06 July 2021: Original Paper
Liver Transplantation: A 10-Year Low-Volume Transplant Center Experience in Kazakhstan
Jamilya Saparbay1ABCDEF*, Janat Spatayev1ADEF, Abylaikhan Sharmenov1AB, Shokan Aytbayev1BCD, Aizhan Uristenova1B, Adilbek Mukazhanov1BCDE, Asan Zhexembayev1ADEDOI: 10.12659/AOT.931786
Ann Transplant 2021; 26:e931786
Abstract
BACKGROUND: Liver transplantation is considered to be the best available treatment option for patients with liver failure. In Kazakhstan, the liver transplantation program was established a decade ago. In this study, we analyzed a low-volume transplant center experience of liver transplantation in Kazakhstan.
MATERIAL AND METHODS: Clinical data of the 64 consecutive liver transplantations from deceased and living donors between 2010 and 2020 were retrieved from electronic records. All data were retrospectively analyzed.
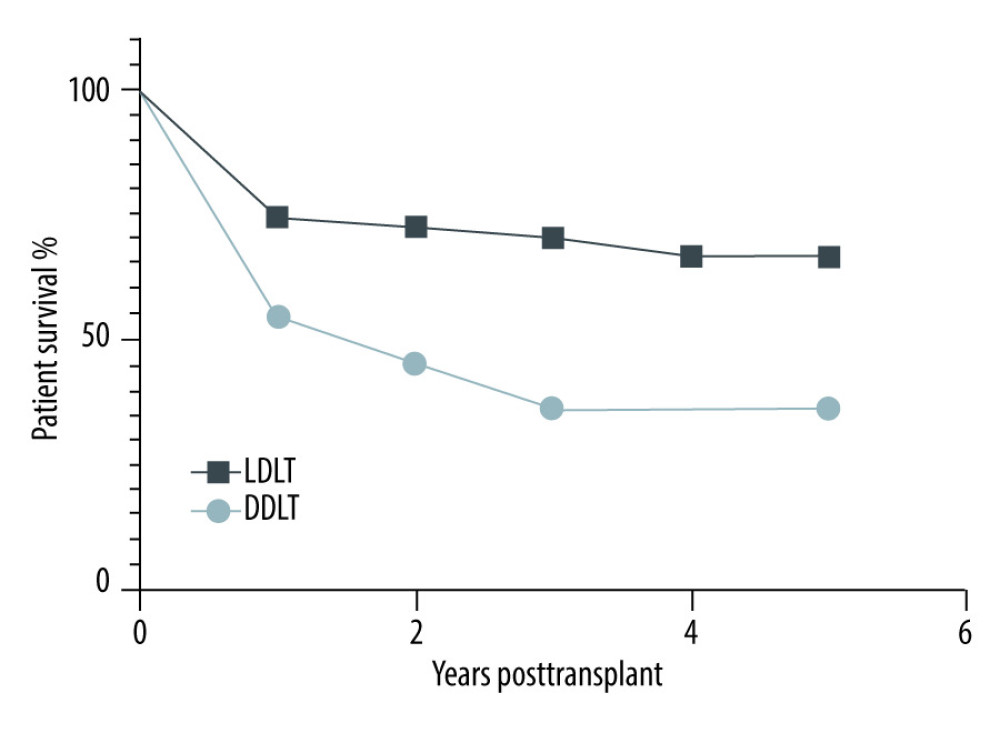
RESULTS: A total of 64 liver transplantations, 11 from deceased donors and 53 from living donors, were carried out in our center between 2010 and 2020. The mean age of the recipient was 44 years, 53% were female, and 47% were male. Hepatitis B+hepatitis D infection was the most common cause of end-stage liver disease (21 cases; 32.8%). The overall patient survival rates for 1, 3, and 5 years were 75%, 69.5%, and 59.6%, respectively, for recipients of a liver transplant from a living donor and 54.5%, 45.5%, and 39% for recipients of a liver transplant from a deceased donor.
CONCLUSIONS: Our clinical outcomes had a high rate of biliary and vascular complications that led to a low survival rate of the recipients. Starting the transplant program in Kazakhstan faced various challenges. In the early period, most transplantations were performed in collaboration with or under the guidance of transplant teams from Russia, Turkey, and South Korea. Improving surgical techniques and protocols of pre- and posttransplantation management could reduce the complications after transplantation.
Keywords: End stage liver disease, Liver Transplantation, Living Donors, Adolescent, Carcinoma, Hepatocellular, Hepatitis B, Hepatitis D, Kazakhstan, Liver Neoplasms, young adult
Background
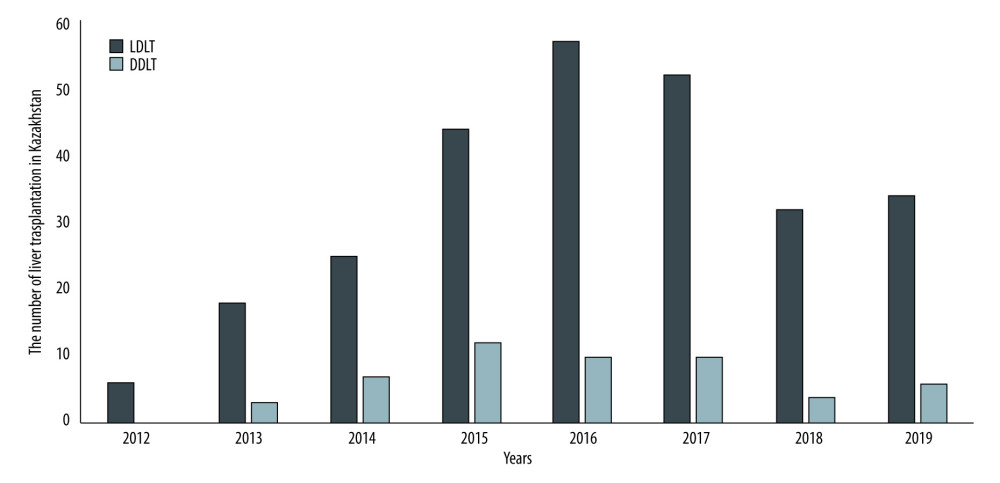
Liver transplantation is the only treatment option for patients with end-stage liver disease (ESLD) [1]. According to the Republican Center for the Coordination of Transplantation of the Ministry of Health of the Republic of Kazakhstan, 127 adults and 8 children are currently registered on the waiting list for liver transplantation [2]. The first successful deceased donor liver transplantation (DDLT) in Kazakhstan was performed in 1996. However, due to the lack of trained transplant surgeons and immunologists, and especially the absence of deceased donor organs, the first case was the only case for almost 10 years. Asian countries have the lowest rate of liver transplantation from deceased donors. By 2010, the rate of deceased organ donation was 0.05–6.0 per million on a population basis, while in the United States it was 33.9 per million [3]. Currently, the organ transplantation program in Kazakhstan is in its early stages. Similar to other Eastern countries, developing transplant services has met a variety of obstacles. An absence of education regarding the importance of organ donation and transplantation is common, alongside a lack of governmental support and poorly developed health care system, and these factors create a barrier for deceased organ donation development [4–6]. Thus, a notable discrepancy exists between the availability of organs and the patients awaiting liver transplantation in Kazakhstan. Living donation is the most attractive option in solving the organ shortage problem [7]. In Kazakhstan, the first successful liver transplantation from a living donor was performed in 2011 in collaboration with a transplant team from Belarus [8]. Since 2013, liver transplantation numbers have been steadily increasing in transplant centers of Kazakhstan (Figure 1). In Kazakhstan, live donor liver transplantation (LDLT) accounted for 80% of the total liver transplantation cases in 2019. Here, we present a 10-year single-center experience of liver transplantation in a low-volume transplant center in Kazakhstan.
Material and Methods
This retrospective study was conducted in compliance with the principles of the Declaration of Helsinki. The Local Ethics Committee of National Research (permit number #7) approved this study.
A total of 64 liver transplantations were performed in our center between January 2011, when the transplant program started in Kazakhstan, and October 2020. Of these transplantations, 11 were from deceased donors and 53 were from living donors. All data concerning recipients and donors were retrieved from electronic data, and we reviewed the course of the recipients. The follow-up period was more than 1 year in all cases. All recipients who received liver transplantation from a deceased or living donor were listed in the National Waiting List of the Republic of Kazakhstan.
Almost all grafts from living donors were right lobe hepatectomy, except 1 left hepatectomy. In all right hepatectomy cases, the middle hepatic vein was left in the donor remnant liver. Remnant liver volume, evaluated by computed tomography, was more than 35%, and graft-to-recipient body weight ratio >0.8 was acceptable in our center. An interposition synthetic graft was used for venous drainage of segments 5 and 8. Biliary reconstruction was completed either with duct-to-duct anastomoses or with hepaticojejunostomy.
Basiliximab (anti-interleukin-2 receptor antagonist) was used for induction therapy. For mainstay therapy, a calcineurin inhibitor-based immunosuppressive regimen was used in our center. Three-component therapy involved tacrolimus, steroid, and mycophenolate mofetil.
Oral nucleoside analogs were used in recipients with hepatitis B virus (HBV)-associated ESLD prior to liver transplantation and continued after liver transplantation as prophylaxis against recurrence.
The demographic and medical data of participants were analyzed retrospectively. Information on the age of recipients and donors, sex of recipients and donors, donor type, the causes of liver cirrhosis, hospital stay length, postsurgical complications, cold ischemic time, operation time, and bleeding volume is presented in Tables 1–3. The overall survival was defined as the time between the diagnosis date and death or the last follow-up visit. The difference between groups was considered significant if the
Results
Among 64 liver transplantations, 11 liver grafts were from deceased donors and 53 were from living donors (50 donors were relatives of the recipient, 3 were unrelated donors). There was 1 LDLT from an ABO-incompatible donor. The major causes of liver cirrhosis were hepatitis B and D in 21 cases (32.8%), hepatitis B in 9 (14%), primary biliary cirrhosis in 9 (14%), autoimmune hepatitis in 7 (10.9%), hepatitis C in 4 cases (6.3%), nonalcoholic steatohepatitis in 3 cases (4.7%), and hepatitis B, C, and D in 1 case (1.5%). In 2 cases, hepatocellular carcinoma was an indication for liver transplantation. Cryptogenic cirrhosis (6.3%) and Chanarin-Dorfman syndrome (1.6%) were rare indications for liver transplantation.
The mean age of transplant recipients was 44 years (range 15–63), with a slight majority of female recipients (53%) compared with male recipients (47%). The mean hospital stay was 40±25 days (range 11–196). Demographic and clinical characteristics of the liver recipients are shown in Table 1.
The mean age of liver donors was 31.5 years (range 19–54); 45 (70.35%) donors were men, and 19 (29.7%) were women. All living donors survived the procedure. Among living related donors, the donor was a parent of the recipient in 12 cases (22.6%), offspring in 17 cases (32.0%), and sibling in 21 cases (39.6%); the 3 unrelated living donors (5.67%) were the spouse of the recipient. No major complications were experienced in living donors. Demographic and clinical characteristics of the liver donors are shown in Table 2.
The mean cold ischemic time was 234.2±153 min, ranging from 330 to 830 min. The mean operation time was 555±114 min. The mean bleeding volume was 1835±1226 mL, ranging from 600 to 8000 mL (Table 3).
Most of the recipients experienced biliary complications (n=21, 32.8%). Among these 21 cases, anastomotic biliary stricture occurred in 8 (38%), bile leakage in 9 (42.8%), nonanastomotic biliary stricture in 1 (4.8%), and cholangitis in 3 (14.4%). Bile leakage was treated conservatively in 3 cases (33.3%) and reoperation was performed in 5 cases (55.5%) to remove the biloma or reanastomosis. Anastomotic biliary stricture needed reoperation with Roux-en-Y hepaticojejunostomy. When high bilirubinemia persisted even after reanastomosis in recipients, percutaneous transhepatic draining of the bile ducts was performed. Portal vein thrombosis occurred in 7 recipients and hepatic artery thrombosis in 4 cases, all after LDLT.
Acute rejection was observed in 7 patients (10.9%) after liver transplantation, with 1 case (9%) occurring after DDLT and 6 cases (11.3%) after LDLT. There was a significant association between donor type (living/deceased) and rejection rate (
The overall patient survival rates for 1, 3, and 5 years were 75%, 69.5%, and 59.6%, respectively, for liver transplant recipient from a living donor and 54.5%, 45.5%, and 39% for a liver recipient from a deceased donor (Figure 2).
Discussion
The National Research Oncology Center is one of the few transplant centers in Kazakhstan, the largest country of central Asia. In this study, we analyzed the outcome of 64 liver transplantations performed in our center since 2010. Clinico-demographic characteristics of donors, recipients, and outcomes were described in this paper. Our data showed that the main cause of ESLD among our patients was HBV, which accords with a systematic review indicating that Kazakhstan has the highest prevalence of HBV in central Asia [9]. Among a total of 64 liver transplantations, 53 recipients received a liver graft from living donors. Worldwide, LDLT has developed significantly, with the highest distribution in Asian countries [10]. The main disadvantage of the living donation is the donor hepatectomy, which can lead to various complications [11]. We made a strict selection of so-called ideal donors; the volume of remnant liver varied from 35% to 40%, liver steatosis was less than 10%, and the mean age was 32.5 years. In our study, living donors did not experience any major complications.
The liver transplant procedure itself is technically complicated, and partial liver transplantation is an even more challenging procedure. Numerous perioperative complications occurred in our case series. Biliary and vascular complications are the major cause of morbidity and mortality after liver transplantation. The most common complication in our study involved biliary complications. The incidence of biliary complications in liver recipients varies from 10% to 15% in DDLT and from 15% to 30% in LDLT [12]. The rate of biliary complications in our study was 32.8%. Most of the cases of biliary stenosis were treated with reoperation and hepaticojejunostomy or transhepatic draining. Hepaticojejunostomy often led to chronic cholangitis and biliary cirrhosis of the liver graft.
Our data revealed that DDLT patients had fewer episodes of acute rejection (9% vs 11.3%), which is not similar to other transplant centers’ data. We suggest that a higher rate of acute rejection in LDLT recipients might be due to a higher rate of hepatic artery thrombosis after LDLT.
Despite the high rate of acute rejection in LDLT patients, overall survival was significantly higher in LDLT recipients than in DDLT recipients (
This study is limited due to the small-size cohort and its retrospective character. A longer observation period, a higher number of liver transplantation cases, and improved donor and recipient selection are necessary for further work.
Conclusions
For developing countries, starting a liver transplant program is challenging due to many factors, such as insufficient financial support and lack of opportunity to train surgeons and other specialists. These factors are essential for the complicated procedure of liver transplantation and efficient follow-up. Improving surgical technique, preventing biliary and vascular complications, and overcoming the organ shortage problems remain to be achieved.
In conclusion, in Kazakhstan, similar to other developing countries, establishment of transplantation programs is essential for health care system improvement.
References
1. Murray KR, Carithers RL, AASLD practice guidelines: Evaluation of the patient for liver transplantation: Hepatology, 2005; 41; 1407-32
2. Republican Center for Coordination of Transplantation (RCCT), Healthcare Ministry of Kazakhstan, Statistics: Waiting List [in Russian]https://transplant.kz/ru/
3. Chen CL, Kabiling CS, Concejero AM, Why does living donor liver transplantation flourish in Asia?: Nat Rev Gastroenterol Hepatol, 2013; 10(12); 746-51
4. Altinors N, Haberal M, Brain death and transplant in Islamic countries: Exp Clin Transplant, 2016; 14(Suppl 3); 48-52
5. Shaheen FAM, Souqiyyeh MZ, How to improve organ donation in the MESOT countries: Ann Transplant, 2004; 9(1); 19-21
6. Shaheen FAM, Souqiyyeh MZ, Current obstacles to organ transplant in Middle Eastern countries: Exp Clin Transplant, 2015; 13(Suppl 1); 1-3
7. Abouna GM, Organ shortage crisis: Problems and possible solutions: Transplant Proc, 2008; 40(1); 34-38
8. Sultanaliev T, Zhexembayev A, Mukazhanov A, Liver transplant outcomes in a newly started program: Exp Clin Transplant, 2015; 13(Suppl 3); 120-22
9. Davlidova S, Haley-Johnson Z, Nyhan K, Prevalence of HIV, HCV and HBV in Central Asia and the Caucasus: A systematic review: Int J Infect Dis, 2021; 104; 510-25
10. Rela M, Reddy MS, Living donor liver transplant (LDLT) is the way forward in Asia: Hepatol Int, 2017; 11(2); 148-51
11. Sun Z, Yu Z, Yu S, Post-operative complications in living liver donors: A single-center experience in China: PLoS One, 2015; 10(8); e0135557
12. Daniel K, Said A, Early biliary complications after liver transplantation: Clin Liver Dis (Hoboken), 2017; 10(3); 63-67
Figures
Tables
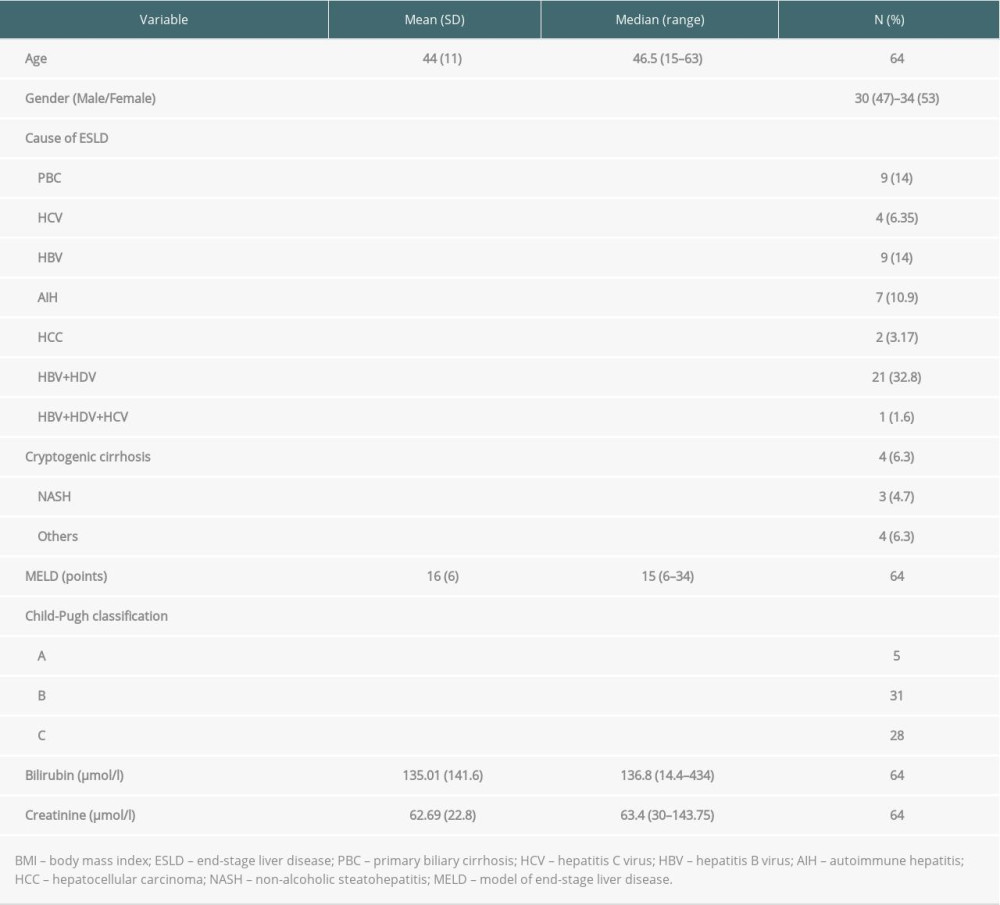 Table 1. Demographics and clinical characteristics of liver recipients.
Table 1. Demographics and clinical characteristics of liver recipients. Table 2. Demographics and clinical characteristics of liver donors.
Table 2. Demographics and clinical characteristics of liver donors.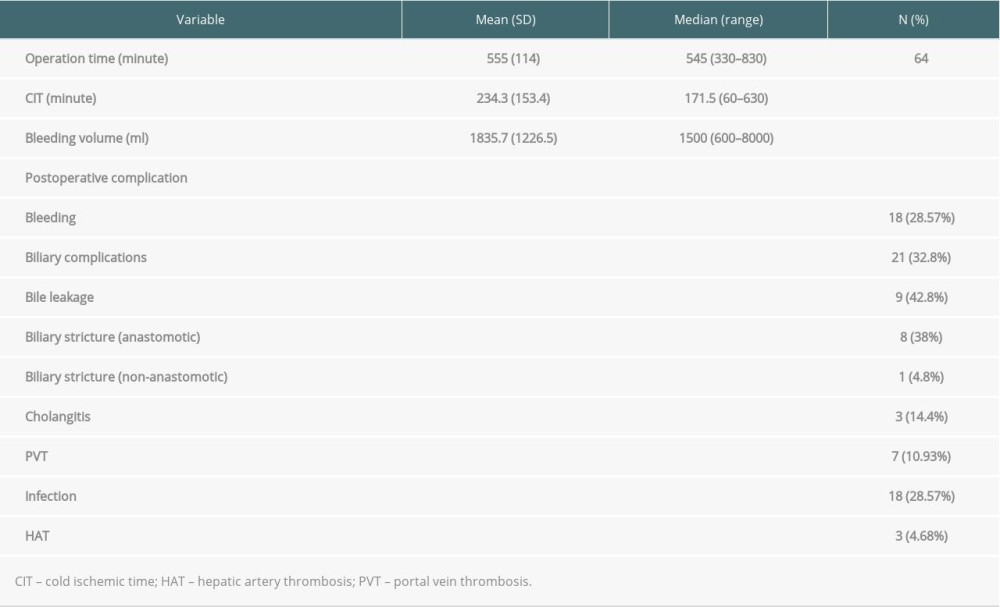 Table 3. Postoperative complications and surgical data.
Table 3. Postoperative complications and surgical data. Table 1. Demographics and clinical characteristics of liver recipients.
Table 1. Demographics and clinical characteristics of liver recipients. Table 2. Demographics and clinical characteristics of liver donors.
Table 2. Demographics and clinical characteristics of liver donors. Table 3. Postoperative complications and surgical data.
Table 3. Postoperative complications and surgical data. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860