23 May 2023: Original Paper
Safety and Efficacy of 4 mg·kg Sugammadex for Simultaneous Pancreas-Kidney Transplantation Recipients: A Prospective Randomized Trial
Jiancheng TangDOI: 10.12659/AOT.940211
Ann Transplant 2023; 28:e940211
Abstract
BACKGROUND: Simultaneous pancreas-kidney transplantation (SPK) is a time-consuming and important surgical procedure, which can provide a physiological mean of achieving normoglycemia and render patients free of dialysis. The potential clinical benefits of sugammadex include fast and predictable reverse deep neuromuscular blockade (NMB), but whether sugammadex affects the function of SPK grafts is uncertain.
MATERIAL AND METHODS: Forty-eight patients were studied and reversed deep NMB with either sugammadex (n=24) or neostigmine (n=24). The safety variables included serum creatinine (Scr), creatinine clearance rate (CCr), serum amylase (AMS), blood glucose (Glu), mean arterial pressure (MAP), and heart rate (HR). Secondary outcomes were time from administration of sugammadex/neostigmine at the scheduled time to recovery of a TOF ratio to 0.7 and 0.9, and post-acute pulmonary complications.
RESULTS: Scr at T2-6 was significantly lower than that at T0-1 (P<0.01), while CCr was higher (P<0.05). Between the 2 groups, Scr, CCr, and AMS were similar at the same timepoints (P>0.05). MAP, HR, and Glu were higher in group S than in group N at T1 (P<0.05). The recovery time of TOF=0.7 was 3 (2.4-4.2) min for group S and 12.1 (10.2-15.9) min for group N (P<0.001), and recovery time to TOFr ≥0.9 was 4.8 (3.6-7.1) min for group S and 23.5 (19.8-30.8) in group S. Compared to group N, group S had lower risk for post-acute pulmonary complications: supplemental oxygen requirements 0 vs 4 (16.7%), pulmonary atelectasis 0 vs 2 (0.83%), pneumonia 1 (4.2%) vs 3 (12.5%), and hypoxemia 1 (4.2%) vs 4 (16.7%).
CONCLUSIONS: Sugammadex administration is safe and effective for SPK transplantation recipients.
Keywords: sugammadex, Pancreas Transplantation, Perioperative Care, Kidney Transplantation, Neuromuscular Blockade, Humans, Neostigmine, gamma-Cyclodextrins, rocuronium, Prospective Studies, Neuromuscular Nondepolarizing Agents, Androstanols, Pancreas
Background
Diabetic nephropathy (DN) is one of the most serious consequences of diabetes mellitus (DM) and has become the leading cause of end-stage kidney disease worldwide [1]. Simultaneous pancreas-kidney transplantation (SPK), an effective therapeutic option, has been widely accepted for patients with end-stage renal disease (ESRD). In our center, a modified SPK transplantation technique for patients with ESRD effectively corrects renal and pancreatic dysfunction, and allows dialysis and insulin therapy to be discontinued. Deep neuromuscular blockade (NMB) is necessary to improve the surgical conditions for surgeons. However, excessive residual neuromuscular blocking agents (NMBAs) may cause delayed recovery from anesthesia, which can lead to severe complications such as postoperative hypoxemia, pulmonary atelectasis, and pneumonia. Insufficient reversal and re-occurrence of NMB increase the risk of postoperative pulmonary complications. As a novel NMB antagonist, sugammadex is a γ-cyclodextrin designed to selectively encapsulate aminosteroid nondepolarizing agent to reverse any degree of NMB [2]. Compared with neostigmine reversal, even deep NMB with sugammadex as the reversal agent can significantly avoid both adverse events. However, because sugammadex is almost completely excreted by the kidneys, with minimal or no self-metabolism [3], it was not initially recommended for use in ESRD patients. Few studies have been conducted on the use of sugammadex in patients with diabetes. Therefore, whether sugammadex affects the function of SPK grafts is still unknown. To address this issue, our study was designed to evaluate the efficacy and safety profile of sugammadex for SPK transplant recipients.
Material and Methods
STUDY DESIGN AND ETHICS APPROVAL:
This prospective, randomized, controlled clinical trial was conducted at the Second Affiliated Hospital of Guangzhou Medical University, China. The study protocol and consent form were approved by the Institutional Ethics Committee of the Second Affiliated Hospital of Guangzhou Medical University (approval number: 2020-hs-67, December 10, 2020). Written informed consent for participation in the trial was obtained from all patients before our evaluation, following the voluntary principle.
PATIENT RECRUITMENT:
The perioperative risk of all patients was calculated in accordance with the American Society of Anesthesiologists (ASA) score, and all were between II and III. Patients were aged 25–62 years and had body mass index (BMI) of 18.5–32.9 kg/m2. The exclusion criteria were: history of allergy to narcotic drugs, cardiopulmonary insufficiency, congestive heart failure, myasthenia gravis, neurological disease, mental illness, severe liver dysfunction, hyperkalaemia, malignant hyperthermia, were unwilling to participate in the study for any reason, or had participated in another study within the 3 months before enrolment. The inclusion criteria were: confirmed diabetic nephropathy on insulin therapy, ability to tolerate SPK surgery and immunosuppression, and creatinine clearance <15 mL/min or on dialysis.
INTERVENTIONS AND PERIOPERATIVE MANAGEMENT:
A total of 54 patients were evaluated for eligibility between July 2021 and May 2022. Each patient had received hemodialysis treatment approximately 1 day before the surgery to maintain a stable internal environment. Four patients were excluded from the study for failing to meet the inclusion criteria, such as refusing to participate, electrolyte disturbance (K+ >6 mmol·L−1) and inadequate outputted volume of urine (<1 ml·kg−1·h−1). Finally, 48 patients were successfully recruited, and subject numbers were entered into SPSS in a 1: 1 ratio by the recruiter to generate a randomization scheme. Eligible patients were randomly allocated to either group S or group N. Twenty-four patients were allocated to group S and received a sugammadex injection, whereas another 24 were allocated to group N and received the neostigmine-atropine complex (Figure 1). The anesthetists and assessors were unaware of the study-group assignments throughout the study.
After a patient was admitted to the operating room, radial artery (avoiding the arteriovenous fistula side) and right internal jugular vein puncture and catheterization were performed under local anesthesia by the same senior attending anesthesiologist. We routinely monitored the patient’s MAP, HR, central venous pressure (CVP), electrocardiograph (ECG), end-tidal carbon dioxide pressure (PETCO2), and pulse oxygen saturation (SpO2). After skin cleansing, 2 electrodes were positioned over the ulnar nerve 1 cm proximal to the wrist at a distance of 3–6 cm. A force transducer ring was attached to the thumb, and the fingers were secured to prevent movement during nerve stimulation.
Anesthesia was induced using a rapid sequence induction procedure. All patients received midazolam 0.06–0.1 mg·kg−1, sufentanil 0.3 μg·kg−1, propofol 1.5–2.0 mg·kg−1, and rocuronium 1 mg·kg−1 for anesthesia induction. Before NMBAs administration, TOF-Watch SX® (Organon, Dublin, Ireland) acceleromyography (electric current 40–60 mA) was calibrated with the built-in calibration model after 5 s of 50-Hz tetanic stimulation preceded by repetitive TOF stimulation for 1 min, and 3 min of repetitive TOF stimulation was required to ensure a stable response after calibration. After the TOF-Watch SX was set up, 1 mg·kg−1 rocuronium was administered within 5 s as a fast-running infusion. Then, tracheal intubation was performed. TOF stimulation was applied at 12-s intervals until the end of surgery.
After mechanical ventilation was initiated, a lung protection strategy, including low tidal volume ventilation, and positive end-expiratory pressure, in addition to lung recruitment manoeuvres during the entire operation, was used. Combined intravenous and inhalational anesthesia for the maintenance protocol and the end-tidal anesthetic gas (ETAG) for sevoflurane were maintained to an appropriate minimal alveolar concentration (MAC), according to the appropriate plasma concentration (Cp) target-controlled infusion (TCI) of propofol and remifentanil to achieve adequate depth of anesthesia. We continuously infused 0.6–0.9 mg·kg−1·h−1 rocuronium bromide to maintain deep NMB [TOF 0, post-tetanic count (PTC) 1–2] under neuromuscular monitoring, which was performed at the adductor pollicis muscle.
During the operation, PETCO2 was controlled at 30–40 mmHg (1 mmHg=0.133 kPa), and a BIS index (Aspect Medical Systems, Inc., Norwood, MA, USA) monitor was used to maintain the depth of anesthesia at a score of 40–60. An automatic anesthesia information system was linked to the patient’s physiological monitor to record BIS data every 5 min. All the patients received restrictive fluid management to maintain normovolemia utilizing the Flotrac sensor® (Edwards Lifesciences Corporation, Irvine, CA, USA) monitor, controlling the stroke volume variation (SVV) at 7–10%. In cases of hypotension, vasopressors were given if necessary to maintain the MAP above 65 mmHg. We injected 0.1 μg·kg−1 sufentanil and 1.0–1.5 mg·kg−1 parecoxib sodium within 30 min before skin incision to avoid remifentanil-induced postoperative hyperalgesia in both groups, which can provide better preemptive analgesia effect with less pulmonary complications. After the last skin suture, inhaled sevoflurane, injection of rocuronium, and remifentanil were discontinued. All the subjects were reversed from NMB with either sugammadex (4 mg·kg−1, group S) at the end of the surgery immediately or the neostigmine (70 μg·kg−1 with a 5 mg maximum dose, group N) injection when the presence of 1–5 PTCs (deep NMB) occurred. All the participants were intubated until the recovery of neuromuscular function to TOFr ≥0.9 with an adequate spontaneous respiration rate (RR ≥12) and TV ≥8 ml/kg. When the anesthesiologist judged the patient BIS index as ≥90 and their Steward Recovery Scores were ≥5, the patient was considered to be completely resuscitated and was returned to the inpatient ward. The other research indexes – Scr, CCr, Glu, AMS, MAP, HR, and SpO2 – were measured at the predetermined timepoints: the start of surgery (T0), 8 h (T1), 12 h (T2), 36 h (T3), 60 h (T4), 84 h (T5), and 108 h (T6) after the surgery. We also monitored the occurrence of hypoxemia (SpO2 <90%) after the extubation within 24 h in the inpatient ward (heart failure and any other underlying pulmonary diseases were excluded). The emerged lung complications, such as pulmonary atelectasis and pneumonia, were evaluated by new symptoms (cough, sputum, fever) and increased white blood cells or imagological examination within 7 days after the surgery.
STATISTICAL ANALYSIS:
We calculated [software: PASS 11 (NCSS, Kaysville, UT, USA)] a minimum requirement of 24 subjects in each arm of the study based on our pilot trial that 100% of patients in the sugammadex group and 40% of the patients in the neostigmine group would reach TOFr ≥0.9 within 40 min, and based on the criteria of a risk alpha of 5%, a power of 80% in a bilateral contrast and estimating a loss rate of 10%, a sample size of 24 subjects in each group was estimated.
The SPSS 22.0 software package was used for data analysis in this study. We used descriptive statistics to evaluate the data, and a nonparametric confidence interval (CI) approach was used to analyze the efficacy. All values of outcome indicators for MAP, HR, Scr, CCr, AMS, and Glu are presented as the means±SDs (standard deviation, SD). The homogeneity of parametric conditions in both groups was evaluated by Levene’s test. A paired
Results
The study flow chart is shown in Figure 1. The demographic characteristics, procedure duration, operation details, and perioperative complications of all the patients are presented in Table 1. No significant differences were observed between the groups with regard to age, sex, cultural background, BMI, ASA score, operation time, anesthesia time, dosage of rocuronium, graft survival rate, or other ventilation-related parameters. All the patients underwent hemodialysis for nearly the same length of time before SPK (P>0.05).
There was no difference between the 2 groups regarding total dosage of rocuronium [5.98 (4.57–7.13)
Nevertheless, the recovery time of the T4/T1 ratio (TOFr) to 0.7/0.9 differed significantly between the 2 groups (P<0.001). The median (95% CI) time of recovery to TOF=0.7 was 3 (2.4–4.2) min for group S and 12.1 (10.2–15.9) min for group N (Figure 4A, P<0.001), and recovery time to TOFr ≥0.9 was 4.8 (3.6–7.1) min for group S and 23.5 (19.8–30.8) min for group N (Figure 4A, P<0.001). The extubation time was 10 (8–16) min in group S
Discussion
SPK transplantation is the most effective treatment for patients with DM and ESRD, which has provided a physiological means of achieving normoglycemia and rendering patients free of dialysis. Our center began to apply a modified SPK transplantation technique for patients with ESRD to prevent serious perioperative complications. The requirements for improving anesthesia management to decrease the risk of perioperative complications became more stringent. This study is the first to evaluate the feasibility of the novel sugammadex for reversal of deep NMB in SPK transplantation recipients.
In this trial, we found that 4 mg·kg−1 sugammadex is safe and effective for patients undergoing SPK transplantation. Previous studies have also found that sugammadex was well tolerated and effective in reversing rocuronium-induced deep NMB, even in patients with severe renal impairment [4–8]. Our protocol can completely, rapidly, and predictably reverse deep NMB. However, in group N, the recovery time was considerably longer, which may be because inhalation anesthetics enhance the NMB effect and inhibit reversal with neostigmine but not sugammadex [9,10].
Because sugammadex is almost completely excreted by the kidneys, with minimal or no self-metabolism [3], sugammadex was not initially recommended for use in end-stage renal disease (ESRD) patients. However, our study has proved that sugammadex administration is safe and effective for patients undergoing SPK transplantation, which is in agreement with other studies [11–14]. The function of new transplant grafts recovered well after the sugammadex or neostigmine administration regimen, and all the functional assessment indicators (Scr, CCr, AMS, and Glu) quickly returned to normal ranges.
The efficacy of the sugammadex-rocuronium compound did not appear to be dependent on the traditional pathway of transhepatic or renal metabolism, and there was also no recurrence of NMB. For patient safety, we selected an appropriate time when the new kidney transplant grafts already had sufficient blood reperfusion and sufficient urine output (≥1 ml·kg−1·h−1) for the neuromuscular blocking antagonist administration.
In addition, some studies have indicated that sugammadex is dialysable. Cammu et al reported that residual sugammadex and the sugammadex-rocuronium complex can be efficiently removed through hemodialysis [15], and this also eliminated the risk of sugammadex accumulation in the SPK recipients and ensured the safety of patients in our trial.
The concentrations of Glu and AMS in both groups returned to normal after SPK transplantation, and their variation tendency was consistent in both groups S and N during the perioperative period. This proves that the 2 groups of neuromuscular blocking antagonists did not affect posttransplant pancreatic function.
It is interesting to note that MAP, HR, and Glu levels in group S had more dramatic fluctuations at T1 than those in group N. This is probably due to the strong stress response caused by the patient’s rapid resuscitation after sugammadex administration. Khuenl-Brady et al also found that patients who received sugammadex recovered from anesthesia faster than those treated with neostigmine after general anesthesia [16]. The muscle spindle afferents theory suggests that cerebral stimulation is produced by excited muscle stretch receptors [17], and sugammadex can increase the stretch receptor activity and afferent input, thus stimulating the brain arousal centers. After thorough and rapid reversal of NMB by sugammadex, activated muscle movement under light anesthesia conditions may cause a stronger stress response (coughing, movement, or bucking), which can lead to a higher mean arterial pressure, faster heart rate, and higher blood glucose concentration. Therefore, MAP, HR, and Glu levels in group S increased significantly, but there was no obvious harm to patients after the recovery of graft function. It may also be that a stronger preemptive analgesic regimen is necessary, such as a local nerve block technique carried out around the incision before the skin suture.
Residual NMB (rNMB) remains a potential problem in anesthesia, as it is a risk factor for postoperative pulmonary complications. Deep NMB often results in rNMB, and upper airway obstruction and postoperative pulmonary complications may be associated with it, especially for patients with advanced age and long-term ESRD. According to some studies [3,18,19], 4 mg·kg−1 sugammadex could reverse deep NMB in patients, even in those with renal impairment. Our study showed that the recovery time of NMB in group S was much shorter than that in group N, even approaching the recovery rate of healthy volunteers with normal renal function [6]. Through our neuromuscular monitoring, rNMB or recurarization did not occur, which guaranteed avoidance of post-acute pulmonary complications. Our results also supported the conclusion that a significant risk of postoperative residual weakness exists when neostigmine is used to reverse deep NMB [20]. Adopting multiple strategies, such as reversing with sugammadex and using a quantitative neuromuscular monitor, can decrease the risk of postoperative pulmonary complications. The incidence of perioperative complications such as hypoxemia, pulmonary atelectasis, and pneumonia in group S was significantly lower than that in group N, which is consistent with many previous studies [21–23].
There are some limitations to our trial that should be considered. First, because subjects were hard to recruit, the number of patients was relatively small. Second, the rehabilitation was conducted in local hospitals, and patients also returned regularly for follow-up visits, but we did not collect data for this phase. An additional limitation is the single-center nature of this study. We believe that expanding the scope of the study and the number of cases would lead to more meaningful results.
Conclusions
To conclude, the efficacy and safety of 4 mg·kg−1 sugammadex were confirmed to reverse deep rocuronium-induced NMB in patients undergoing SPK transplantation.
Figures
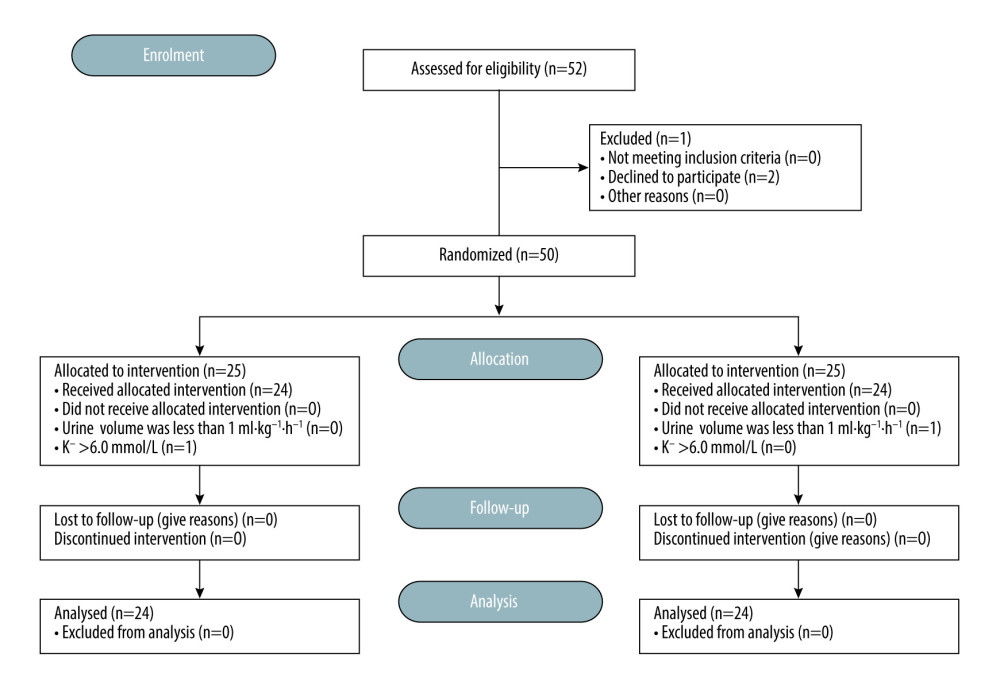 Figure 1. Patient flow through the study, in accordance with CONSORT guidelines. (This figure was made by Microsoft® Word 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 1. Patient flow through the study, in accordance with CONSORT guidelines. (This figure was made by Microsoft® Word 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). 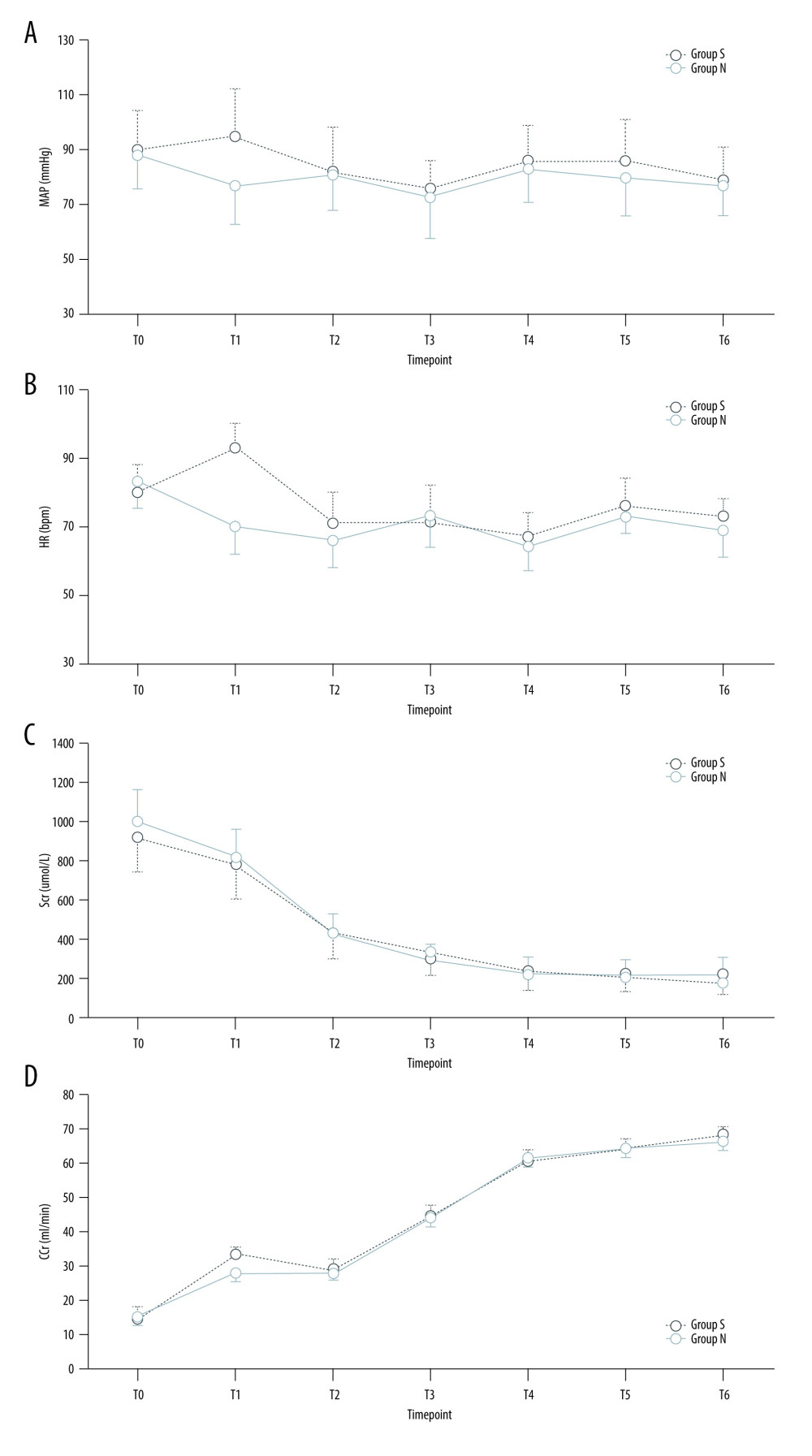 Figure 2. (A) Tendency of MAP of 2 groups. (B) Tendency of HR of 2 groups. (C) Tendency of Scr of 2 groups. (D) Tendency of CCr of 2 groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase; Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 2. (A) Tendency of MAP of 2 groups. (B) Tendency of HR of 2 groups. (C) Tendency of Scr of 2 groups. (D) Tendency of CCr of 2 groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase; Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). 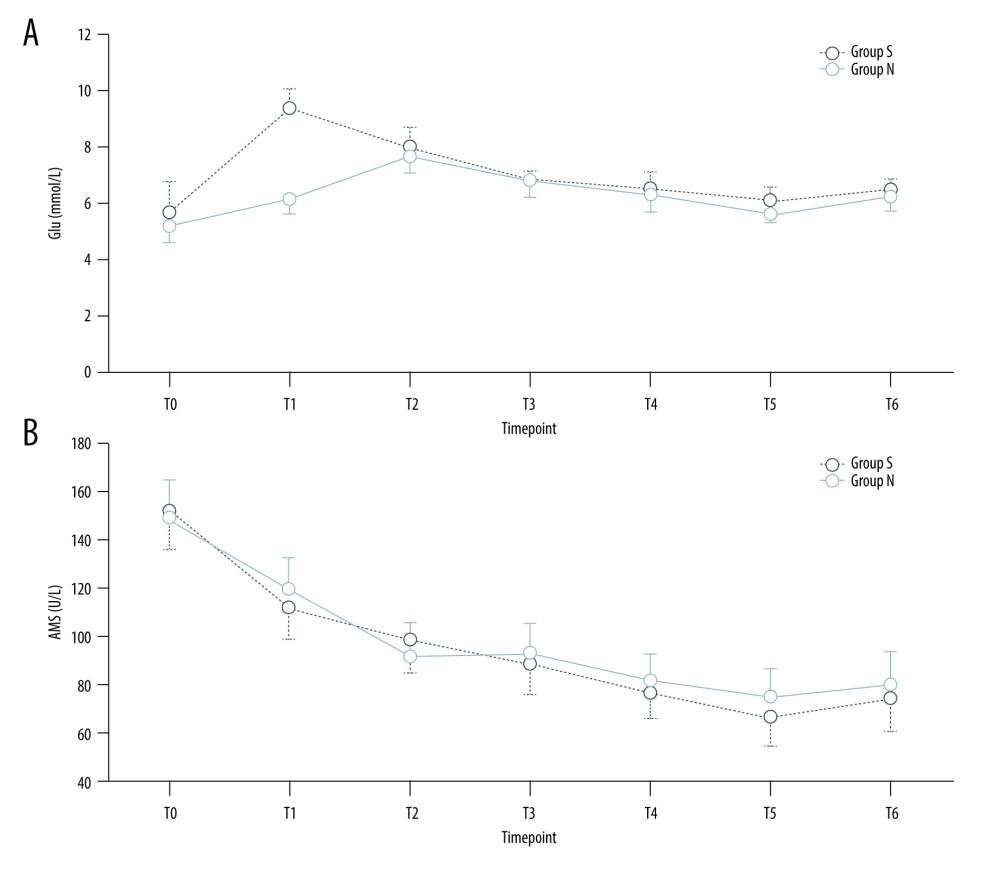 Figure 3. (A) Tendency of Glu of 2 groups. (B) Tendency of AMS of 2 groups. Plots of MAP, HR, Scr, CCr, Glu, and AMS of patients in both groups S (deep blue) and N (light blue) in each timepoint. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP, mean arterial pressure; HR, heart rate; Scr, serum creatinine; CCr, creatinine clearance rate; AMS, serum amylase and Glu, blood glucose; S, sugammadex; N, neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 3. (A) Tendency of Glu of 2 groups. (B) Tendency of AMS of 2 groups. Plots of MAP, HR, Scr, CCr, Glu, and AMS of patients in both groups S (deep blue) and N (light blue) in each timepoint. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP, mean arterial pressure; HR, heart rate; Scr, serum creatinine; CCr, creatinine clearance rate; AMS, serum amylase and Glu, blood glucose; S, sugammadex; N, neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). 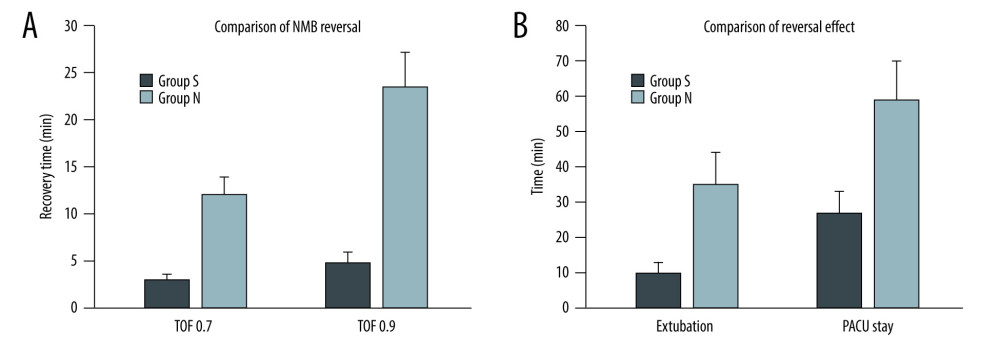 Figure 4. (A) Time of recovery to TOF=0.7/0.9 for both groups. (B) Extubation time and length of PACU stay in both groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase, Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 4. (A) Time of recovery to TOF=0.7/0.9 for both groups. (B) Extubation time and length of PACU stay in both groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase, Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). References
1. Umanath K, Lewis JB, Update on diabetic nephropathy: Core curriculum 2018: Am J Kidney Dis, 2018; 71; 884-95
2. Herring WJ, Woo T, Assaid CA, Sugammadex efficacy for reversal of rocuronium- and vecuronium-induced neuromuscular blockade: A pooled analysis of 26 studies: J Clin Anesth, 2017; 41; 84-91
3. Peeters P, Passier P, Smeets J, Sugammadex is cleared rapidly and primarily unchanged via renal excretion: Biopharm Drug Dispos, 2011; 32; 159-67
4. Panhuizen IF, Gold SJ, Buerkle C: Br J Anesth, 2015; 114; 777-84
5. Kim YS, Lim BG, Won YJ, Efficacy and safety of sugammadex for the reversal of rocuronium-induced neuromuscular blockade in patients with end-stage renal disease: A systematic review and meta-analysis: Medicina (Kaunas), 2021; 57(11); 1259
6. Carlos RV, Torres ML, de Boer HD, The use of rocuronium and sugammadex in paediatric renal transplantation: Two case reports: Eur J Anaesthesiol, 2016; 33; 383-86
7. Jones RK, Caldwell JE, Brull SJ, Reversal of profound rocuronium-induced blockade with sugammadex: A randomized comparison with neostigmine: Anesthesiology, 2008; 109(5); 816-24
8. Hristovska AM, Duch P, Allingstrup M, Efficacy and safety of sugammadex versus neostigmine in reversing neuromuscular blockade in adults: Cochrane Database Syst Rev, 2017; 8; CD012763
9. Lowry DW, Mirakhur RK, McCarthy GJ, Neuromuscular effects of rocuronium during sevoflurane, isoflurane, and intravenous anesthesia: Anesth Analg, 1998; 87(4); 936-40
10. Reid JE, Breslin DS, Mirakhur RK, Neostigmine antagonism of rocuronium block during anesthesia with sevoflurane, isoflurane or propofol: Can J Anaesth, 2001; 48(4); 351-55
11. Dias BH, Rana AAM, Olakkengil SA, Development and implementation of an enhanced recovery after surgery protocol for renal transplantation: ANZ J Surg, 2019; 89; 1319-23
12. Staals LM, Snoeck MM, Driessen JJ, Multicentre, parallel-group, comparative trial evaluating the efficacy and safety of sugammadex in patients with end-stage renal failure or normal renal function: Br J Anesth, 2008; 101; 492-97
13. Staals LM, Snoeck MM, Driessen JJ, Reduced clearance of rocuronium and sugammadex in patients with severe to end-stage renal failure: A pharmacokinetic study: Br J Anesth, 2010; 104; 31-39
14. Cammu G, Van Vlem B, van den Heuvel M, Dialysability of sugammadex and its complex with rocuronium in intensive care patients with severe renal impairment: Br J Anesth, 2012; 109; 382-90
15. Souza CM, Tardelli MA, Tedesco H, Efficacy and safety of sugammadex in the reversal of deep neuromuscular blockade induced by rocuronium in patients with end-stage renal disease: A comparative prospective clinical trial: Eur J Anaesthesiol, 2015; 32; 681-86
16. Khuenl-Brady KS, Wattwil M, Vanacker BF, Sugammadex provides faster reversal of vecuronium-induced neuromuscular blockade compared with neostigmine: A multicenter, randomized,controlled trial: Anesth Analg, 2010; 110; 64-73
17. Lanier WL, Iaizzo PA, Milde JH, The cerebral and systemic effects of movement in response to a noxious stimulus in lightly anesthetized dogs. Possible modulation of cerebral function by muscle afferents: Anesthesiology, 1994; 80(2); 392-401
18. Kang WS, Oh CS, Rhee KY, Deep neuromuscular blockade during spinal surgery reduces intra-operative blood loss: A randomised clinical trial: Eur J Anaesthesiol, 2020; 37(3); 187-95
19. Matsui M, Konishi J, Suzuki T, Reversibility of rocuronium-induced deep neuromuscular block with sugammadex in infants and children – a randomized study: Biol Pharm Bull, 2019; 42(10); 1637-640
20. Saager L, Maiese EM, Bash LD, Incidence, risk factors, and consequences of residual neuromuscular block in the United States: The prospective, observational, multicenter RECITE-US study: J Clin Anesth, 2019; 55; 33-41
21. Han J, Ryu JH, Koo BW, Effects of sugammadex on post-operative pulmonary complications in laparoscopic gastrectomy: A retrospective cohort study: J Clin Med, 2020; 9(4); 1232
22. Yu J, Park JY, Lee Y, Sugammadex versus neostigmine on postoperative pulmonary complications after robot-assisted laparoscopic prostatectomy: A propensity score-matched analysis: J Anesth, 2021; 35(2); 262-69
23. Kheterpal S, Vaughn MT, Dubovoy TZ, Sugammadex versus neostigmine for reversal of neuromuscular blockade and postoperative pulmonary complications (STRONGER): A multicenter matched cohort analysis: Anesthesiology, 2020; 132(6); 1371-81
Figures
 Figure 1. Patient flow through the study, in accordance with CONSORT guidelines. (This figure was made by Microsoft® Word 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 1. Patient flow through the study, in accordance with CONSORT guidelines. (This figure was made by Microsoft® Word 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). Figure 2. (A) Tendency of MAP of 2 groups. (B) Tendency of HR of 2 groups. (C) Tendency of Scr of 2 groups. (D) Tendency of CCr of 2 groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase; Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 2. (A) Tendency of MAP of 2 groups. (B) Tendency of HR of 2 groups. (C) Tendency of Scr of 2 groups. (D) Tendency of CCr of 2 groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase; Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). Figure 3. (A) Tendency of Glu of 2 groups. (B) Tendency of AMS of 2 groups. Plots of MAP, HR, Scr, CCr, Glu, and AMS of patients in both groups S (deep blue) and N (light blue) in each timepoint. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP, mean arterial pressure; HR, heart rate; Scr, serum creatinine; CCr, creatinine clearance rate; AMS, serum amylase and Glu, blood glucose; S, sugammadex; N, neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 3. (A) Tendency of Glu of 2 groups. (B) Tendency of AMS of 2 groups. Plots of MAP, HR, Scr, CCr, Glu, and AMS of patients in both groups S (deep blue) and N (light blue) in each timepoint. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP, mean arterial pressure; HR, heart rate; Scr, serum creatinine; CCr, creatinine clearance rate; AMS, serum amylase and Glu, blood glucose; S, sugammadex; N, neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). Figure 4. (A) Time of recovery to TOF=0.7/0.9 for both groups. (B) Extubation time and length of PACU stay in both groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase, Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation).
Figure 4. (A) Time of recovery to TOF=0.7/0.9 for both groups. (B) Extubation time and length of PACU stay in both groups. Scr and AMS at T2–6 were significantly lower than that at T0–1 (P<0.01), CCr at T2–6 was significantly higher than that at T0–1 (P<0.01). There was no significantly significant difference in the concentration of Scr, CCr, and AMS between the 2 groups at the same timepoint (P>0.05). MAP, HR, and Glu at T1 in group S were higher compared with group N (P<0.05). MAP – mean arterial pressure; HR – heart rate; Scr – serum creatinine; CCr – creatinine clearance rate; AMS – serum amylase, Glu – blood glucose; S – sugammadex; N – neostigmine. T0, the start of surgery; 8 hours (T1), 12 hours (T2), 36 hours (T3), 60 hours (T4), 84 hours (T5), and 108 hours (T6) after the surgery completed. (This figure was made using Microsoft® Excel® 2019MSO (2303 Build 16.0.16227.20202) 64, Microsoft Corporation). Tables
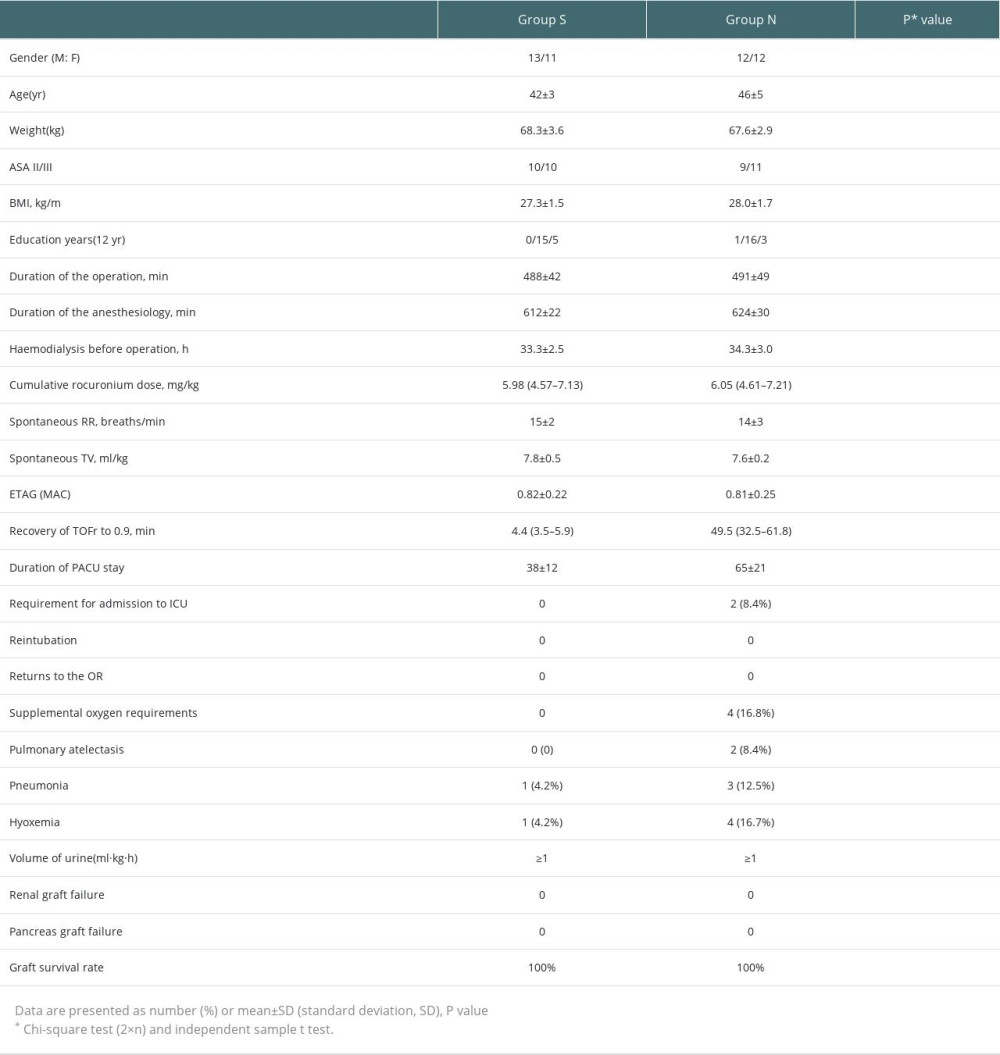 Table 1. The patient characteristics and details of surgery between the 2 groups.
Table 1. The patient characteristics and details of surgery between the 2 groups.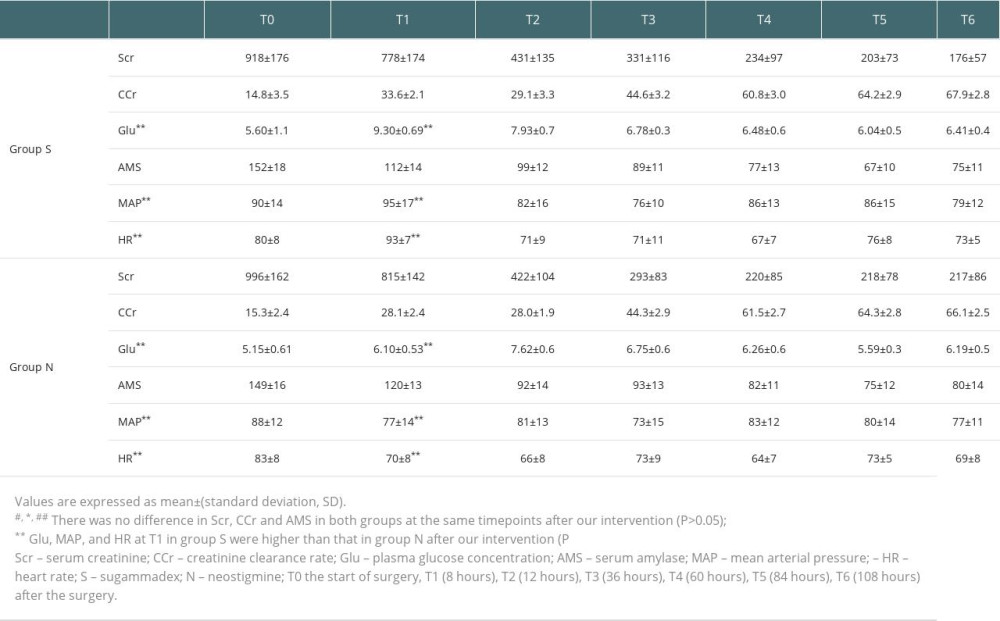 Table 2. The changes of Scr, CCr, AMS, Glu, MAP and HR of two groups patients.
Table 2. The changes of Scr, CCr, AMS, Glu, MAP and HR of two groups patients. Table 1. The patient characteristics and details of surgery between the 2 groups.
Table 1. The patient characteristics and details of surgery between the 2 groups. Table 2. The changes of Scr, CCr, AMS, Glu, MAP and HR of two groups patients.
Table 2. The changes of Scr, CCr, AMS, Glu, MAP and HR of two groups patients. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








