08 April 2025: Original Paper
Long-Term Outcomes of Left Renal Vein Ligation in Living Donor Liver Transplantation: A 20-Year Study
Hyo Jung Ko DOI: 10.12659/AOT.947492
Ann Transplant 2025; 30:e947492
Abstract
BACKGROUND: Persistence of large spontaneous splenorenal shunt (SSRS) can result in graft failure in adult living donor liver transplantation (LDLT) due to portal flow steal; thus, it is necessary to block SSRS to ensure sufficient portal blood flow.
MATERIAL AND METHODS: We performed a retrospective 20-year observational follow-up study subsequent to a prior prospective study to evaluate the long-term outcomes following ligation of the proximal left renal vein (LRV). Between October 2001 and January 2005, 44 liver cirrhosis patients underwent LDLT with LRV ligation. These patients were followed up until April 2024 or patient death.
RESULTS: Portal flow was significantly increased after LRV ligation. Renal function recovered uneventfully after LDLT in 40 patients. Eighteen patients died due to cancer recurrence (n=6), pneumonia (n=3), and other causes (n=9), thus 1-, 5-, 10-, and 20-year overall patient survival rates were 95.5%, 86.4%, 81.8%, and 59.1%, respectively. Solitary atrophy of the left kidney was not observed. SSRS was completely resolved in 20 patients, but the other 20 patients showed persistently identifiable SSRS of variable extents to date or at patient death. The LRV was reopened to make large SSRS in another 4 patients, in which retrograde transvenous obliteration was performed in 2 for variceal bleeding control and in another 2 for portal flow augmentation.
CONCLUSIONS: This study demonstrated that LRV ligation is a safe and effective method to control SSRS. Currently, direct ligation of the SSRS is preferred, but LRV ligation still can be a good therapeutic option when direct access to SSRS is not feasible.
Keywords: Hemodynamics, Liver Cirrhosis, Transplantation, Humans, Renal Veins, Male, Liver Transplantation, Female, Living Donors, Middle Aged, Retrospective Studies, adult, Ligation, Treatment Outcome, Follow-Up Studies
Introduction
In patients with liver cirrhosis and portal hypertension, it is common for portal blood flow to be redirected into the systemic circulation through collateral vessels. As liver cirrhosis progresses, increased intrahepatic vascular resistance can lead to a reversal of portal flow, causing reduced liver perfusion [1–5]. When deceased donor liver transplantation is performed with a whole liver graft, it often leads to a significant drop in portal pressure and restoration of normal intrahepatic vascular resistance, which helps to resolve collateral circulation issues [6]. However, in adult living donor liver transplantation (LDLT), the smaller graft size may not fully restore intrahepatic resistance, potentially resulting in persistent portal hypertension and inadequate perfusion of the graft. Large spontaneous splenorenal shunts (SSRS) can divert blood away from the liver graft, which can result in portal steal syndrome, leading to graft hypoperfusion after LDLT. While direct ligation of the SSRS is a common method to address this issue, it is technically challenging and carries a potential risk of severe bleeding [7–9]. As a safer alternative, intraoperative ligation of the proximal left renal vein (LRV) near the inferior vena cava (IVC) has been used to manage SSRS. Our previously study indicated that LRV ligation is both safe and effective in preventing the portal steal phenomenon [10], but its real-world long-term results have not been reported yet in literature. This study aimed to evaluate the 20-year outcomes of LDLT with LRV ligation, focused on maintenance of the renal function and the hemodynamic changes of SSRS.
Material and Methods
PATENT SELECTION:
From October 2001 to January 2005, 44 patients underwent concurrent RLV ligation during LDLT out of 665 cases of adult LDLT as a prospective study [10]. These patients had large SSRS >10 mm in diameter at the insertion site into the LRV. All patients of this prior study group were selected in the present retrospective observational study for very-long-term follow-up.
TECHNIQUES FOR LRV LIGATION:
During the recipient surgery, the proximal LRV was isolated adjacent to the IVC using the Kocher maneuver, which was done prior to the recipient hepatectomy. Once the hepatectomy was completed, the amount of portal flow bleeding from the severed end of the main portal vein (PV) was observed both with and without clamping of the LRV. This comparative evaluation was made to assess the extent of shunting blood flow through the SSRS. After confirming that clamping the LRV significantly increased portal blood flow, the LRV was ligated immediately following portal reperfusion. A secure tight tie-ligation using a synthetic umbilical tape was made at the isolated portion of the proximal LRV inserting into the IVC (Figure 1). Two large metal clips were applied over the tie site to identify the site of LRV ligation.
POST-TRANSPLANT FOLLOW-UP:
The institutional protocols for post-transplant follow-up with imaging studies have been described elsewhere [11,12]. LDLT recipients underwent dynamic computed tomography (CT) scanning and Doppler ultrasonography (DUS) before and after LDLT to assess changes in the splanchnic collaterals and portal blood flow. Abdominal DUS was performed every day during the first post-transplant week to assess the flow status of the hepatic artery, portal vein, and outflow hepatic vein. Abdominal CT scanning was performed weekly during the first post-transplant month. Follow-up abdominal CT scanning was performed every 6 months during the first post-transplant year, annually during the first 5 years, and then every 2 years lifelong. For patients with impaired renal function or adverse effects of CT contrast media, DUS or magnetic resonance imaging studies were performed instead. Renal function tests, including urine output, serum creatinine, estimated glomerular filtration rate (eGFR), and urinalysis, were frequently performed during the first 3 post-transplant months. Thereafter, blood chemistry studies, including serum creatinine and eGFR, were performed at every outpatient clinic visit.
Patient follow-up was continued until September 2024 or until patient death, through a comprehensive review of institutional medical records and in collaboration with the National Health Insurance Service of Korea.
The study protocol received approval from the institutional review board at Asan Medical Center (approval no. 2021-1611). Due to the retrospective nature of this research, the requirement for informed consent was waived. The study adhered to the ethics guidelines outlined in the World Medical Association Declaration of Helsinki 2013.
STATISTICAL ANALYSIS:
Numerical data are presented as means and the standard deviation, accompanied by range. To assess survival rates, we employed the Kaplan-Meier method. Statistical analyses were conducted using SPSS version 22 (IBM, New York, NY, USA).
Results
PATIENT DEMOGRAPHICS:
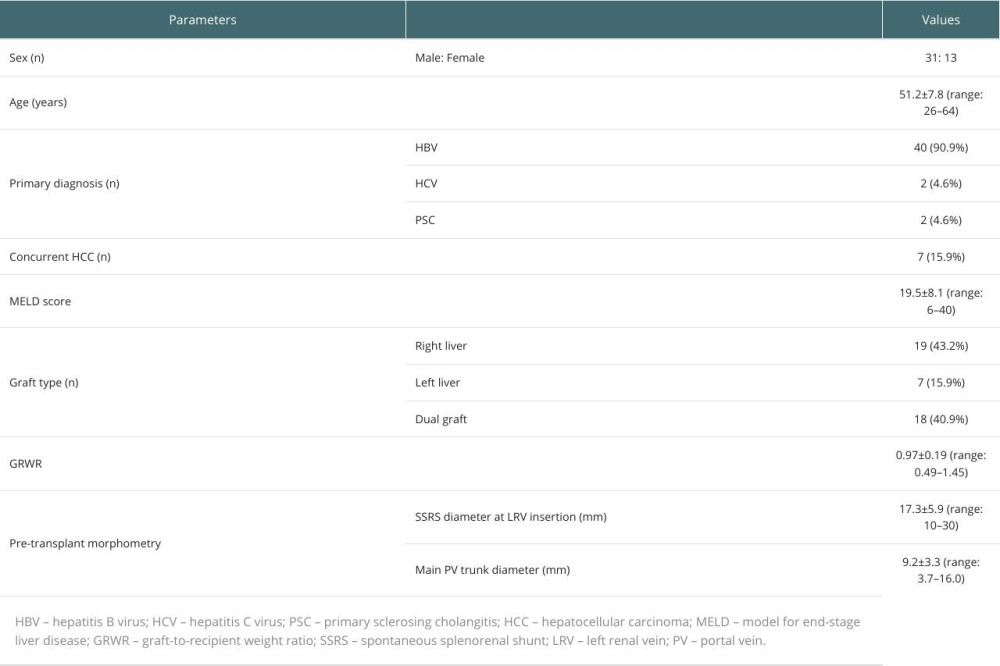
Patient and graft profiles are summarized in Table 1. There were 31 males and 13 females. Primary diagnoses for LDLT were hepatitis B virus-associated liver cirrhosis in 40 patients, hepatitis C virus-associated liver cirrhosis in 2, and primary sclerosing cholangitis in 2. The mean model for end-stage liver disease score was 19.5±8.1 (range: 6–40). The mean graft-to-recipient weight ratio was 0.97±0.19 (range: 0.49–1.45).
PERIOPERATIVE CHANGES IN MORPHOMETRIC FEATURES AND HEMODYNAMICS:
Pre-transplant CT scanning showed that the diameter of SSRS at the insertion into the LRV was 17.3±5.9 mm (range: 10–30 mm), and the diameter of the main PV trunk was 9.2±3.3 mm (range: 3.7–16.0 mm). Pre-transplant DUS showed definite PV stenosis (<10 mm at the main PV trunk) in 24 patients (54.5%), occlusion of PV flow in 7 patients (15.9%), and centrifugal portal flow in 3 patients (6.8%).
Intraoperative DUS showed a significant increase in portal flow between pre- and post-ligation results of the LRV after engraftment. There were no LRV ligation-associated complications.
Post-transplant DUS showed that the flow velocity of the main PV reached the peak values of 85.4±39.5 cm/s (range: 35–238 cm/s) on the post-transplant day 1, and gradually decreased to 53.4±25.0 cm/s (range: 21–120 cm/s) on day 7 according to regeneration of the partial liver graft.
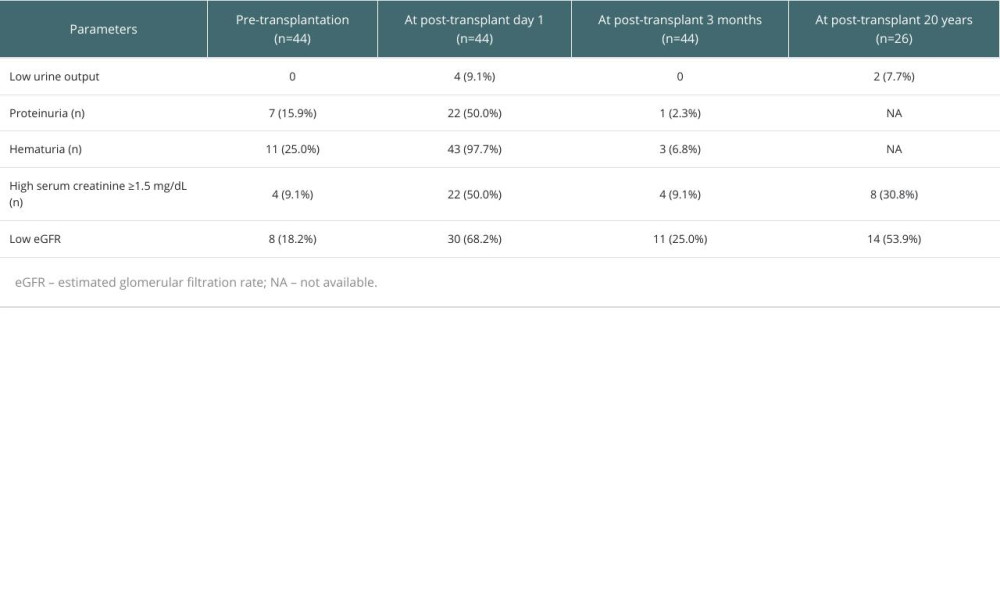
PERI-TRANSPLANT AND LONG-TERM CHANGES IN RENAL FUNCTION:
Peri-transplant and 20-year changes in renal function before and after LDLT with LRV ligation are summarized in Table 2.
Hypo-attenuation of the left kidney, compared with the normally-appearing right kidney, was observed in 34 (77.3%) patients on day-7 CT scans, but such perfusion abnormality disappeared in all patients on 3-month CT scans.
At 3 months after transplant, normalization of the renal function was observed in 40 of 44 patients (90.9%). At the time of writing or patient death, 3 patients (6.8%) showed marked atrophy of both kidneys leading to end-stage renal disease requiring hemodialysis, which was not associated with LRV ligation. Solitary atrophy of the left kidney was not observed at all. At the last or latest follow-up blood tests, 4 of 44 patients (9.1%) received renal replacement therapy due to diabetic nephropathy (n=3) and glomerulonephritis (n=1).
Mild fluctuating increases of the serum creatinine (1.5–2 mg/dL) were observed in 9 of 44 patients (20.5%). The remaining 31 patients (70.5%) showed normal serum creatinine levels.
In the 26 patients who survived more than 20 years at the time of writing, 2 patients (7.7%) received hemodialysis due to chronic renal failure, 6 patients had mildly impaired renal function (serum creatinine 1.5–2 mg/dL with eGFR <60 mL/min), and the remaining 18 patients (69.2%) had normal serum creatinine levels; however, of them, low eGFR <60 mL/min was observed in 6 patients (33.3%). As a cross-sectional analysis at 20 years after transplant, 2 patients (7.7%) had chronic renal failure, 12 patients (46.2%) had mildly impaired renal function regarding either serum creatinine or eGFR, and only 12 patients (46.2%) had normally preserved renal function. Calcineurin inhibitor-free immunosuppressive regimens with or without dosage reduction of entecavir or tenofovir were administered to 8 of these 26 patients (30.8%) to avoid drug-associated nephrotoxicity.
LONG-TERM PATIENT SURVIVAL:
Eighteen patients died due to cancer recurrence (n=6), pneumonia (n=3), and other causes (n=9). The 1-, 5-, 10-, and 20-year overall patient survival rates were 95.5%, 86.4%, 81.8%, and 59.1%, respectively (Figure 2).
LONG-TERM CHANGES IN MORPHOMETRIC FEATURES AND HEMODYNAMICS OF SSRS:
At the time of writing or patient death, SSRS was completely resolved in 20 patients (45.5%) (Figure 3), but another 20 patients (45.5%) had persistently identifiable SSRS of variable extents (Figure 4). In addition, the ligated LRV was reopened and made large SSRS in 4 patients (9.1%) (Figure 5). Of these, radiologic intervention for retrograde intravenous obliteration was performed in 2 each for variceal bleeding control and portal flow augmentation (Figure 6).
Discussion
Sufficient portal inflow is essential for uneventful regeneration of a partial liver graft after LDLT [13,14]. In LDLT for cirrhotic patients with large collateral shunts, detrimental portal flow steal through the collaterals can occur due to increased intrahepatic vascular resistance [13]. Persistence of the spontaneous portosystemic shunts (SPSS) after LDLT with a small-for-size graft is associated with negative outcomes because increased intrahepatic vascular resistance can aggravate portosystemic shunting. Thus, ligation of these collateral veins is recommended [2,4], but direct ligation of these collateral veins is often technically difficult and vulnerable to accidental bleeding [7–9]. As an alternative and easier method to direct ligation, we have performed LRV ligation, and previously demonstrated that it was technically safe and effective [10].
The present study assessed the long-term outcomes of LRV ligation following LDLT, focused on maintenance of the renal function and the hemodynamic changes of SSRS. LRV ligation induced temporal renal complications during the early post-transplant period in a small proportion of patients, probably due to high renal vein pressure, but they spontaneously resolved within a few weeks after transplantation. Isolated atrophy of the left kidney was not observed during long-term follow-up over 20 years. During the follow-up period, post-transplant chronic renal failure requiring renal replacement therapy occurred in 4 of 44 patients, but there was no evidence that LRV ligation was associated with such renal dysfunction in these 4 patients. Their underlying causes of chronic renal failure were diabetic nephropathy in 3 and glomerulonephritis in 1. At post-transplant 20 years in 26 patients who survived more than 20 years, 2 had chronic renal failure, 12 had mildly impaired renal function, and another 12 had normal renal function. Thus, it is reasonable to suggest that LRV ligation is technically safe and hemodynamically effective. All 44 patients in the present study recovered from LDLT despite wide-ranging graft-to-recipient weight ratios and model for end-stage liver disease scores. We confirmed that LRV ligation was not associated with long-term renal dysfunction because left renal blood flow was effectively drained through the left gonadal vein or other collateral veins.
LRV ligation can effectively increase portal blood flow and block SSRS during the early post-transplant period. Over several years following LDLT with LRV ligation, SSRS completely resolved and disappeared in 20 patients, whereas another 20 patients showed persistently identifiable SSRS of variable extents despite the ligated state of the LRV. Meanwhile, 4 patients showed reopened LRV, probably due to mechanical dilatation of the ligation site from loosening of the ties. We currently recommend a more secure ligation technique using linear stapling and overlapped over-and-over running sutures, instead of simple tie-ligation.
Reopening of the LRV resulted in reappearance of large SSRS. Two of them underwent retrograde transvenous obliteration for variceal bleeding control and portal flow augmentation, and both underwent balloon-occluded retrograde transvenous obliteration and plug-assisted retrograde transvenous obliteration [15,16]. Recent technical advances in interventional radiology facilitate control of residual SSRS collaterals that were missed during LDLT or newly developed after LDLT. Proximal splenic vein embolization is reported to effectively interrupt complicated large SSRS in adult LDLT [17].
Currently, our preferred method for control of SSRS is direct ligation of the SSRS at its insertion site to the LRV after full exposure of the SSRS and LRV. We have successfully performed this procedure in hundreds of LDLT cases, but it carries potential risk of accidental bleeding during vascular isolation and it can leave some residual SSRS collateral veins. A Korean single-center study presented a 7.7% rate of major complications related to direct SSRS ligation [9]. Considering these drawbacks of direct SSRS ligation, LRV ligation is still worthy of clinical application in some selected situations with complex SSRS. Several studies confirmed the technical ease and hemodynamic effectiveness of LRV ligation in LDLT and in deceased donor liver transplantation [10,18–24].
A consensus conference guideline for prevention of small-for-size syndrome in LDLT suggests evidence-based management of SPSS [25]. The pathophysiology of development of small-for-size syndrome is related to the increased portal flow and associated shear stress it causes on a small graft. Consequently, it is counterintuitive to ligate the SPSS in small-for-size syndrome because it can worsen the hyperdynamic circulation. However, a large, persistent SPSS can divert the portal flow after transplantation, leading to portal steal syndrome, with consequent graft hypoperfusion and dysfunction. Thus, it is suggested that large SPSS (greater than 10 mm) should be ligated to prevent portal steal. All patients in the present study met the conditions of this suggestion. The decision to ligate SPSS versus leaving them undisturbed needs to be carefully weighed after considering all factors, because ligation probably gives a more controlled situation of portal hemodynamics, and reduces the need for re-exploration or radiological interventions in the post-transplant period for SPSS with portal flow steal [26].
The present study has several limitations. It was retrospective and followed a previous single-center prospective study. Most patients had associated with hepatitis B virus infection. The number of study patients was too small to carry out statistical analysis. Detailed morphometric and hemodynamics analyses on the status of the whole SPSS were not performed.
Conclusions
The results of this study demonstrate that proximal LRV ligation is a safe and effective method to control SSRS. Currently, direct ligation of the SSRS is preferred, but LRV ligation still can be a good therapeutic option when direct access to SSRS is not feasible.
Figures
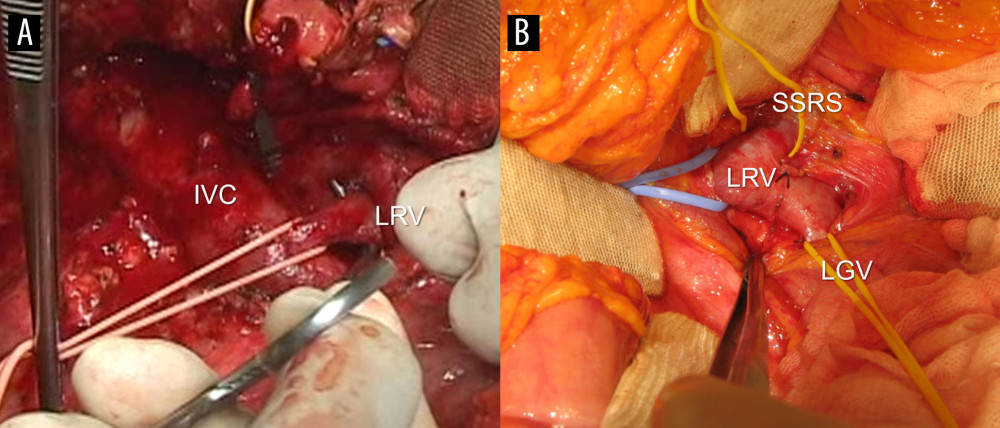 Figure 1. Intraoperative photographs showing vascular isolation. (A) The proximal left renal vein (LRV) was isolated from the inferior vena cava (IVC) after Kocher maneuver. The LRV was ligated at the vessel-loop encircled site with a synthetic umbilical tape. (B) The proximal LRV was isolated with individual encircling of the spontaneous splenorenal shunt (SSRS) and left gonadal vein (LGV).
Figure 1. Intraoperative photographs showing vascular isolation. (A) The proximal left renal vein (LRV) was isolated from the inferior vena cava (IVC) after Kocher maneuver. The LRV was ligated at the vessel-loop encircled site with a synthetic umbilical tape. (B) The proximal LRV was isolated with individual encircling of the spontaneous splenorenal shunt (SSRS) and left gonadal vein (LGV). 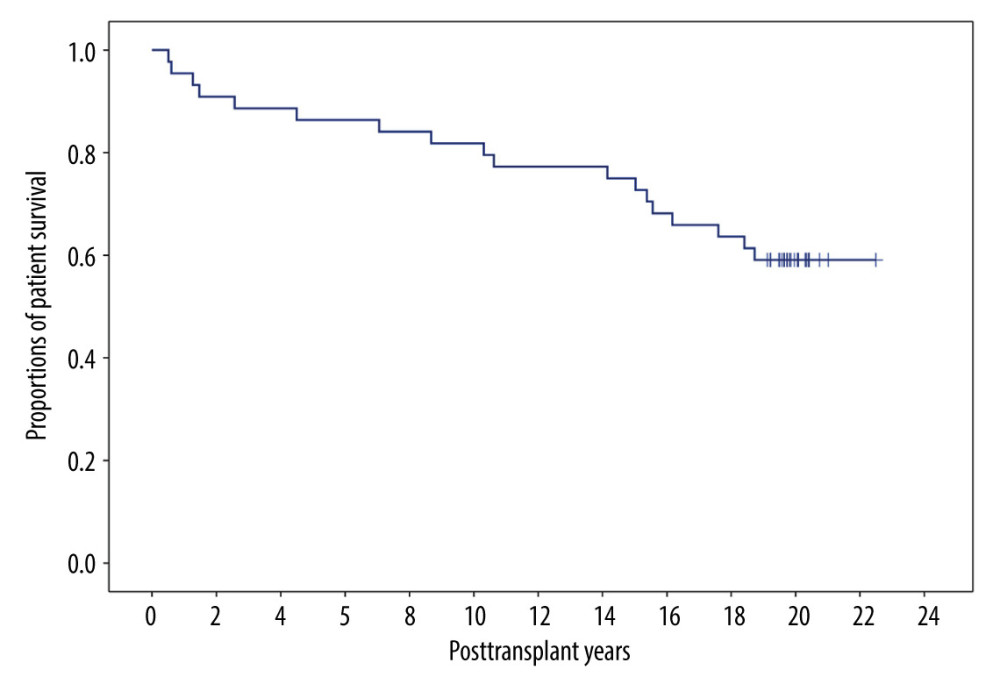 Figure 2. Overall patient survival curve.
Figure 2. Overall patient survival curve. 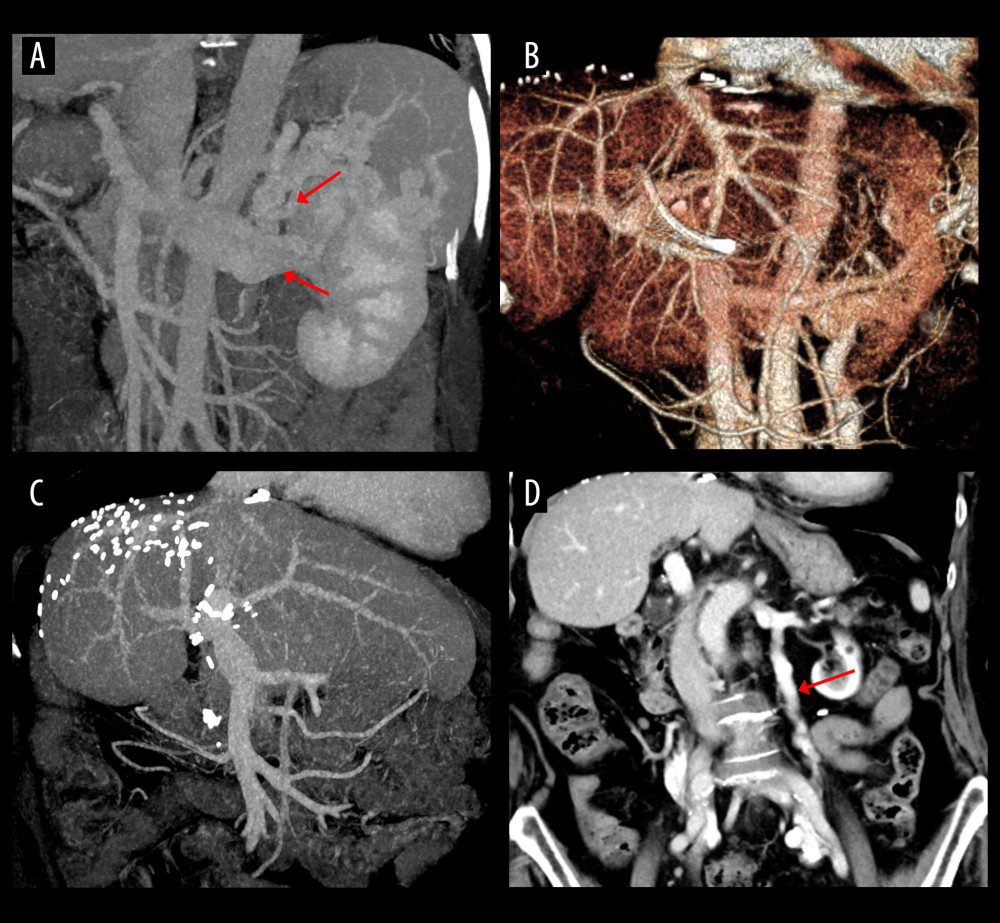 Figure 3. Computed tomography (CT) scans showing resolution of the spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) SSRS disappeared at CT scan taken 3 months after transplantation. (C, D) Follow-up CT scans taken 21 years after transplantation showed absence of SSRS with left renal vein outflow drainage through the enlarged left gonadal vein (arrow).
Figure 3. Computed tomography (CT) scans showing resolution of the spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) SSRS disappeared at CT scan taken 3 months after transplantation. (C, D) Follow-up CT scans taken 21 years after transplantation showed absence of SSRS with left renal vein outflow drainage through the enlarged left gonadal vein (arrow). 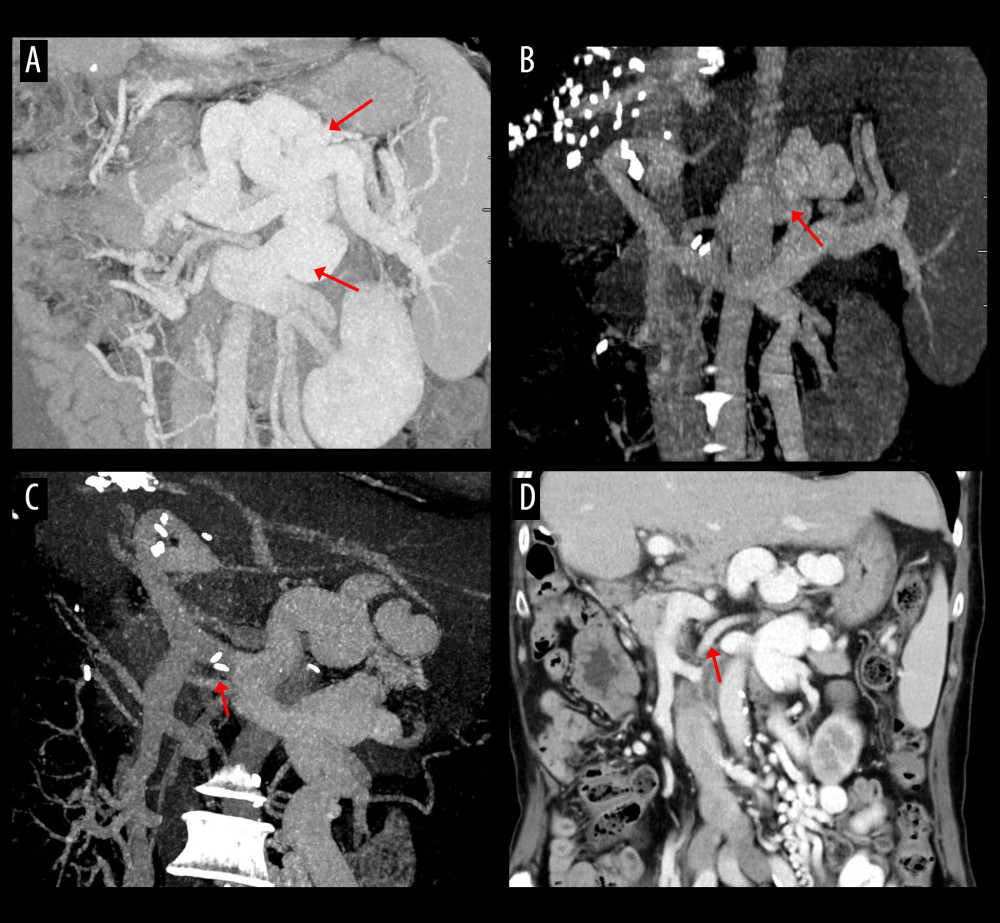 Figure 4. Computed tomography (CT) scans showing persistence of spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Some SSRS branches (arrow) were persistently identified at CT scan taken 6 months after transplantation. (C, D) Follow-up CT scans taken 20 years after transplantation showed persistence of SSRS with the ligated stump of the proximal left renal vein (arrows).
Figure 4. Computed tomography (CT) scans showing persistence of spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Some SSRS branches (arrow) were persistently identified at CT scan taken 6 months after transplantation. (C, D) Follow-up CT scans taken 20 years after transplantation showed persistence of SSRS with the ligated stump of the proximal left renal vein (arrows). 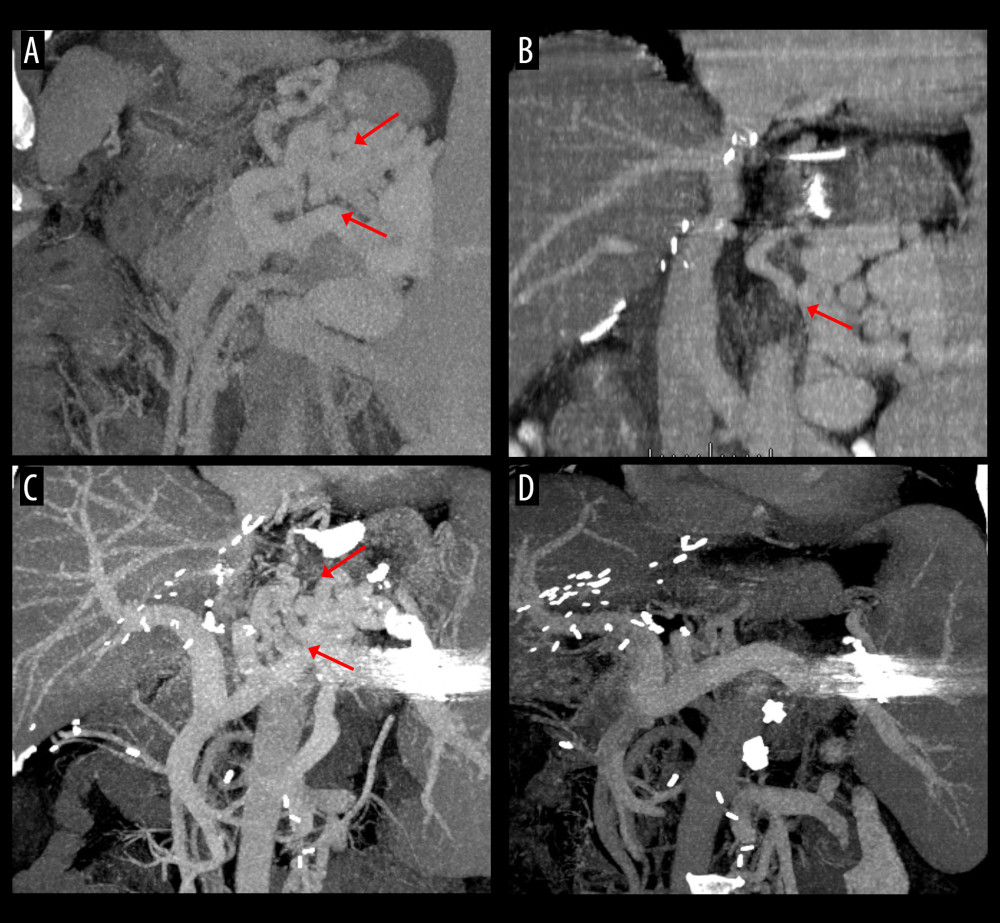 Figure 5. Computed tomography (CT) scans showing interventional occlusion of the spontaneous splenorenal shunt (SSRS) through retrograde transvenous obliteration. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Small SSRS branches (arrow) were identified at CT scan taken 1 year after transplantation. (C) Large SSRS branches (arrows) were identified at CT scan taken 7 years after transplantation with reopening of the left renal vein. (D) Follow-up CT scan taken 9 years after transplantation (1 year after retrograde transvenous obliteration) showed disappearance of SSRS.
Figure 5. Computed tomography (CT) scans showing interventional occlusion of the spontaneous splenorenal shunt (SSRS) through retrograde transvenous obliteration. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Small SSRS branches (arrow) were identified at CT scan taken 1 year after transplantation. (C) Large SSRS branches (arrows) were identified at CT scan taken 7 years after transplantation with reopening of the left renal vein. (D) Follow-up CT scan taken 9 years after transplantation (1 year after retrograde transvenous obliteration) showed disappearance of SSRS. 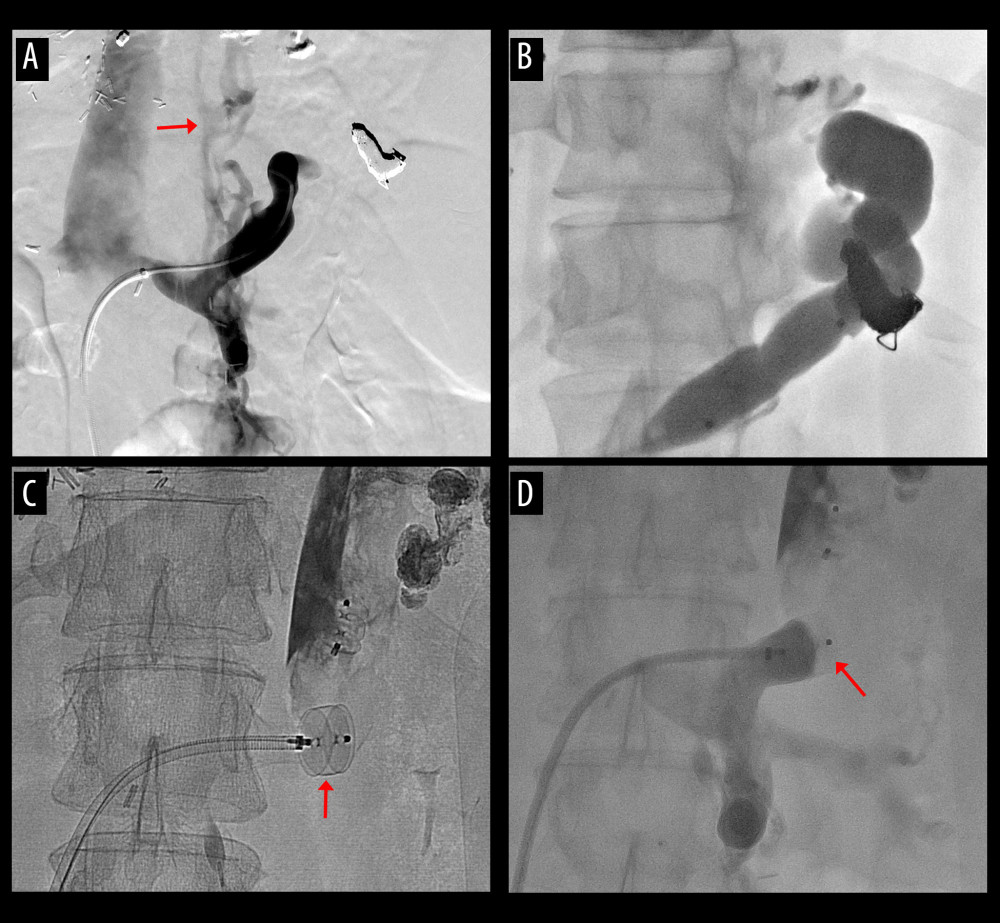 Figure 6. Procedures of plug-assisted retrograde transvenous obliteration (PARTO). (A) Retrograde venography shows development of the spontaneous splenorenal shunt (SSRS) with esophageal varices (arrow). (B, C) Distal branches are embolized and the large SSRS was occluded with a vascular plug (arrow). (D) The SSRS was completely occluded after PARTO (arrow).
Figure 6. Procedures of plug-assisted retrograde transvenous obliteration (PARTO). (A) Retrograde venography shows development of the spontaneous splenorenal shunt (SSRS) with esophageal varices (arrow). (B, C) Distal branches are embolized and the large SSRS was occluded with a vascular plug (arrow). (D) The SSRS was completely occluded after PARTO (arrow). References
1. De Carlis L, Del Favero E, Rondinara G, The role of spontaneous portosystemic shunts in the course of orthotopic liver transplantation: Transpl Int, 1992; 59-14
2. Kita Y, Harihara Y, Sano K, Reversible hepatofugal portal flow after liver transplantation using a small-for-size graft from a living donor: Transpl Int, 2001; 14; 217-22
3. Sekido H, Matsuo K, Takeda K, Severe fatty change of the graft liver caused by a portosystemic shunt of mesenteric varices: Transpl Int, 2002; 15; 259-62
4. Fujimoto M, Moriyasu F, Nada T, Influence of spontaneous portosystemic collateral pathways on portal hemodynamics in living-related liver transplantation in children. Doppler ultrasonographic study: Transplantation, 1995; 60; 41-45
5. Margarit C, Lazaro JL, Charco R, Liver transplantation in patients with splenorenal shunts: Intraoperative flow measurements to indicate shunt occlusion: Liver Transpl Surg, 1999; 5; 35-39
6. Paulsen AW, Klintmalm GB, Direct measurement of hepatic blood flow in native and transplanted organs, with accompanying systemic hemodynamics: Hepatology, 1992; 16; 100-1
7. Settmacher U, Nussler NC, Glanemann M, Venous complications after orthotopic liver transplantation: Clin Transplant, 2000; 14; 235-41
8. Troisi R, Hesse UJ, Decruyenaere J, Functional, life-threatening disorders and splenectomy following liver transplantation: Clin Transplant, 1999; 13; 380-88
9. Kim H, Yoon KC, Lee KW, Tips and pitfalls in direct ligation of large spontaneous splenorenal shunt during liver transplantation: Liver Transpl, 2017; 23; 899-906
10. Lee SG, Moon DB, Ahn CS, Ligation of left renal vein for large spontaneous splenorenal shunt to prevent portal flow steal in adult living donor liver transplantation: Transpl Int, 2007; 20; 45-50
11. Kim M, Hwang S, Ahn CS, Twenty-year longitudinal follow-up after liver transplantation: A single-center experience with 251 consecutive patients: Korean J Transplant, 2022; 36; 45-53
12. Oh RK, Hwang S, Song GW, Donor sex and donor-recipient sex disparity do not affect hepatocellular carcinoma recurrence after living donor liver transplantation: Ann Surg Treat Res, 2023; 105; 133-40
13. Kawasaki S, Makuuchi M, Ishizone S, Liver regeneration in recipients and donors after transplantation: Lancet, 1992; 339; 580-81
14. Yagi S, Iida T, Taniguchi K, Impact of portal venous pressure on regeneration and graft damage after living-donor liver transplantation: Liver Transpl, 2005; 11; 68-75
15. Saad WE, Chick JFB, Srinivasa RN, Two-year outcomes of balloon-occluded retrograde transvenous obliteration of gastric varices in liver transplant recipients: A multi-institutional study: Diagn Interv Imaging, 2017; 98; 801-8
16. Kim GH, Gwon DI, Ko GY, Short-term results of plug-assisted retrograde transvenous obliteration for portal steal from complicated portosystemic shunts in living-donor liver transplantation: J Vasc Interv Radiol, 2023; 34; 645-52
17. Kang WH, Moon DB, Ko GY, Application of proximal splenic vein embolization to interrupt complicated large splenorenal shunts in adult living donor liver transplantation: Ann Surg, 2022; 276; e834-e41
18. Tang R, Han D, Li M, Left renal vein ligation for large splenorenal shunt during liver transplantation: ANZ J Surg, 2017; 87; 767-72
19. Nguyen MC, Sage Silski L, Alebrahim M, Left renal vein ligation for spontaneous splenorenal shunts during deceased-donor orthotopic liver transplant is safe and can mitigate complications from portal steal: A case series: Exp Clin Transplant, 2021; 19; 374-77
20. Shankar S, Rammohan A, Balasubramanian B, Renal implications of left renal vein ligation for portal flow augmentation in liver transplantation: World J Surg, 2021; 45; 2567-71
21. Golse N, Bucur PO, Faitot F, Spontaneous splenorenal shunt in liver transplantation: results of left renal vein ligation versus renoportal anastomosis: Transplantation, 2015; 99; 2576-85
22. Golse N, Mohkam K, Rode A, Surgical management of large spontaneous portosystemic splenorenal shunts during liver transplantation: Splenectomy or left renal vein ligation?: Transplant Proc, 2015; 47; 1866-76
23. Kousoulas L, Ringe KI, Winkler M, Ligation of left renal vein for spontaneous splenorenal shunt to prevent portal hypoperfusion after orthotopic liver transplantation: Case Rep Transplant, 2013; 2013; 842538
24. Liu Y, Liu B, Cao J, Wang Y, Ligation of left renal vein to improve portal vein blood flow during liver transplantation for alcoholic cirrhosis: A case report: Asian J Surg, 2024; 47; 4980-81
25. Hakeem AR, Mathew JS, Aunés CV, Preventing small-for-size syndrome in living donor liver transplantation: Guidelines from the ILTS-iLDLT-LTSI Consensus Conference: Transplantation, 2023; 107; 2203-15
26. Reddy MS, Rela M, Portosystemic collaterals in living donor liver transplantation: What is all the fuss about?: Liver Transpl, 2017; 23; 537-44
Figures
 Figure 1. Intraoperative photographs showing vascular isolation. (A) The proximal left renal vein (LRV) was isolated from the inferior vena cava (IVC) after Kocher maneuver. The LRV was ligated at the vessel-loop encircled site with a synthetic umbilical tape. (B) The proximal LRV was isolated with individual encircling of the spontaneous splenorenal shunt (SSRS) and left gonadal vein (LGV).
Figure 1. Intraoperative photographs showing vascular isolation. (A) The proximal left renal vein (LRV) was isolated from the inferior vena cava (IVC) after Kocher maneuver. The LRV was ligated at the vessel-loop encircled site with a synthetic umbilical tape. (B) The proximal LRV was isolated with individual encircling of the spontaneous splenorenal shunt (SSRS) and left gonadal vein (LGV). Figure 2. Overall patient survival curve.
Figure 2. Overall patient survival curve. Figure 3. Computed tomography (CT) scans showing resolution of the spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) SSRS disappeared at CT scan taken 3 months after transplantation. (C, D) Follow-up CT scans taken 21 years after transplantation showed absence of SSRS with left renal vein outflow drainage through the enlarged left gonadal vein (arrow).
Figure 3. Computed tomography (CT) scans showing resolution of the spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) SSRS disappeared at CT scan taken 3 months after transplantation. (C, D) Follow-up CT scans taken 21 years after transplantation showed absence of SSRS with left renal vein outflow drainage through the enlarged left gonadal vein (arrow). Figure 4. Computed tomography (CT) scans showing persistence of spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Some SSRS branches (arrow) were persistently identified at CT scan taken 6 months after transplantation. (C, D) Follow-up CT scans taken 20 years after transplantation showed persistence of SSRS with the ligated stump of the proximal left renal vein (arrows).
Figure 4. Computed tomography (CT) scans showing persistence of spontaneous splenorenal shunt (SSRS) after left renal vein ligation. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Some SSRS branches (arrow) were persistently identified at CT scan taken 6 months after transplantation. (C, D) Follow-up CT scans taken 20 years after transplantation showed persistence of SSRS with the ligated stump of the proximal left renal vein (arrows). Figure 5. Computed tomography (CT) scans showing interventional occlusion of the spontaneous splenorenal shunt (SSRS) through retrograde transvenous obliteration. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Small SSRS branches (arrow) were identified at CT scan taken 1 year after transplantation. (C) Large SSRS branches (arrows) were identified at CT scan taken 7 years after transplantation with reopening of the left renal vein. (D) Follow-up CT scan taken 9 years after transplantation (1 year after retrograde transvenous obliteration) showed disappearance of SSRS.
Figure 5. Computed tomography (CT) scans showing interventional occlusion of the spontaneous splenorenal shunt (SSRS) through retrograde transvenous obliteration. (A) Large SSRS branches (arrows) were identified at the pre-transplant CT. (B) Small SSRS branches (arrow) were identified at CT scan taken 1 year after transplantation. (C) Large SSRS branches (arrows) were identified at CT scan taken 7 years after transplantation with reopening of the left renal vein. (D) Follow-up CT scan taken 9 years after transplantation (1 year after retrograde transvenous obliteration) showed disappearance of SSRS. Figure 6. Procedures of plug-assisted retrograde transvenous obliteration (PARTO). (A) Retrograde venography shows development of the spontaneous splenorenal shunt (SSRS) with esophageal varices (arrow). (B, C) Distal branches are embolized and the large SSRS was occluded with a vascular plug (arrow). (D) The SSRS was completely occluded after PARTO (arrow).
Figure 6. Procedures of plug-assisted retrograde transvenous obliteration (PARTO). (A) Retrograde venography shows development of the spontaneous splenorenal shunt (SSRS) with esophageal varices (arrow). (B, C) Distal branches are embolized and the large SSRS was occluded with a vascular plug (arrow). (D) The SSRS was completely occluded after PARTO (arrow). In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588