplantation; M – month; MPA – mycophenolic acid; RND – randomization; TAC – tacrolimus. Created using Microsoft Office (2016, Microsoft). 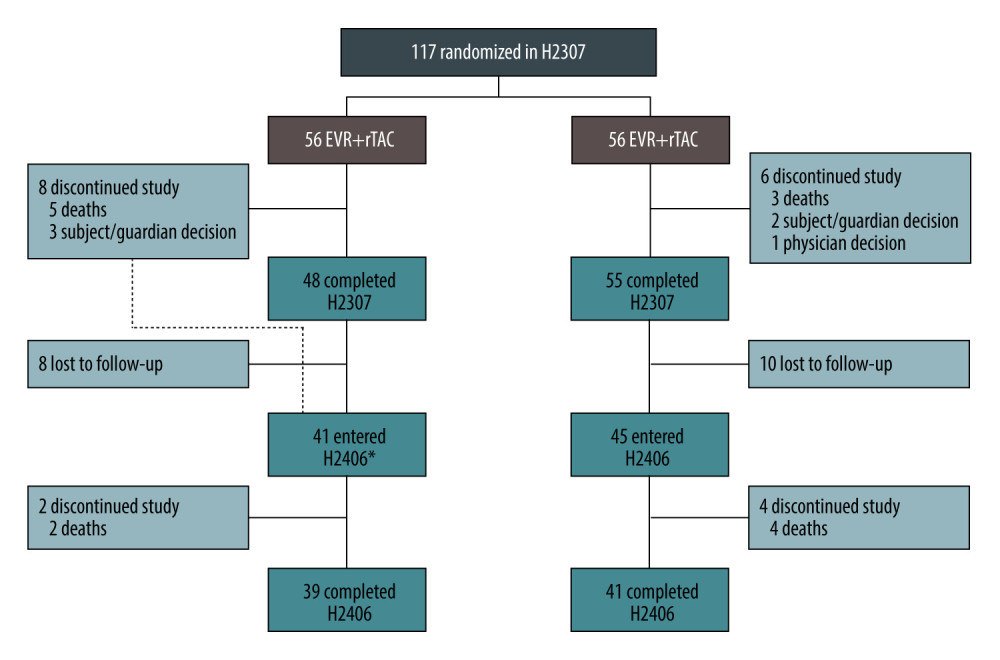 Figure 2. Patient disposition up to Month 60. * One patient discontinued the core study, but was enrolled in H2406. Created using Microsoft Office (2016, Microsoft).
Figure 2. Patient disposition up to Month 60. * One patient discontinued the core study, but was enrolled in H2406. Created using Microsoft Office (2016, Microsoft). 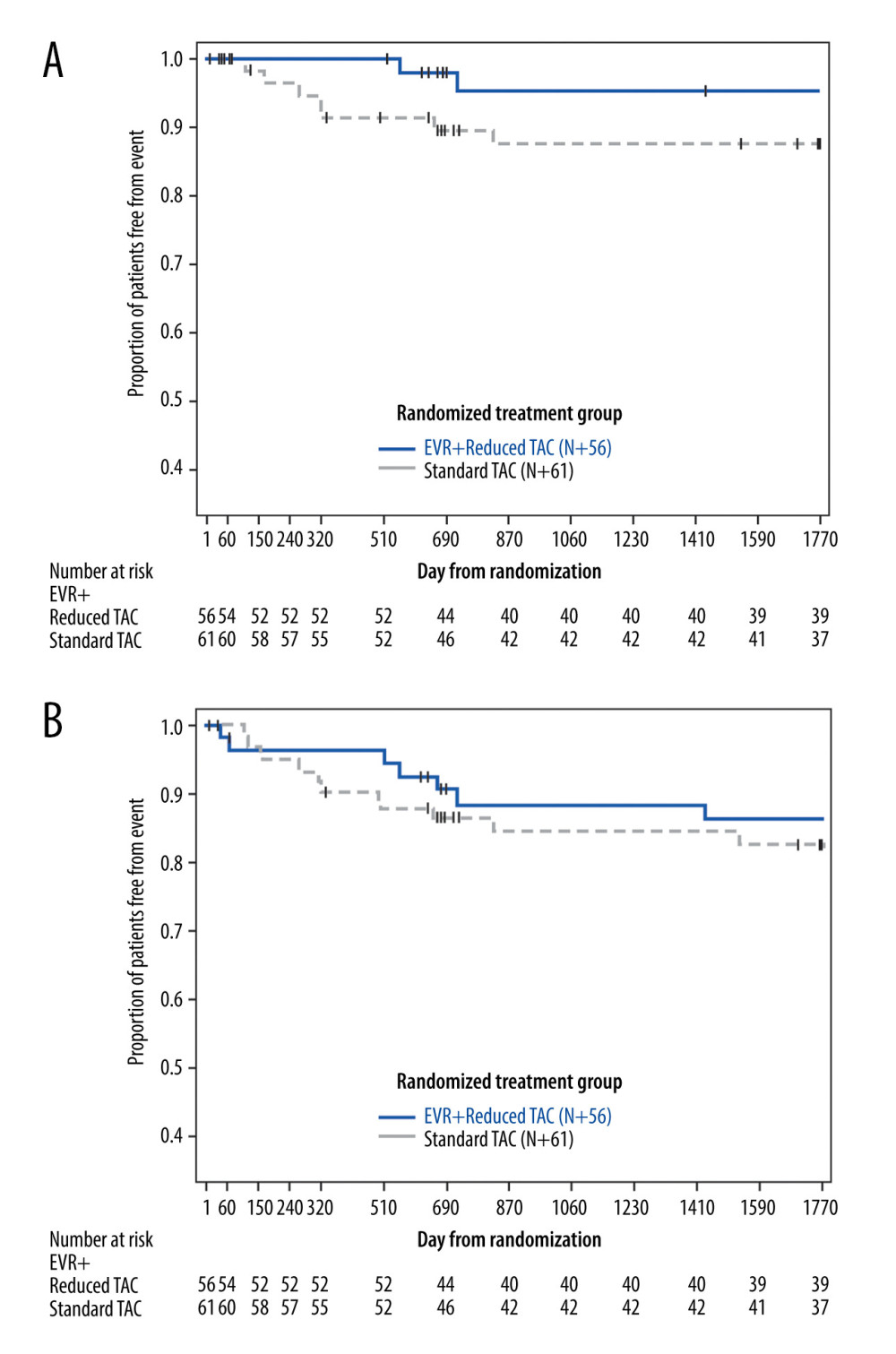 Figure 3. Kaplan-Meier plot for proportion of patients (ITT population) free from HCC recurrence (A) and free from HCC recurrence or death (B). EVR+rTAC – everolimus plus reduced tacrolimus; HCC – hepatocellular carcinoma; ITT – intent-to-treat; sTAC – standard tacrolimus. Created using SAS version 9.4.
Figure 3. Kaplan-Meier plot for proportion of patients (ITT population) free from HCC recurrence (A) and free from HCC recurrence or death (B). EVR+rTAC – everolimus plus reduced tacrolimus; HCC – hepatocellular carcinoma; ITT – intent-to-treat; sTAC – standard tacrolimus. Created using SAS version 9.4. 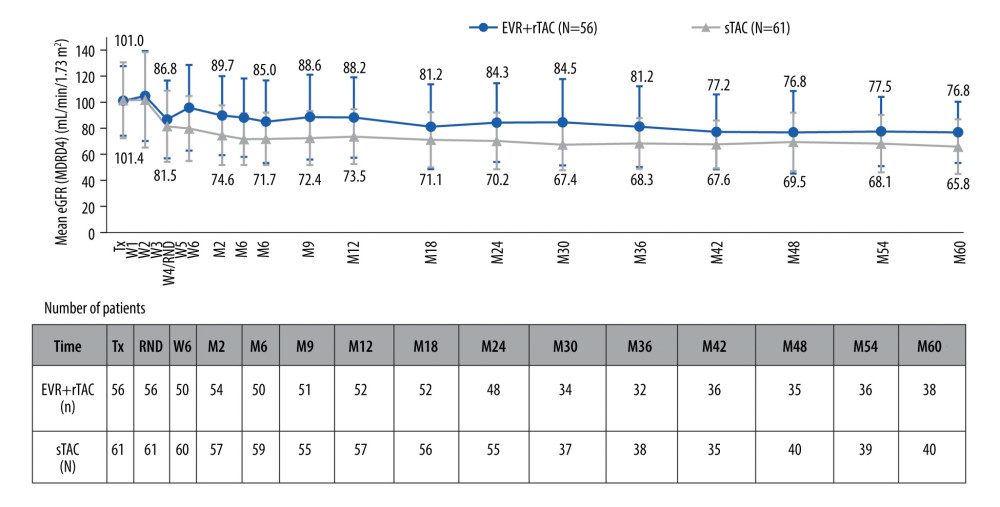 Figure 4. Evolution of renal function until Month 60 (in ITT population). eGFR – estimated glomerular filtration rate; MDRD4 – modification of diet in renal diseases-4; EVR+rTAC – everolimus plus reduced tacrolimus; M – month; RND – randomization; sTAC – standard tacrolimus; Tx – transplantation; W – week. Created using Microsoft Office (2016, Microsoft).
Figure 4. Evolution of renal function until Month 60 (in ITT population). eGFR – estimated glomerular filtration rate; MDRD4 – modification of diet in renal diseases-4; EVR+rTAC – everolimus plus reduced tacrolimus; M – month; RND – randomization; sTAC – standard tacrolimus; Tx – transplantation; W – week. Created using Microsoft Office (2016, Microsoft). Tables
Table 1. Demographics and baseline characteristics in ITT patients who followed to the H2406 study.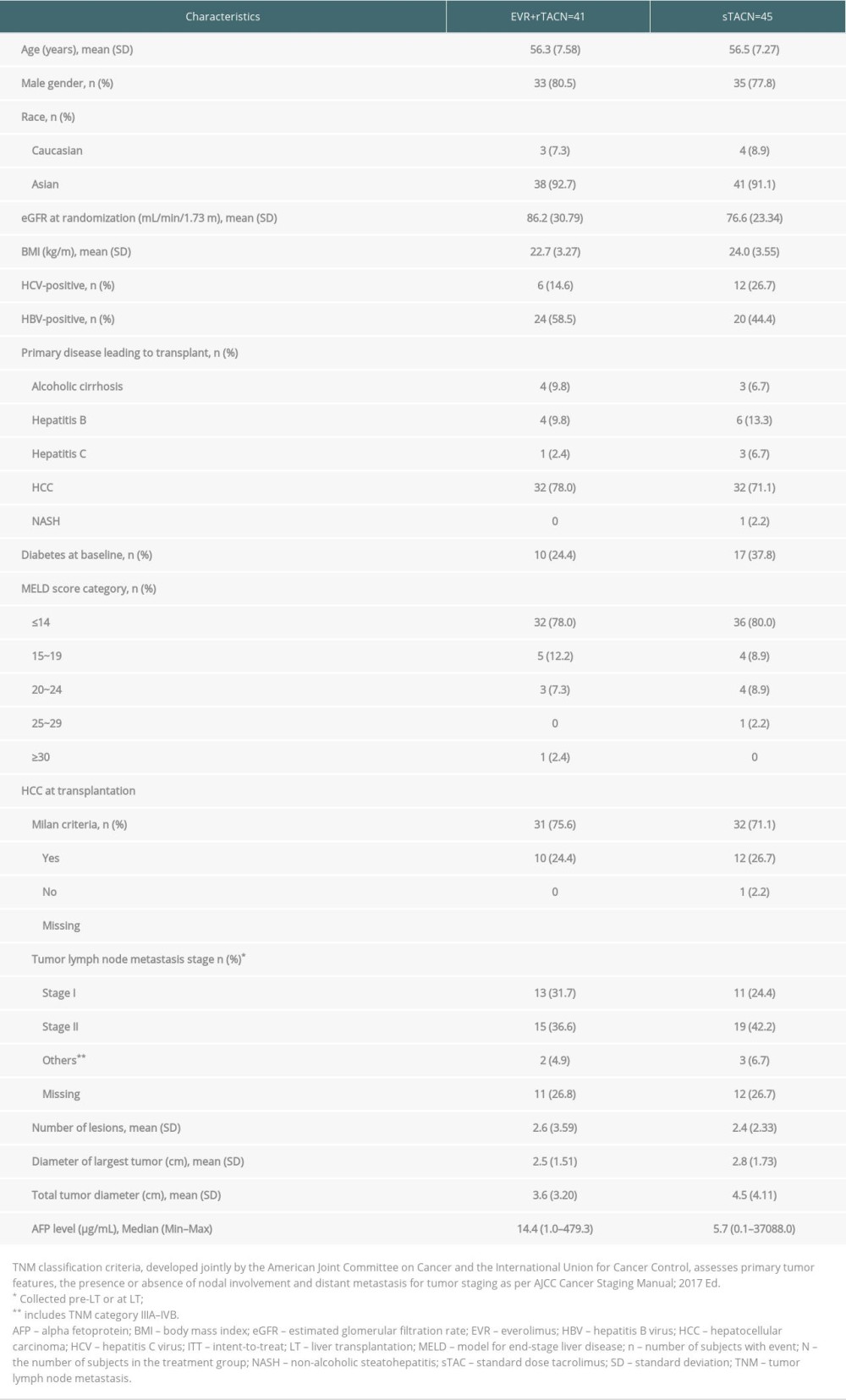 Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint.
Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint.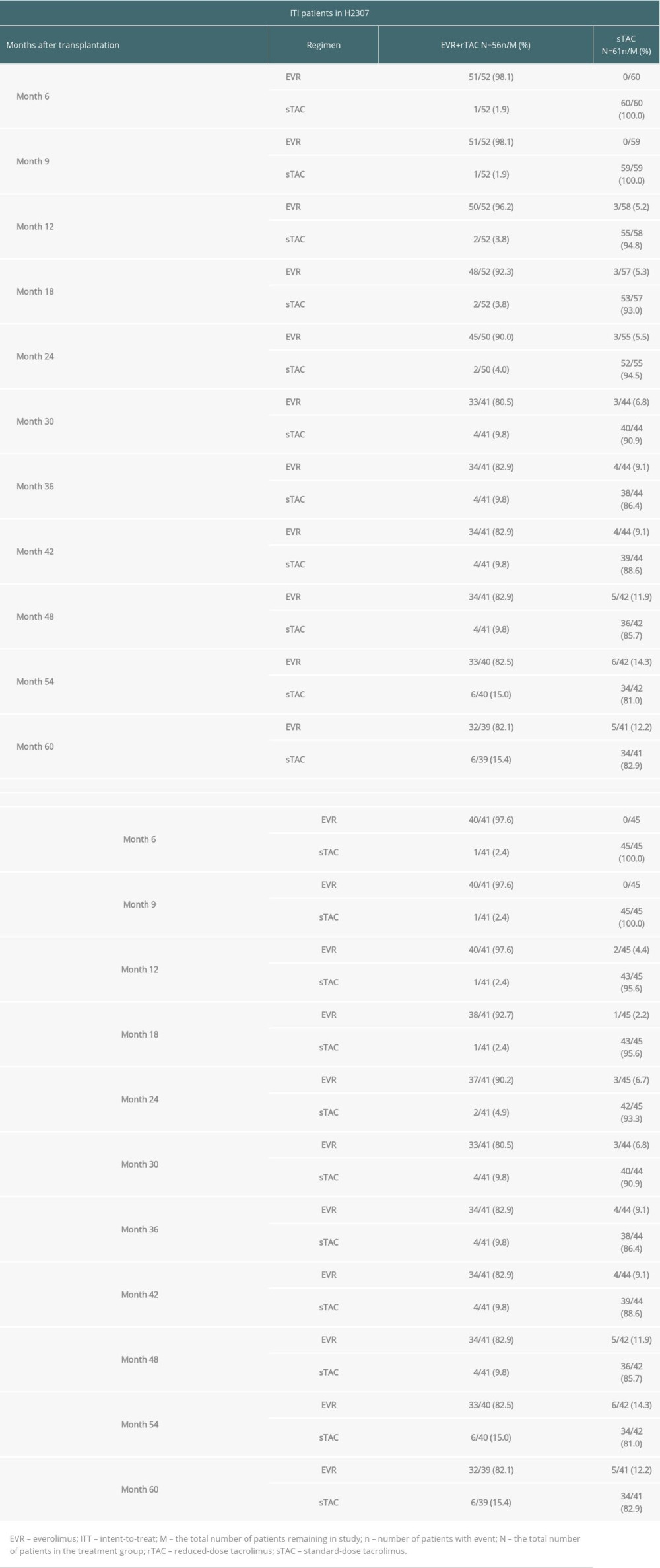 Table 3. Efficacy results for patients in ITT population.
Table 3. Efficacy results for patients in ITT population.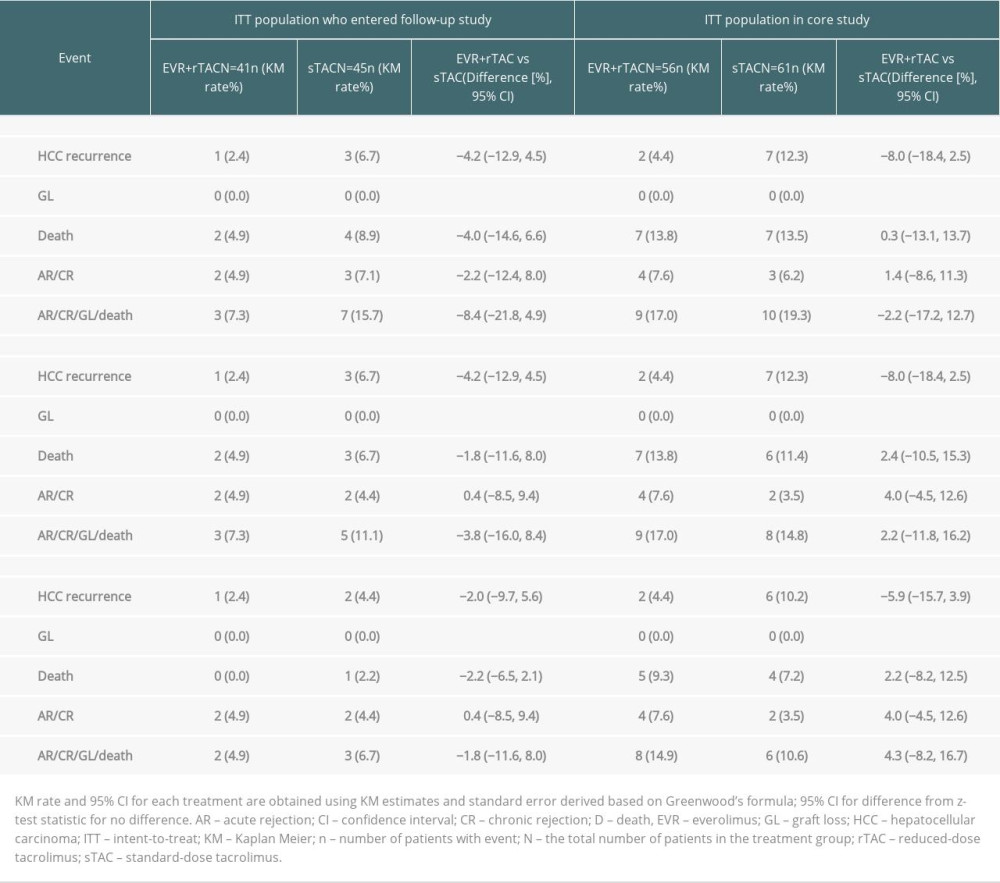 Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT).
Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT).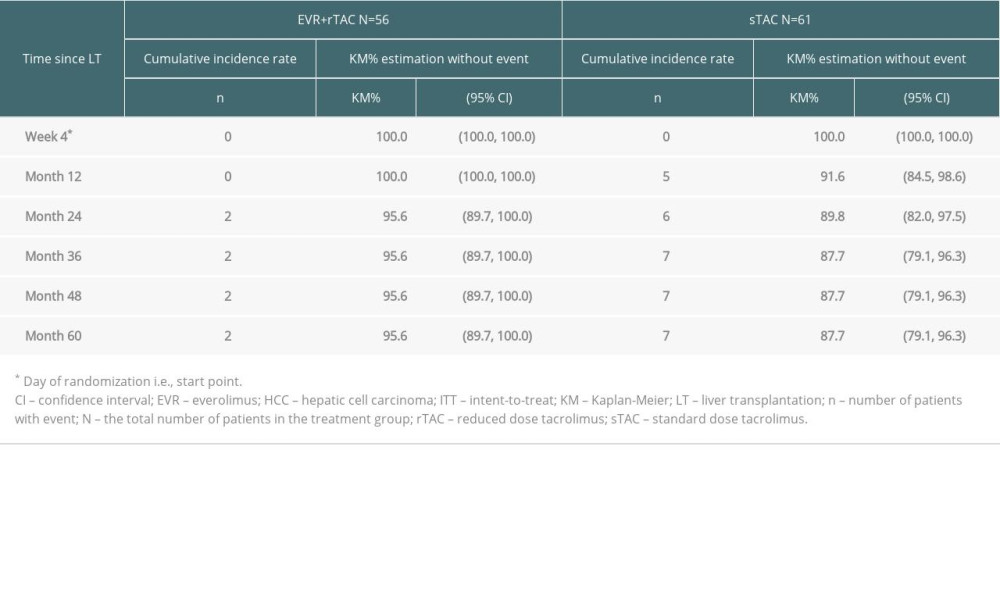 Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT).
Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT).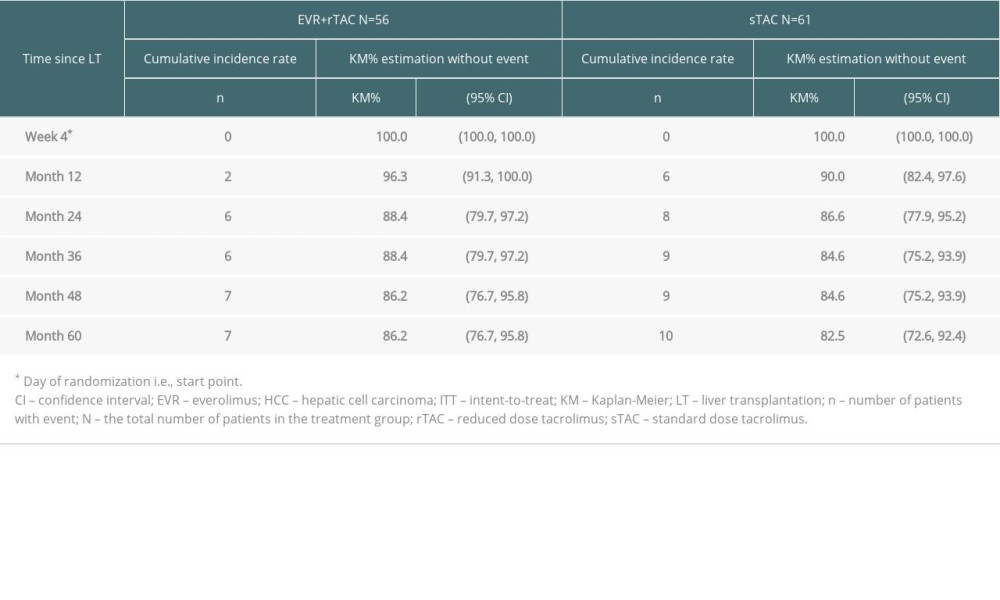 Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*).
Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*).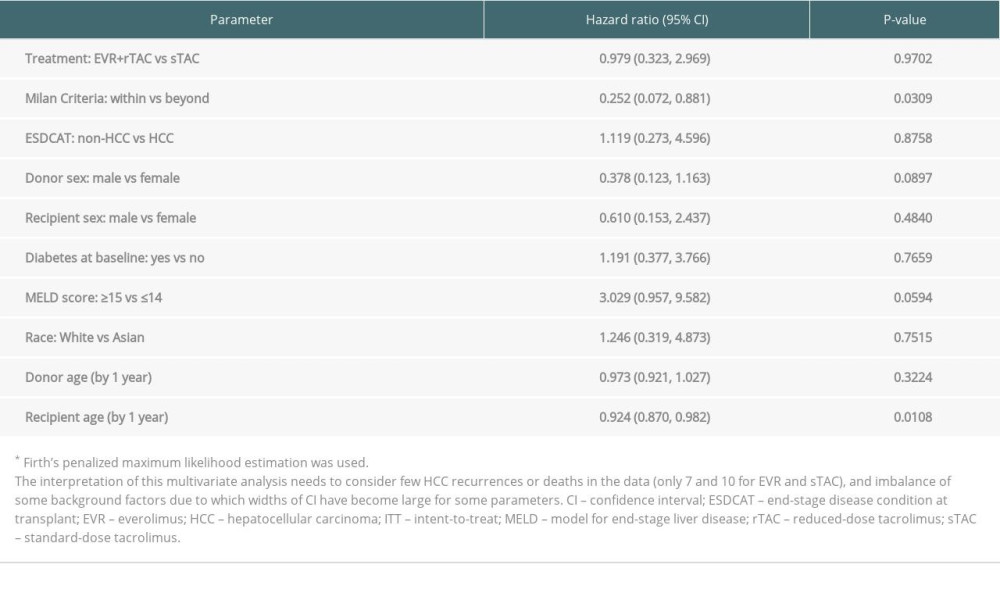 Table 7. Safety events for the patients who entered in H2406 in the AT population.
Table 7. Safety events for the patients who entered in H2406 in the AT population.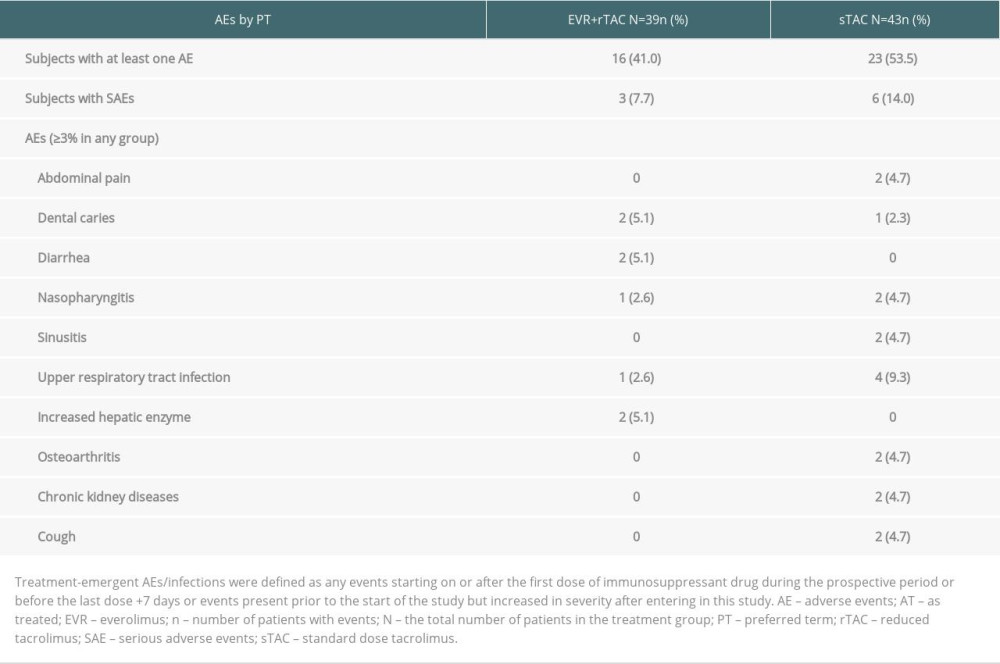
References
1. Rodríguez-Perálvarez M, De la Mata M, Burroughs AK, Liver transplantation: Immunosuppression and oncology: Curr Opin Organ Transplant, 2014; 19(3); 253-60
2. Farkas SA, Schnitzbauer AA, Kirchner G, Calcineurin inhibitor minimization protocols in liver transplantation: Transplant Int, 2009; 22; 49-60
3. Cheung A, Spotlight on impactful research: Long-term calcineurin inhibitor therapy and brain function in patients after liver transplantation: Clin Liver Dis (Hoboken), 2020; 15(4); 141-43
4. Grigg SE, Sarri GL, Gow PJ, Systematic review with meta-analysis: Sirolimus- or everolimus-based immunosuppression following liver transplantation for hepatocellular carcinoma: Aliment Pharmacol Ther, 2019; 49; 1260-73
5. De Simone P, Nevens F, De Carlis L, Everolimus with reduced tacrolimus improves renal function in de novo liver transplant recipients: A randomized controlled trial: Am J Transplant, 2012; 12(11); 3008-20
6. Saliba F, De Simone P, Nevens F, Renal function at two years in liver transplant patients receiving everolimus: Results of a randomized, multicenter study: Am J Transplant, 2013; 13(7); 1734-45
7. Fischer L, Saliba F, Kaiser GM, Three-year outcomes in de novo liver transplant patients receiving everolimus with reduced tacrolimus: Follow-up results from a randomized, multicenter study: Transplantation, 2015; 99(7); 1455-62
8. Jeng LB, Lee SG, Soin AS, Efficacy and safety of everolimus with reduced tacrolimus in living-donor liver transplant recipients: 12-month results of a randomized multicenter study: Am J Transplant, 2018; 18(6); 1435-46
9. Lee SG, Jeng LB, Saliba F, Efficacy and safety of everolimus with reduced tacrolimus in liver transplant recipients: 24-month results from the pooled analysis of two randomized controlled trials: Transplantation, 2021; 105; 1564-75
10. Cholongitas E, Mamou C, Rodríguez-Castro , Mammalian target of rapamycin inhibitors are associated with lower rates of hepatocellular carcinoma recurrence after liver transplantation: A systematic review: Transpl Int, 2014; 27; 1039-49
11. Sterneck M, Kaiser GM, Heyne N, Long-term follow-up of five yr shows superior renal function with everolimus plus early calcineurin inhibitor withdrawal in the PROTECT randomized liver transplantation study: Clin Transplant, 2016; 30(6); 741-48
12. Saliba F, Dharancy S, Salame E, Time to conversion to an everolimus-based regimen: Renal outcomes in liver transplant recipients from the EVEROLIVER registry: Liver Transpl, 2020; 26(11); 1465-76
13. Toniutto P, Fornasiere E, Fumolo E, Risk factors for hepatocellular carcinoma recurrence after liver transplantation: Hepatoma Res, 2020; 6; 50
14. Wang P, Wang C, Li H, Impact of age on the prognosis after liver transplantation for patients with hepatocellular carcinoma: A single-center experience: Onco Targets Ther, 2015; 8; 3775-81
15. Chen CL, Kabiling CS, Concejero AM, Why does living donor liver transplantation flourish in Asia?: Nat Rev Gastroenterol Hepatol, 2013; 10(12); 746-51
16. Shukla A, Vadeyar H, Rela M, Liver transplantation: East versus West: J Clin Exp Hepatol, 2013(3); 243-53
17. Chan SC, Cheung TT, Chan AC, New insights after the first 1000 liver transplantations at The University of Hong Kong: Asian J Surg, 2016; 39(4); 202-10
18. Tarantino G, Magistri P, Ballarin R, Oncological impact of M-Tor inhibitor immunosuppressive therapy after liver transplantation for hepatocellular carcinoma: Review of the literature: Front Pharmacol, 2016; 7; 387
19. Kang I, Lee JG, Choi SH, Impact of everolimus on survival after liver transplantation for hepatocellular carcinoma: Clin Mol Hepatol, 2021; 27(4); 589-602
20. Yan X, Huang S, Yang Y, Sirolimus or everolimus improves survival after liver transplantation for hepatocellular carcinoma: A systematic review and meta-analysis: Liver Transpl, 2022; 28(6); 1063-77
21. Suh KS, Jeng L-B, Soin AS, Renal function outcomes with everolimus plus reduced-exposure tacrolimus in de novo living donor liver transplantation: 24-month results from the H2307 study: Transplantation, 2018; 102; S21-S22
22. Saliba F, Duvoux C, Gugenheim J, Efficacy and safety of everolimus and mycophenolic acid with early tacrolimus withdrawal after liver transplantation: A multicenter randomized trial: Am J Transplant, 2017; 17(7); 1843-52
23. Saliba F, Duvoux C, Dharancy S, Five-year outcomes in liver transplant patients receiving everolimus with or without a calcineurin inhibitor: Results from the CERTITUDE study: Liver Int, 2022; 42(11); 2513-23
24. Sung PS, Han JW, Seo C, Real-life experience of mTOR inhibitors in liver transplant recipients in a region where living donation is predominant: Front Pharmacol, 2021; 12; 685176
Figures
 Figure 1. Design of the follow-up study. * Per the center’s choice; ** All patients received TAC (C0: 5–15 ng/mL) during the run-in phase; *** CS in both arms per the local practice; # variable period between the end of study visit in the H2307 study and the start of data collection for study H2406; ## treatment as per the local clinical practice during H2406. C0 – trough level; CS – corticosteroid; EOS – end of study; EVR – everolimus; HCC – hepatocellular carcinoma; LT – liver transplantation; M – month; MPA – mycophenolic acid; RND – randomization; TAC – tacrolimus. Created using Microsoft Office (2016, Microsoft).
Figure 1. Design of the follow-up study. * Per the center’s choice; ** All patients received TAC (C0: 5–15 ng/mL) during the run-in phase; *** CS in both arms per the local practice; # variable period between the end of study visit in the H2307 study and the start of data collection for study H2406; ## treatment as per the local clinical practice during H2406. C0 – trough level; CS – corticosteroid; EOS – end of study; EVR – everolimus; HCC – hepatocellular carcinoma; LT – liver transplantation; M – month; MPA – mycophenolic acid; RND – randomization; TAC – tacrolimus. Created using Microsoft Office (2016, Microsoft). Figure 2. Patient disposition up to Month 60. * One patient discontinued the core study, but was enrolled in H2406. Created using Microsoft Office (2016, Microsoft).
Figure 2. Patient disposition up to Month 60. * One patient discontinued the core study, but was enrolled in H2406. Created using Microsoft Office (2016, Microsoft). Figure 3. Kaplan-Meier plot for proportion of patients (ITT population) free from HCC recurrence (A) and free from HCC recurrence or death (B). EVR+rTAC – everolimus plus reduced tacrolimus; HCC – hepatocellular carcinoma; ITT – intent-to-treat; sTAC – standard tacrolimus. Created using SAS version 9.4.
Figure 3. Kaplan-Meier plot for proportion of patients (ITT population) free from HCC recurrence (A) and free from HCC recurrence or death (B). EVR+rTAC – everolimus plus reduced tacrolimus; HCC – hepatocellular carcinoma; ITT – intent-to-treat; sTAC – standard tacrolimus. Created using SAS version 9.4. Figure 4. Evolution of renal function until Month 60 (in ITT population). eGFR – estimated glomerular filtration rate; MDRD4 – modification of diet in renal diseases-4; EVR+rTAC – everolimus plus reduced tacrolimus; M – month; RND – randomization; sTAC – standard tacrolimus; Tx – transplantation; W – week. Created using Microsoft Office (2016, Microsoft).
Figure 4. Evolution of renal function until Month 60 (in ITT population). eGFR – estimated glomerular filtration rate; MDRD4 – modification of diet in renal diseases-4; EVR+rTAC – everolimus plus reduced tacrolimus; M – month; RND – randomization; sTAC – standard tacrolimus; Tx – transplantation; W – week. Created using Microsoft Office (2016, Microsoft). Tables
 Table 1. Demographics and baseline characteristics in ITT patients who followed to the H2406 study.
Table 1. Demographics and baseline characteristics in ITT patients who followed to the H2406 study. Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint.
Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint. Table 3. Efficacy results for patients in ITT population.
Table 3. Efficacy results for patients in ITT population. Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT).
Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT). Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT).
Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT). Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*).
Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*). Table 7. Safety events for the patients who entered in H2406 in the AT population.
Table 7. Safety events for the patients who entered in H2406 in the AT population. Table 1. Demographics and baseline characteristics in ITT patients who followed to the H2406 study.
Table 1. Demographics and baseline characteristics in ITT patients who followed to the H2406 study. Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint.
Table 2. Number and percentage of patients in either immunosuppressive regimen by randomized treatment group and timepoint. Table 3. Efficacy results for patients in ITT population.
Table 3. Efficacy results for patients in ITT population. Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT).
Table 4. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence (ITT). Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT).
Table 5. Kaplan-Meier estimates of the proportion of subjects free from HCC recurrence or death (ITT). Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*).
Table 6. Multivariate Cox regression analysis for time to HCC recurrence or death (ie, death by any cause or HCC recurrence, whichever occurred earlier; ITT population*). Table 7. Safety events for the patients who entered in H2406 in the AT population.
Table 7. Safety events for the patients who entered in H2406 in the AT population. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








