15 March 2022: Case Report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Uremia, and Insulin-Dependent Diabetes
Xitao HongDOI: 10.12659/AOT.935860
Ann Transplant 2022; 27:e935860
Abstract
BACKGROUND: Abdominal organ cluster transplantation for the treatment of upper abdominal end-stage diseases is a serious conundrum for surgeons.
CASE REPORT: We performed clinical assessment of quadruple organ transplantation (liver, pancreas, duodenum, and kidney) for a patient with end-stage liver disease, post-chronic hepatitis B cirrhosis, uremia, and insulin-dependent diabetes mellitus, and explored the optimal surgical procedure. Simultaneous classic orthotopic liver, pancreas-duodenum, and heterotopic renal transplantation was performed on a 46-year-old man. The process was an improvement of surgery implemented with a single vascular anastomosis (Y graft of the superior mesenteric artery and the celiac artery open together in the common iliac artery). The pancreatic secretions and bile were drained through a modified uncut jejunal loop anastomosis, and the donor’s kidneys were placed in the right iliac fossa. The patient was prescribed basiliximab, glucocorticoid, tacrolimus, and mycophenolate mofetil for immunosuppression. The hepatic function recovered satisfactorily on postoperative day (POD) 3, and pancreatic function recovered satisfactorily in postoperative month (POM) 1. Hydronephrosis occurred in the transplanted kidney, with elevated creatinine on POD 15. Consequently, renal pelvic puncture and drainage were performed. His creatinine dropped to a normal level on POD 42. No allograft rejections or other complications, like pancreatic leakage, thrombosis, or localized infections, occurred. The patient had normal liver, renal, and pancreas functions with insulin-independent after POD 365.
CONCLUSIONS: Simultaneous classic orthotopic liver, pancreas-duodenum, and heterotopic renal transplantation is a promising therapeutic option for patients with insulin-dependent diabetes combined with end-stage hepatic and renal disease, and our center’s experience can provide a reference for clinical multiorgan transplantation.
Keywords: Islets of Langerhans Transplantation, Kidney Transplantation, Liver Cirrhosis, Liver Transplantation, Organ Transplantation, Transplantation, Isogeneic, Diabetes Mellitus, Type 1, Duodenum, Hepatitis B, Humans, Insulin, Male, Pancreas, Pancreas Transplantation, Uremia
Background
Abdominal multiorgan transplantation is effective for patients with multiple organ failure (MOF). Combined liver-kidney transplantation (CLKT), simultaneous kidney-pancreas transplantation (SKPT) with enteric drainage of pancreatic exocrine secretions, and simultaneous orthotopic liver and heterotopic pancreas-duodenum transplantation (SLPT) can be indicated for patients with failure of both organs. All of these combinations have been reported in the literature [1].
Combined liver, pancreas-duodenum, and kidney transplantation (LPDKT) has only been performed in a few cases as a therapeutic approach for patients with end-stage liver disease combined with insulin-dependent diabetes mellitus with renal failure. Such patients are rare, and there is no causal connection among end-stage liver disease, diabetes, and chronic renal failure. Patients with simultaneous failure of 3 organs often have difficulty tolerating surgery due to their poor physical condition [2]. LPDKT is complex because the perioperative hemodynamics are unstable, and it is difficult to adjust the use of drugs during and after surgery, which makes successfully combined transplantation challenging. According to statistics, the 5-year survival rates of diabetic patients after liver transplantation are significantly lower than those of nondiabetic patients, so diabetic patients must strictly control their blood glucose level after organ transplantation to keep it in the normal range [3,4]. Herein, we discuss a case where 1 patient with end-stage liver disease, post-chronic hepatitis B cirrhosis, uremia, and insulin-dependent diabetes mellitus (IDDM) was successfully transplanted and recovered, the details of surgical techniques, and immunosuppressive treatment regimens.
Case Report
ETHICS STATEMENT:
The study was approved by the Institutional Ethics Committee for Clinical Research and Animal Trials of the First Affiliated Hospital of Sun Yat-sen University, and an informed consent waiver was granted by the IEC given the retrospective nature and minimal risk of the study. The patient gave consent for his details and accompanying images to be published.
Discussion
Classic orthotopic liver transplantation combined with heterotopic pancreas transplantation is the standard treatment for chronic hepatitis B cirrhosis, IDDM, and uremia. There are few reports (Table 3 [7–11]) on the treatment of combined LPDKT in patients with end-stage liver disease, renal disease, and IDDM because patients with all these conditions are rare. There is no conclusive evidence yet to suggest an internal causative relationship among end-stage liver disease, chronic renal failure, and diabetes.
Surgical treatment of patients with multiple organ failure is often difficult because of the patients’ poor physical condition. The complex surgical process, unstable hemodynamic changes [12], and many contradictions regarding the adjustments of medication during and after the operation increase the difficulty of a successful combined LPDKT. However, this type of surgery will reduce the complications of pancreas transplantation affecting a previously transplanted liver, such as vascular embolism, pancreatitis, or a local abscess. Moreover, it can also reduce the damage to the liver produced by pancreatic complications when reoperation is required, and it will facilitate the operation, but this technique is very difficult [13]. Some studies have shown that combined liver transplantation can reduce the recipient’s rejection of the transplanted organs, while the insulin and glucagon produced by the pancreas have nutritional protective effects on the liver [14,15]. The pancreas relies on portal vein reflux, which makes the metabolism of insulin more stable [16].
Therefore, combined pancreas transplantation should be considered for patients with organ failure complicated with diabetes. The grafts should all be taken from the same donor with a similar genetic background and antigenicity. This will produce fewer donor-specific antibodies than grafts from different donors. The symptoms of diabetes can be corrected at the same time as uremia and liver failure. Combined en bloc liver-pancreas transplantation has been proven to be effective in treating end-stage liver disease with IDDM [17,18]. Zhang et al [7] reported the long-term effects of liver-kidney-pancreas transplant for patients with advanced liver disease, hepatitis B cirrhosis, chronic renal failure, and chronic pancreatitis caused by insulin-dependent diabetes, and achieved satisfactory long-term effects after 14 years of follow-up, without any rejection or other complications. Kornberg et al [19] reported SLPT for 14 liver cirrhosis and IDDM patients. All recipients could discontinue insulin therapy shortly after the operation. The 7-year survival rate of the recipients was as high as 64.2%, and only 1 patient experienced diabetes relapse. Li et al [9] reported simultaneous LPDKT in a patient with hepatitis B cirrhosis, uremia, and IDDM. The patient was hospitalized for 47 days and did well with normalization of transplanted organ function. Therefore, combined LPDKT is effective in the treatment of diabetes with liver and kidney failure and has better long-term effects than combined liver-kidney transplantation.
Like the original technique of pancreas graft exocrine drainage, the bladder-drained grafts always had to be revised to enteric drainage owing to complications such as urinary tract infections, metabolic acidosis, and reflux pancreatitis from urinary retention. Bladder drainage may have a selective benefit to allow for an earlier diagnosis of graft rejection by allowing monitoring of urinary amylase levels, but its status has been questioned because of improvements in immunosuppression. Subsequently, enteric drainage becomes another most physiologic approach for drainage of exocrine secretions.
According to data from the United Network for Organ Sharing (UNOS) and the International Pancreatic Transplant Registry (IPTR) [20,21], from 2006 to 2016, 8.4% of pancreases underwent bladder drainage, 18.5% underwent enteric drainage using a Roux limb, and 66.4% underwent enteric drainage without a Roux limb. The survival rates of recipients and grafts for all strategies were similar. Enteric drainage is preferred because it can drain pancreatic secretions into the dysfunctional intestine, thereby transferring the contents of the small intestine from the transplanted duodenal-jejunal anastomosis. However, it can be easier to achieve graft rescue if the duodenum-jejunum anastomosis leaks when perforation occurs [22].
To improve the surgical process, the donor’s superior mesenteric artery and celiac arteries were anastomosed to a Y graft of the internal iliac artery and external iliac artery from the same donor to allow for a single vascular anastomosis (the common iliac artery) (Supplementary Figure 1). The artery and the recipient’s hepatic artery were anastomosed in an end-to-end fashion. The procedure of single opening vascular reconstruction is completed during the pruning of the organ clusters, which can appropriately prolong the vascular pedicle and shorten the anhepatic phase to make the anastomosis operation more convenient. It has been reported [23] that a long anhepatic phase will aggravate the ischemia-reperfusion injury (IRI) of the graft, which is of great significance for the recovery of postoperative organ function. After reperfusion of the organ cluster, we performed a modified uncut jejunal loop anastomosis (a Warren anastomosis) between the graft duodenum and recipient’s jejunum for exocrine pancreatic and biliary drainage. Warren anastomosis is a new anastomosis method for choledochojejunostomy, performed behind the antrum pyloricum after lifting the jejunum 15–20 cm away from the ligament of Treitz to the hilar of the liver and anastomosed end-to-side with the bile duct stump, and then the proximal jejunum is anastomosed side-to-side with the bile-draining jejunal loops 40 cm from the bile-enteric anastomosis to form a loop [24,25]. Compared with patients undergoing Roux-en-Y choledochojejunostomy, this surgical procedure is performed without cutting off the jejunum. Only the intestinal tube is ligated, and its normal electrophysiological conduction is not disrupted. Therefore, it is more conducive to the drainage of pancreatic juice, bile, and intestinal contents, reducing the incidence of intestinal loop motility disorders. The patient’s anus exhausts faster, and gastrointestinal function is recovered faster after surgery.
Donor organs are easy to procure and trim for multiple organ transplantation. Only 4 to 5 vessels need to be anastomosed during transplantation to make a jejunal anastomosis without a biliary anastomosis. In addition, the bile and pancreatic juice enter the recipient directly through the duodenum, which is more in line with the normal physiological function. Therefore, classic orthotopic liver transplantation, pancreas-partial duodenal transplantation, and heterotopic kidney transplantation are the best choices. In this case, the patient had end-stage liver disease, post-chronic hepatitis B cirrhosis, uremia, and insulin-dependent diabetes mellitus. After combined transplantation, exogenous insulin was stopped. The function of the liver and kidney and blood glucose levels returned to normal and satisfactory results were achieved.
Allograft rejection is still one of the important factors affecting the efficacy of organ cluster transplantation [26]. There is no perfect immunosuppressive program for multiple organ transplantation due to the small number of cases. At present, many researchers advocate that a combined immunosuppressive regimen should be used after hepatopancreas-duodenal organ cluster transplantation. The quadruple immunosuppressive regimen of this patient was based on basiliximab, tacrolimus, MMF, and Pred, and no allograft rejection occurred within 47 days after transplantation. Basiliximab was used to induce immunity at the end of the operation and on POD 4. The concentration of tacrolimus was stabilized to 8–10 μg/L in the first 3 months. The application of hormones can easily induce postoperative hyperglycemia and diabetes recurrence, and it is recommended that postoperative hormones be quickly reduced and discontinued early for such patients. Our patient recovered and was discharged home after 47 days of hospitalization, insulin-independent, and maintaining a healthy diet and good physical condition 365 days after transplant.
Decompensated cirrhosis secondary to hepatitis B is an absolute indication for liver transplantation, but it is necessary to prevent hepatitis B virus reinfection in the early period after surgery. We used high-dose anti-hepatitis B immunoglobulin to prevent hepatitis B virus infection during and after surgery, checked his HsBAb and HBV DNA regularly, and adjusted the dosage of anti-hepatitis B immunoglobulin. Preventing the recurrence of hepatitis is key for long-term survival [27].
This article reports a rare case in which the patient was diagnosed with decompensated cirrhosis secondary to hepatitis B with chronic renal function failure and uremia, IDDM, and kidney stones. We discussed the necessity of choosing this technique. The main advantage of this technique is its rapidity because of rebuilding the vessels in ex vitro and simplicity since it involves only 4 large vascular anastomoses and 1 duodeno-jejunostomy with no separate biliary anastomosis. This new choledochojejunostomy anastomosis method promotes the postoperative recovery of patients. We used a quadruple immunosuppressive regimen, including basiliximab, tacrolimus, mycophenolate mofetil (MMF), and prednisone acetate (Pred), and gradually transitioned to maintenance therapy with tacrolimus alone. We hope that our center’s experience can provide a reference for the selection and perioperative management of clinical multiorgan transplantation.
Conclusions
Simultaneous classic orthotopic liver, pancreas-duodenum, and heterotopic renal transplantation is a promising therapeutic option for patients with insulin-dependent diabetes combined with end-stage hepatic and renal disease, and our center’s experience can provide a reference for clinical multiorgan transplantation.
Figures
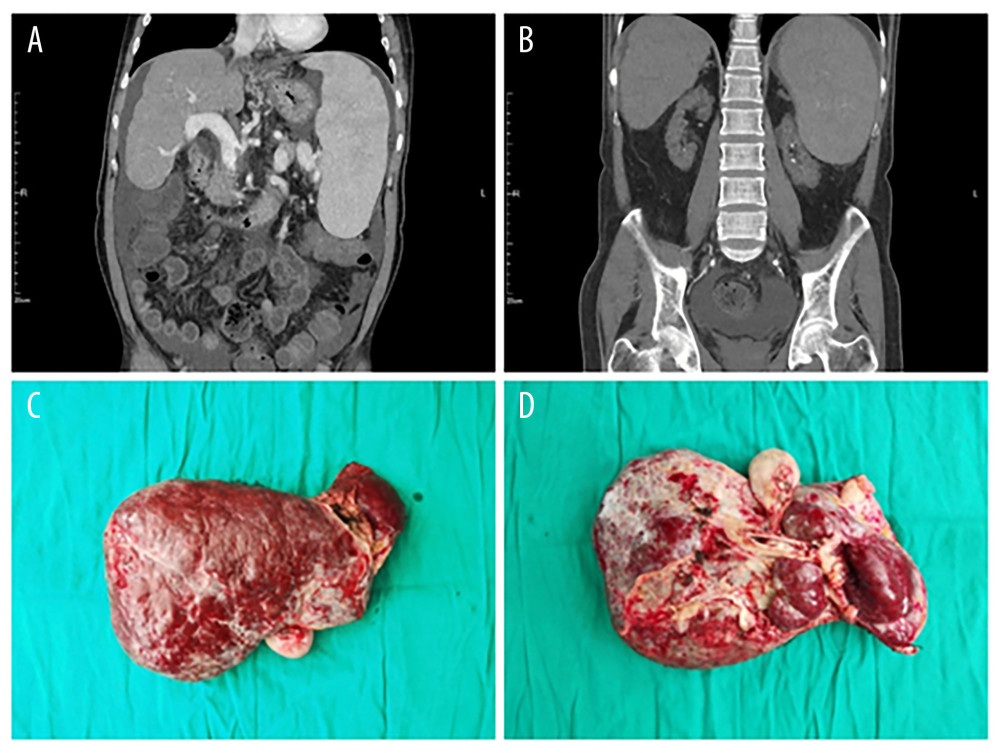 Figure 1. (A, B) Preoperative CT examination shows severe liver cirrhosis, splenomegaly, and atrophy of both kidneys. (C, D) Intraoperative resection of the diseased liver.
Figure 1. (A, B) Preoperative CT examination shows severe liver cirrhosis, splenomegaly, and atrophy of both kidneys. (C, D) Intraoperative resection of the diseased liver. 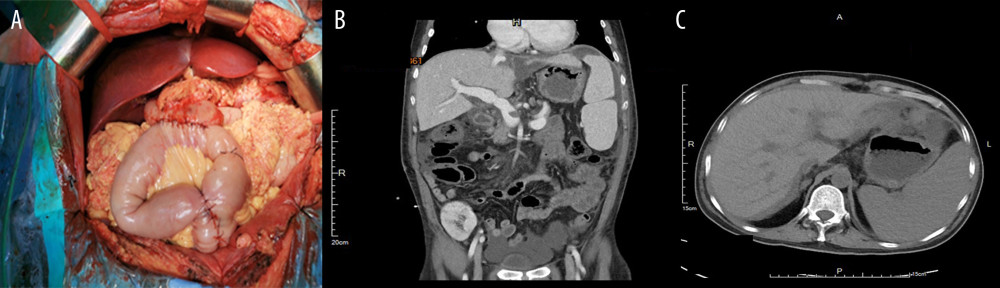 Figure 2. (A) Intraoperative anastomosis. (B, C) Postoperative CT scan.
Figure 2. (A) Intraoperative anastomosis. (B, C) Postoperative CT scan. 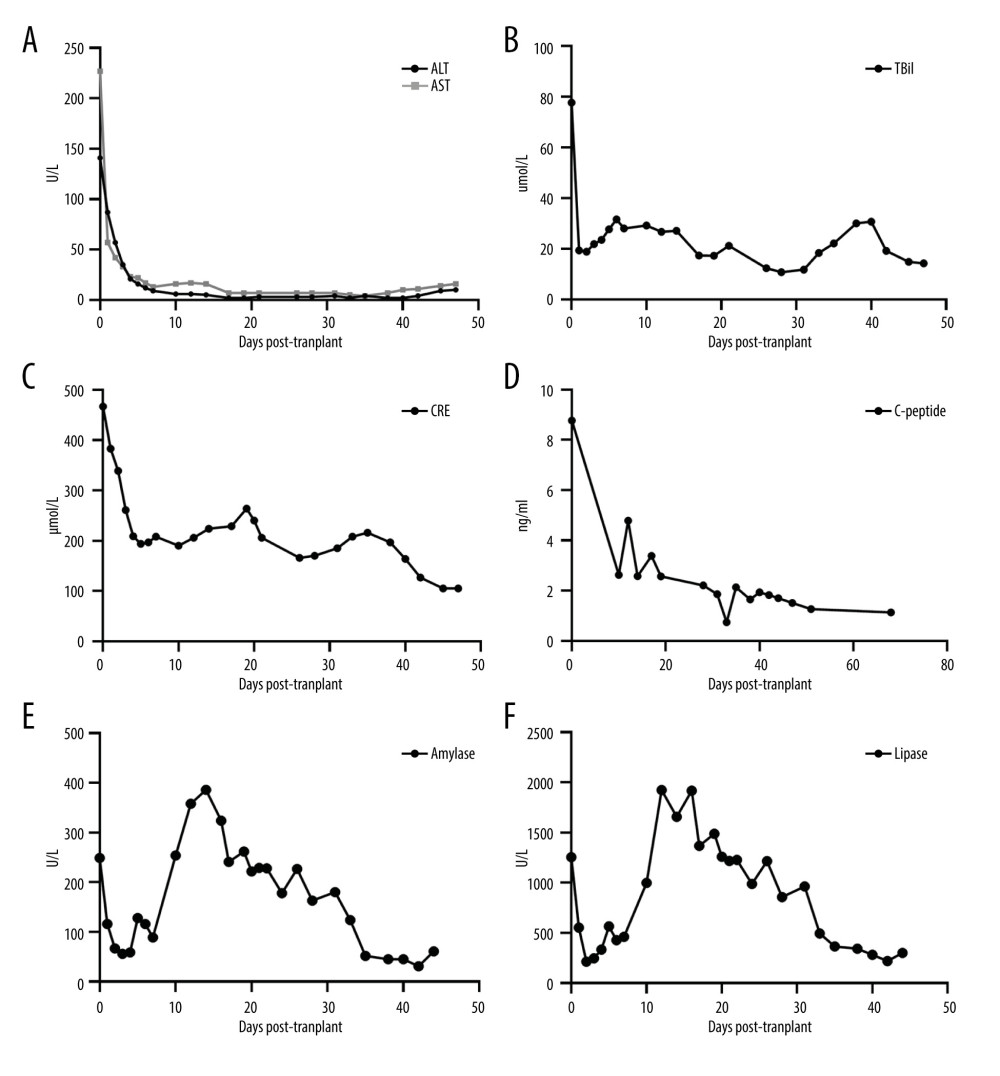 Figure 3. Changes in organ functions after transplantation. (A) Alanine aminotransferase (ALT) and aspartate aminotransferase (AST). (B) Total bilirubin (TBil). (C) Creatinine (CRE). (D) C-peptide. (E) Amylase. (F) Lipase.
Figure 3. Changes in organ functions after transplantation. (A) Alanine aminotransferase (ALT) and aspartate aminotransferase (AST). (B) Total bilirubin (TBil). (C) Creatinine (CRE). (D) C-peptide. (E) Amylase. (F) Lipase. References
1. Yu LX, Liu XY, Xu J, Clinical evaluation of abdominal multiorgan transplantation: Di Yi Jun Yi Da Xue Xue Bao, 2004; 24(2); 148-51
2. Xu J, Yu LX, Deng WFSimultaneous liver-pancreas-duodenum transplantation: One case report and review of the literature: Zhonghua Wai Ke Za Zhi, 2006; 44(3); 157-60 [in Chinese]
3. John PR, Thuluvath PJ, Outcome of liver transplantation in patients with diabetes mellitus: A case-control study: Hepatology, 2001; 34(5); 889-95
4. Cheng CY, Chen CH, Wu MF, Risk factors in and long-term survival of patients with post-transplantation diabetes mellitus: A retrospective cohort study: Int J Environ Res Public Health, 2020; 17(12); 4581
5. Nakache R, Merhav H, Rapid en bloc liver-pancreas-kidney procurement: More pancreas with better vascular supply: Transplant Proc, 2000; 32(4); 755-56
6. Samoylova ML, Borle D, Ravindra KV, Pancreas transplantation: Indications, techniques, and outcomes: Surg Clin North Am, 2019; 99(1); 87-101
7. Zhang G, Qin W, Yuan J, A 14-year follow-up of a combined liver-pancreas-kidney transplantation: Case report and literature review: Front Med (Lausanne), 2020; 7; 148
8. Tzakis AG, Nunnelley MJ, Tekin A, Liver, pancreas and kidney transplantation for the treatment of Wolcott-Rallison syndrome: Am J Transplant, 2015; 15(2); 565-67
9. Li J, Guo QJ, Cai JZ, Simultaneous liver, pancreas-duodenum and kidney transplantation in a patient with hepatitis B cirrhosis, uremia and insulin dependent diabetes mellitus: World J Gastroenterol, 2017; 23(45); 8104-8
10. Rivera E, Gupta S, Chavers B, En bloc multiorgan transplant (liver, pancreas, and kidney) for acute liver and renal failure in a patient with Wolcott-Rallison syndrome: Liver Transpl, 2016; 22(3); 371-74
11. Caicedo LA, Villegas JI, Serrano O, En-bloc transplant of the liver, kidney and pancreas: Experience from a Latin American transplant center: Am J Case Rep, 2017; 18; 114-18
12. Matuszkiewicz-Rowińska J, Wieliczko M, Małyszko J, Renal replacement therapy before, during, and after orthotopic liver transplantation: Ann Transplant, 2013; 18; 248-55
13. Benzing C, Hau HM, Atanasov G, Outcome and complications of combined liver and pancreas resections: A retrospective analysis: Acta Chir Belg, 2016; 116(6); 340-45
14. Haydon G, Neuberger J, Liver transplantation in cirrhotic patients with diabetes mellitus: Liver Transpl, 2001; 7(3); 234-37
15. Siegel AB, Lim EA, Wang S, Diabetes, body mass index, and outcomes in hepatocellular carcinoma patients undergoing liver transplantation: Transplantation, 2012; 94(5); 539-43
16. Gaber AO, Shokouh-Amiri H, Hathaway DK, Pancreas transplantation with portal venous and enteric drainage eliminates hyperinsulinemia and reduces postoperative complications: Transplant Proc, 1993; 25(1 Pt 2); 1176-78
17. Mekeel KL, Langham MR, Gonzalez-Perralta R, Combined en bloc liver pancreas transplantation for children with CF: Liver Transpl, 2007; 13(3); 406-9
18. Trotter JF, Bak TE, Wachs ME, Combined liver-pancreas transplantation in a patient with primary sclerosing cholangitis and insulin-dependent diabetes mellitus: Transplantation, 2000; 70(10); 1469-71
19. Kornberg A, Küpper B, Bärthel E, Combined en-bloc liver-pancreas transplantation in patients with liver cirrhosis and insulin-dependent type 2 diabetes mellitus: Transplantation, 2009; 87(4); 542-45
20. Gruessner AC, Gruessner RW, Pancreas transplantation of US and Non-US cases from 2005 to 2014 as reported to the United Network for Organ Sharing (UNOS) and the International Pancreas Transplant Registry (IPTR): Rev Diabet Stud, 2016; 13(1); 35-58
21. Ziaja J, Wullstein C, Woeste G, Bechstein WO, High duodeno-jejunal anastomosis as a safe method of enteric drainage in simultaneous pancreas and kidney transplantation: Ann Transplant, 2002; 7(3); 46-49
22. Amin I, Butler AJ, Defries G, A single-centre experience of Roux-en-Y enteric drainage for pancreas transplantation: Transpl Int, 2017; 30(4); 410-19
23. Ijtsma AJ, van der Hilst CS, de Boer MT, The clinical relevance of the anhepatic phase during liver transplantation: Liver Transpl, 2009; 15(9); 1050-55
24. Yang XW, Yang J, Wang K, A new anastomosis method for choledochojejunostomy by the way behind antrue pyloricum: Chin Med J (Engl), 2013; 126(24); 4633-37
25. Huang L, Liang LJ, Lai JMDetermination of the postoperative effect on intestinal structure and myoelectric motility in rabbits: Modified uncut jejunal loop versus Roux-en-Y biliodigestive anastomosis.]: Zhonghua Wai Ke Za Zhi, 2008; 46(11); 839-42 [in Chinese]
26. Miller ML, Daniels MD, Wang T, Spontaneous restoration of transplantation tolerance after acute rejection: Nat Commun, 2015; 6; 7566
27. Karasu Z, Ozacar T, Akyildiz M, Low-dose hepatitis B immune globulin and higher-dose lamivudine combination to prevent hepatitis B virus recurrence after liver transplantation: Antivir Ther, 2004; 9(6); 921-27
Figures
 Figure 1. (A, B) Preoperative CT examination shows severe liver cirrhosis, splenomegaly, and atrophy of both kidneys. (C, D) Intraoperative resection of the diseased liver.
Figure 1. (A, B) Preoperative CT examination shows severe liver cirrhosis, splenomegaly, and atrophy of both kidneys. (C, D) Intraoperative resection of the diseased liver. Figure 2. (A) Intraoperative anastomosis. (B, C) Postoperative CT scan.
Figure 2. (A) Intraoperative anastomosis. (B, C) Postoperative CT scan. Figure 3. Changes in organ functions after transplantation. (A) Alanine aminotransferase (ALT) and aspartate aminotransferase (AST). (B) Total bilirubin (TBil). (C) Creatinine (CRE). (D) C-peptide. (E) Amylase. (F) Lipase.
Figure 3. Changes in organ functions after transplantation. (A) Alanine aminotransferase (ALT) and aspartate aminotransferase (AST). (B) Total bilirubin (TBil). (C) Creatinine (CRE). (D) C-peptide. (E) Amylase. (F) Lipase. Tables
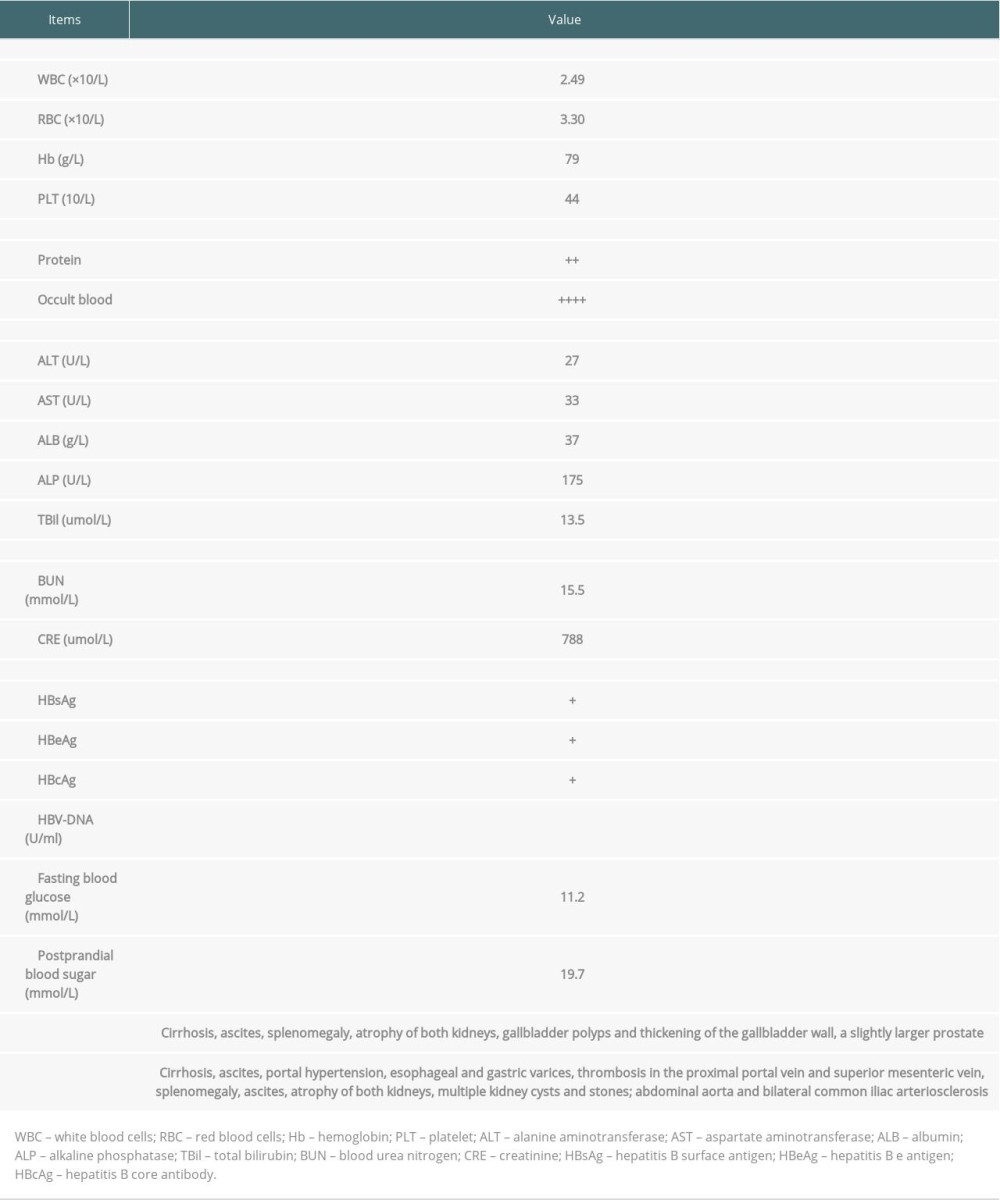 Table 1. Preoperative laboratory examination results of the patient.
Table 1. Preoperative laboratory examination results of the patient.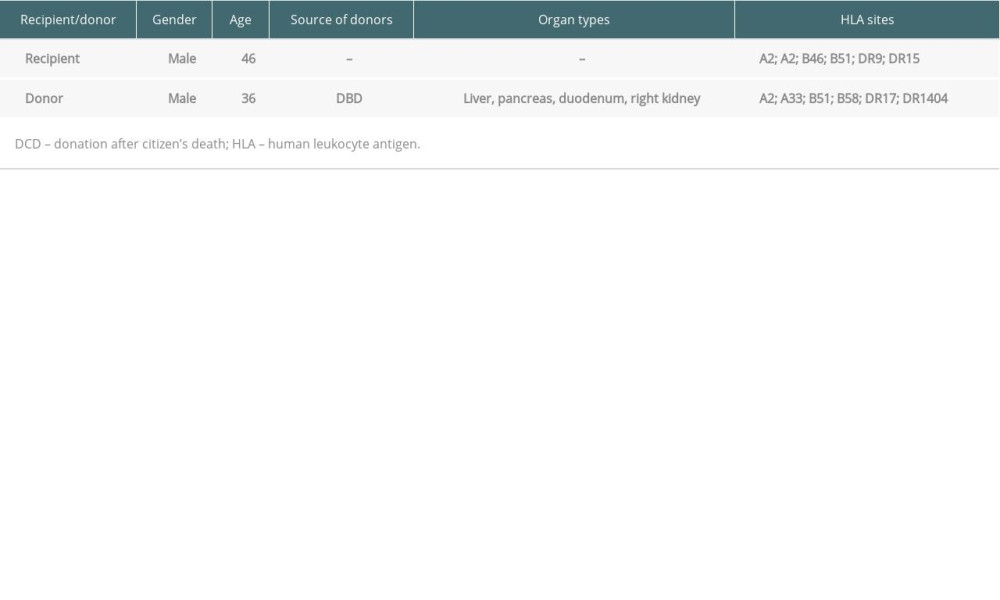 Table 2. Basic characteristics of the recipient and donors.
Table 2. Basic characteristics of the recipient and donors.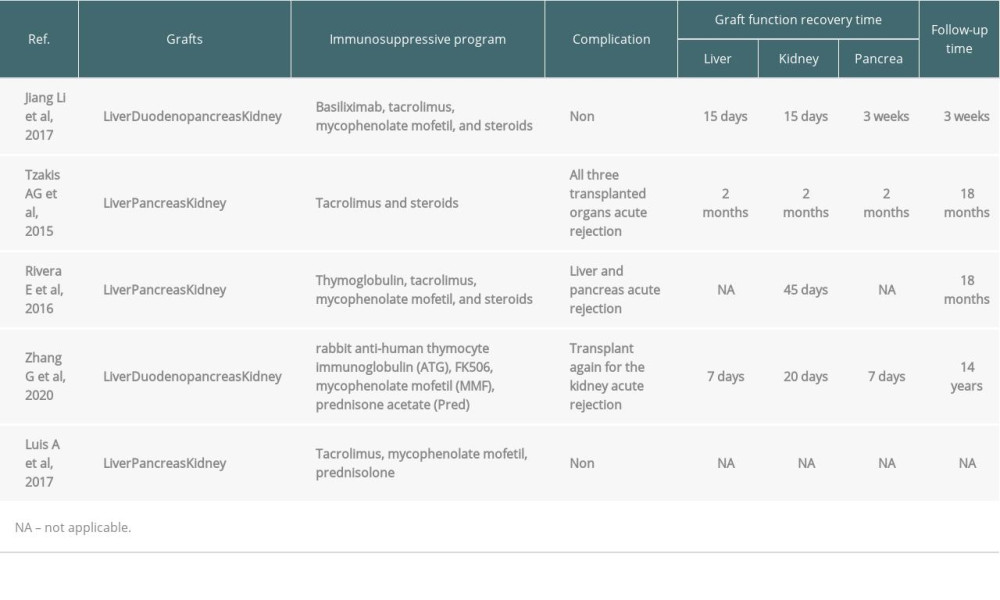 Table 3. Multiorgan liver-pancreas-kidney transplantation.
Table 3. Multiorgan liver-pancreas-kidney transplantation. Table 1. Preoperative laboratory examination results of the patient.
Table 1. Preoperative laboratory examination results of the patient. Table 2. Basic characteristics of the recipient and donors.
Table 2. Basic characteristics of the recipient and donors. Table 3. Multiorgan liver-pancreas-kidney transplantation.
Table 3. Multiorgan liver-pancreas-kidney transplantation. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








