10 November 2020: Original Paper
Renal Function in Patients with or without a Left Ventricular Assist Device Implant During Listing for a Heart Transplant
Armin Zittermann1ABCDEF*, Rene Schramm1DE, Tobias Becker1BC, Ellen von Rössing1BC, Detlef Hinse2BC, Stefan Wlost1BC, Michiel Morshuis1BD, Jan F. Gummert1BD, Uwe Fuchs1ABDDOI: 10.12659/AOT.925653
Ann Transplant 2020; 25:e925653
Abstract
BACKGROUND: Left ventricular assist device (LVAD) implantation may improve kidney function, but in patients awaiting heart transplantation, the long-term effects of LVAD implantation on renal function and subsequent clinical outcome are unclear.
MATERIAL AND METHODS: We analyzed data in patients with LVAD implants (n=139) and without LVAD implants (n=1038) who were listed for a heart transplant at our institution between 2000 and 2019. The primary endpoint was an impairment in renal function (decrease of creatinine-based estimated glomerular filtration rate [eGFR] by ≥30%) up to a maximum of 2 years after listing. Secondary endpoints were chronic kidney disease stage 4 or 5, heart transplantation, survival during listing, and 1-year survival after transplantation.
RESULTS: Values for eGFR increased after LVAD implantation (P=0.001) and were higher at the time of waitlisting in the LVAD group than in the non-LVAD group (P=0.002), but were similar between groups at the end of waitlisting (P=0.75). Two-year freedom from renal impairment was 50.6% and 66.7% in the LVAD and non-LVAD groups, respectively, with a multivariable-adjusted hazard ratio for the LVAD versus the non-LVAD group of 1.78 (95% confidence interval 1.19–2.68; P=0.005). Two-year freedom from chronic kidney disease stages 4–5 was similar between study groups (LVAD group: 83.5%; non-LVAD group: 80.1%; =0.50). The 2-year probability of transplantation was slightly lower in the LVAD group than in the non-LVAD group (50.0% and 55.8%, respectively, P=0.017). However, 2-year survival on the waiting list and 1-year survival after transplantation did not differ significantly between study groups (P-values >0.20).
CONCLUSIONS: Our data indicate a transient improvement in creatinine-based eGFR values by LVAD implantation without influencing survival.
Keywords: Glomerular Filtration Rate, Heart Transplantation, Renal Insufficiency, Chronic, Ventricular Function, Left, Heart-Assist Devices, Kidney, Stroke Volume
Background
Heart failure (HF) is a medical problem affecting almost 23 million people globally [1]. Five percent of patients develop end-stage HF that is refractory to maximal medical treatment [2]. For these patients, heart transplantation is the last treatment option, with median survival exceeding 10 years [3]. Due to donor heart shortage, left ventricular assist devices (LVADs) are increasingly used as a bridge to transplant [4]. LVADs have proven to be effective in improving survival and quality of life in patients with refractory HF [5], and 30% of all LVADs are implanted as a bridge to transplant, with an additional 23% as a bridge to candidacy in anticipation of listing [6].
HF is often associated with renal impairment, resulting in a pathophysiologic disorder that is called cardiorenal syndrome [7]. Approximately 50% of patients with chronic HF have an estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2 [8]. Several observational studies reported poor clinical outcomes in patients with HF in cases with impaired kidney function [9,10]. Moreover, renal replacement therapy was an important independent predictor of mortality in patients with end-stage HF who needed extracorporeal cardiopulmonary resuscitation [11]. With regard to heart transplantation, chronic kidney disease (CKD) stages 4–5 are considered to be relative contraindications [12].
LVAD implantation is associated with a transient decrease in serum creatinine concentrations and an increase in eGFR values [13,14]. Since the long-term effects of LVAD implantation in patients with refractory HF on kidney function are currently not clear [15], we aimed to investigate renal function, the probability of transplantation, and overall mortality in patients with or without an LVAD awaiting heart transplantation.
Material and Methods
PATIENTS:
This data analysis summarizes results obtained at our institution between January 2000 and April 2019. Patients with or without an LVAD implant who were listed for a heart transplant were eligible for inclusion. Patients who received an LVAD implant as destination therapy were not included in our data analysis. Listing criteria included the following parameters [16,17]: New York Heart Association (NYHA) functional class III or IV, maximal oxygen uptake <10–14 mL/kg/min, central venous pressure >15 mmHg, left ventricular ejection fraction <20%, left ventricular end-diastolic diameter >20 mmHg, a cardiac index <2.2 L/min/m2, plasma sodium <135 mmol/L, or LVAD implantation. Hemodynamic criteria for LVAD implantation were (i) systolic blood pressure <80 mmHg, (ii) pulmonary capillary wedge pressure >20 mmHg, and a cardiac index <2 L/min/m2 despite maximal medical treatment [18,19]. Only those patients with LVAD implants whose device implantation was within the last 6 months of listing were considered. Patients with a biventricular assist device or total artificial heart implant at time of waitlisting and patients with missing data regarding renal function were excluded from data analysis, as were patients with CKD stages 4 and 5, respectively. The patient selection process is described in Figure 1.
The study was performed in accordance with the Strengthening the Reporting of Observational studies in Epidemiology statement for cohort studies (
STUDY DESIGN:
The study cohort was divided into 2 groups: patients with an LVAD implant (designated the LVAD group) and those without an LVAD implant (designated the non-LVAD group). In this way, 139 patients were allocated to the LVAD group and 1038 to the non-LVAD group. For our analysis, we assessed 20 baseline characteristics that are routinely and prospectively collected and entered into a database at our institution. In addition, we assessed the last eGFR values before listing, the first eGFR values after listing, eGFR values at the time of renal impairment (see below), and the last eGFR values during listing. In the LVAD group, we also assessed the last eGFR values before device implant. GFR values were calculated using the simplified Modification of Diet in Renal Disease (MDRD) formula [20].
ENDPOINTS:
The primary endpoint was an impairment in renal function up to a maximum of 2 years after listing. Renal impairment was considered if eGFR values decreased by 30% or more. Patients were censored at the time they received a transplant or died. Secondary endpoints were CKD stage 4 or 5, heart transplantation, and 2-year survival during listing. Moreover, 1-year survival after transplantation was assessed in those patients who receive a transplant within 2 years of listing.
STATISTICS:
We report categorical variables as numbers and percentages of observations, and continuous data as means with standard deviation (normally distributed data) or medians with 25th to 75th percentiles, where appropriate. Normal distribution of data was tested using the Kolmogorov-Smirnov test and was assumed if P-values were >0.05. We used the unpaired t test, the Mann-Whitney test, and Fisher’s exact test to assess group differences in continuous and categorical baseline variables, where appropriate. Comparisons within study groups were performed using the paired t test or Wilcoxon’s rank sum test, where appropriate. Between-group comparisons of creatinine and eGFR values were based on analysis of covariance (ANCOVA) with adjustments for those baseline characteristics that differed significantly between study groups (sex, heart rate, blood pressure, cardiac index, left ventricular ejection fraction, left ventricular end-diastolic diameter, pulmonary capillary wedge pressure, diagnosis, NYHA functional class, sodium, bilirubin; see Table 1). Nonnormally distributed data were log-transformed before analysis.
We generated Kaplan-Meier estimates by study group for the primary and secondary endpoints as a function of time. The log-rank test was used to test for statistical differences between study groups. We also performed unadjusted and multivariable-adjusted Cox regression analysis on the primary and secondary endpoints. Results are presented as hazard ratios (HRs) and 95% confidence intervals (CIs). Adjustments were made for those baseline characteristics that differed significantly between study groups. The proportionality of hazard assumption was assessed by evaluation of time-dependent variables, which were the cross-products of the predictor variables with event-free outcomes. The proportionality of hazard assumption was satisfied for all endpoints (
Results
BASELINE CHARACTERISTICS:
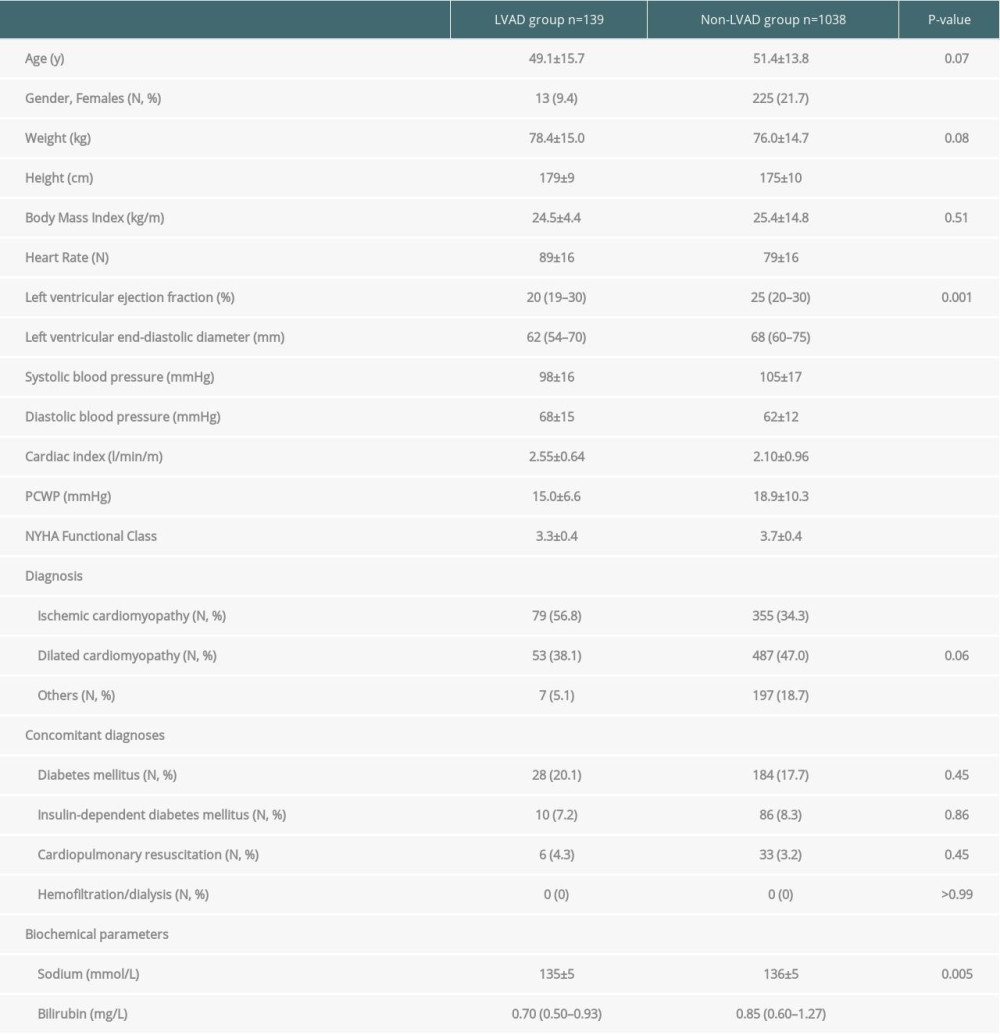
The characteristics of the study cohort at time of waitlisting are presented in Table 1. Briefly, the percentage of women was significantly lower in the LVAD group than in the non-LVAD group. Moreover, cardiac-related parameters differed between groups with significantly higher values for heart rate, diastolic blood pressure, and cardiac index, and significantly lower values for left ventricular ejection fraction, left ventricular end-diastolic diameter, systolic blood pressure, pulmonary capillary wedge pressure, and NYHA functional class in the LVAD group than in the non-LVAD group. In the LVAD group, heart failure was more often due to ischemic cardiomyopathy than in the non-LVAD group. However, concomitant diagnoses such as diabetes mellitus and resuscitation were similar between study groups, and none of the patients needed hemofiltration or dialysis in either the LVAD group or the non-LVAD group.
In the LVAD group, the majority of patients received HeartWare implants (HeartWare International Inc. Framingham, MA, USA; n=56) and HeartMate II implants (Thoratec Corp., Pleasanton, CA, USA; n=26). The remaining LVAD patients received DuraHeart (Terumo Heart Inc., Ann Arbor, MI, USA; n=19), Thoratec (Thoratec Corp.; n=13), NovaCor (WorldHeart Inc., Oakland, CA, USA; n=10), VentrAssist (Ventracor Ltd., Chatswood, NSW, Australia; n=5), and HeartMate I and 3 (Thoratec Corp.; n=4 and 6, respectively) implants.
CREATININE AND EGFR VALUES:
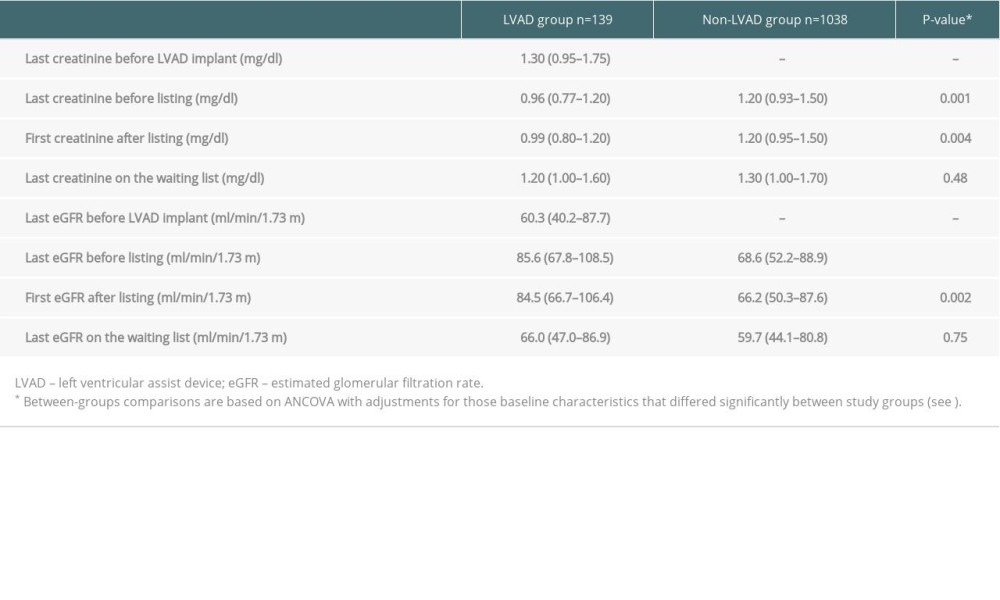
Table 2 presents data on serum creatinine and eGFR values by study group. Briefly, the last creatinine concentrations before listing were significantly lower and the last eGFR values before listing were significantly higher in the LVAD group than in the non-LVAD group, as were the first creatinine and eGFR values after listing. At the time of waitlisting, 17% of patients in the LVAD group and 39% of patients in the non-LVAD group had eGFR values below 60 mL/min/1.73 m2. In both the LVAD and non-LVAD groups, there was a significant rise in serum creatinine and a significant decline in eGFR between the first and the last measurement on the waiting list (all P-values <0.001). Despite the substantial differences in eGFR values at the time of waitlisting, the last creatinine and eGFR values on the waiting list did not differ significantly between study groups.
With respect to the LVAD group, creatinine concentrations were lower at the time of waitlisting, whereas eGFR values were higher than before device implantation (both
PRIMARY AND SECONDARY ENDPOINTS:
LVAD implantation was on average performed 32 days (25th–75th percentiles: 22–56 days) before listing. In the LVAD and non-LVAD groups, mean follow-up was 15.2 months (SD: 9.4 months) and 11.8 months (SD: 10.0 months), respectively (
The primary endpoint was reached by 36.7% (n=51) of patients in the LVAD group and by 20.7% (n=215) of patients in the non-LVAD group. Two-year freedom from renal impairment in the LVAD and non-LVAD groups was 50.6% and 66.7%, respectively (Figure 2), with an unadjusted and multivariable-adjusted HR for the LVAD versus the non-LVAD group of 1.51 (95% CI, 1.11–2.05; P=0.008) and 1.78 (95% CI: 1.19–2.68; P=0.005), respectively. Two-year freedom from CKD stages 4–5 was, however, similar between study groups (LVAD group: 83.5%; non-LVAD group: 80.1%), with an unadjusted and multivariable-adjusted HR for the LVAD versus the non-LVAD group of 0.83 (95% CI: 0.47–1.44; P=0.50) and 0.96 (95% CI: 0.50–1.88; P=0.92), respectively.
The 2-year probability of transplantation was significantly lower in the LVAD group compared with the non-LVAD group (50.0% and 55.8%, respectively) (Figure 3), with an unadjusted and multivariable-adjusted HR for nontransplantation in the LVAD versus the non-LVAD group of 1.37 (95% CI: 1.06–1.77; P=0.018) and 2.63 (95% CI: 1.86–3.72; P<0.001), respectively. Nevertheless, 2-year survival on the waiting list did not differ significantly between study groups (LVAD group: 85.9%; non-LVAD group: 85.7%) (Figure 4). The corresponding unadjusted and multivariable-adjusted HR for 2-year mortality on the waiting list was 0.95 (95% CI: 0.53–1.72) for the LVAD group versus 0.62 (95% CI: 0.25–1.53; P=0.30) for the non-LVAD group. Of the 13 patients in the LVAD group who died during follow-up, 5 died of intracranial bleeding (38%), 5 of sepsis (38%), 1 of sudden cardiac death (8%), and 1 of myocardial infarction (8%), and 1 cause of death was unknown (8%). Of the 79 patients who died in the non-LVAD group, 4 died of intracranial bleeding (5%), 13 of sepsis (16%), 18 of cardiogenic shock (23%), 17 of sudden cardiac death (22%), 12 of multiorgan failure (15%), 2 of gastrointestinal bleeding (2%), and 7 of other causes (9%), and 6 causes of death were unknown (8%). In those patients who received transplants within 2 years of listing (LVAD group: n=64; non-LVAD group: n=552), 1-year mortality after transplantation did not differ significantly between the LVAD and non-LVAD groups (20.4% and 15.5%, respectively; P=0.23; Figure 5).
SENSITIVITY ANALYSIS:
Two patients in the LVAD group were weaned and 68 patients in the non-LVAD needed an LVAD implant during follow-up. Exclusion of the weaned patients did not substantially influence results of the primary endpoint. Briefly, 2-year freedom from renal impairment in the LVAD and non-LVAD groups was 50.6% and 66.7%, respectively, with an unadjusted and multivariable-adjusted HR for the LVAD versus the non-LVAD group of 1.55 (95% CI, 1.14–2.11;
Inclusion of the 68 patients, who initially did not receive an LVAD implant, in the non-LVAD group also did not substantially influence results of the primary endpoint. Briefly, 2-year freedom from renal impairment in the LVAD and non-LVAD groups was 50.6% and 65.7%, respectively, with an unadjusted and multivariable-adjusted HR for the LVAD versus the non-LVAD group of 1.50 (95% CI, 1.11–2.03;
Discussion
This study has 3 major findings. First, at the time of waitlisting, the eGFR values, calculated by creatinine-based formula, were significantly higher in the LVAD group than in the non-LVAD group. Second, freedom from renal impairment and the probability of transplantation were lower in the LVAD than in the non-LVAD group. Third, despite higher eGFR values at the time of waitlisting in the LVAD group compared with the non-LVAD group, survival during listing and survival after transplantation were similar between the 2 study groups.
Before LVAD implantation, eGFR values were significantly lower than after LVAD implantation and at time of waitlisting. Therefore, the higher eGFR values in the LVAD group compared with the non-LVAD group at the time of waitlisting can reliably be explained by an LVAD-induced increase in eGFR values. Similar results have been reported by others [21–23]. Moreover, earlier studies also provided evidence that the increase in eGFR after LVAD implantation may be transient, but long-term data were limited. Our results clearly support the assumption of a transient increase in eGFR values from LVAD implantation. Whether or not this transient increase indicates a true effect on kidney function is unclear. On the one hand, some evidence suggests that due to muscle wasting following LVAD implant, creatinine-based formulae may incorrectly estimate early post-LVAD GFR values [23–25]. Cystatin C-based assessment of kidney function could not confirm a transient increase in eGFR [25]. Moreover, it has been suggested that in frail and sarcopenic patients, such as patients with LVAD implants, creatinine-based formulae for estimating GFR values may overestimate kidney function [23]. Notably, substantial muscle catabolism can occur within a few days of immobilization [26], resulting in lower serum creatinine levels and thus higher eGFR values in the medium term. On the other hand, our LVAD patients served as their own controls. In addition, the results of several creatinine-based formulae have recently been compared against a reference method of measuring glomerular filtration rate (51Cr-EDTA method) in patients with heart failure [27]. Data demonstrated correlation coefficients between 0.81 and 0.90 for all creatinine-based formulae, including the simplified MDRD formula. Therefore, the transient increase in eGFR values in our patients with an LVAD implant may indeed reflect a true transient improvement in kidney function.
The differences in calculated kidney function between study groups have not obviously influenced clinical outcome such as survival on the waiting list and survival after heart transplantation. Notably, the decline in eGFR values during listing was more pronounced in the LVAD group than in the non-LVAD group, and the last eGFR values on the waiting list did not differ significantly between study groups. There is some evidence that in patients with implantation of continuous flow devices, beneficial renal effects may at least in part be counteracted by LVAD-related deleterious effects on renal function [28]. Moreover, we cannot definitely rule out that infectious or clotting complications adversely affected results in the LVAD group. Unfortunately, however, complications were not systematically assessed. The more pronounced decline in eGFR values in the LVAD group compared with the non-LVAD group was also confirmed by the lower 2-year freedom from renal impairment. However, 2-year freedom from severe kidney disease such as CKD stages 4–5 was similar in both study groups. Moreover, during listing more than 75% of the patients in our 2 study groups had eGFR values above 45 mL/min/1.73 m2. The similar clinical outcomes in both study groups are in line with the similar incidence of a severe decline in kidney function during listing. There is evidence that a substantial association of poor kidney function with mortality only occurs in those patients who have eGFR values below 45 mL/min/1.73 m2, but not in patients with eGFR values above 45 mL/min/1.73 m2 [29]. However, mortality is exceptionally high in patients with pre-existing end-stage renal disease, despite LVAD implantation [30,31]. Regarding the association between pretransplant eGFR values and posttransplant mortality, eGFR values <30 have been particularly associated with higher mortality after heart transplantation [32]. In contrast, pretransplantation kidney function was not predictive of mortality after heart transplantation in a study with a low percentage of patients with measured GFR values <30 mL/min/1.73 m2 [33]. Moreover, LVAD implantation obviously does not affect kidney function after heart transplantation [34].
The REMATCH (Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure) study [35], a randomized controlled clinical trial, has demonstrated that, compared with optimal medical treatment, LVAD implantation results in a transient survival benefit. In our investigation, however, study groups were not randomized, and important predictors of survival in patients with heart failure, such as cardiac index and plasma sodium, were significantly lower in the LVAD group than in the non-LVAD group. This difference may at least in part explain why LVAD patients had similar, but not better survival than non-LVAD patients. Notably, the multivariable-adjusted HR for 2-year mortality on the waiting list was lower than the unadjusted HR for the LVAD versus non-LVAD group (0.62
The significant difference in the probability of transplantation between patients with and without an LVAD implant has already been reported by our group [36]. This difference may be due to the allocation scheme in the Eurotransplant region, because inotrope dependence in non-LVAD patients justifies high-urgency status and those patients are prioritized over LVAD patients without life-threatening device-related complications [37]. It remains elusive at present whether the planned introduction of a Cardiac Allocation Score may alter the allocation of donor hearts to LVAD and non-LVAD patients, as it should balance both urgency and transplant benefit.
Our study has both strengths and limitations. Strengths are the relatively large number of patients, the homogeneous groups of patients, and the clear listing criteria. Limitations are as follows. First, the retrospective study design resulted in missing eGFR data just before listing, immediately after listing, or at the end of listing in a substantial number of patients. Second, several patients with an LVAD implant had to be excluded from data analysis because the device was implanted long before or long after listing. Since these patients may also differ in their clinical characteristics from those patients who were included in the data analysis, this exclusion may have biased study results to some extent. Third, GFR values were only estimated by the simplified MDRD formula, and creatinine concentrations may have been influenced by factors that are unrelated to kidney function such as changes in muscle mass. Fourth, there was a predominance of male patients in our study groups, but this is a general phenomenon in patients awaiting heart transplantation [38] and thus unrelated to our investigation. Fifth, censoring of patients that received transplants and deceased patients may have biased outcome results. Therefore, any overstating of our results should be avoided. Finally, despite clear listing criteria for a heart transplant for both study cohorts, criteria are not completely identical. This became obvious by significant differences in various baseline characteristics between study groups and may also have biased study results. However, adjustment for baseline covariates is a generally accepted measure to address this problem. Notably, unadjusted and multivariable-adjusted study results did not differ substantially. Therefore, the central goal of the present study (comparison of patients with and without an LVAD implant) may largely be unaffected by this limitation.
Conclusions
In summary, our data indicate that LVAD implantation results in a transient improvement in kidney function. Our data, however, also indicate that this LVAD-induced effect does not have an impact on survival, either on the waitlist or after heart transplantation. In contrast to severe kidney failure, mild to moderate renal dysfunction may therefore be considered a relative risk factor in heart failure patients only.
Figures
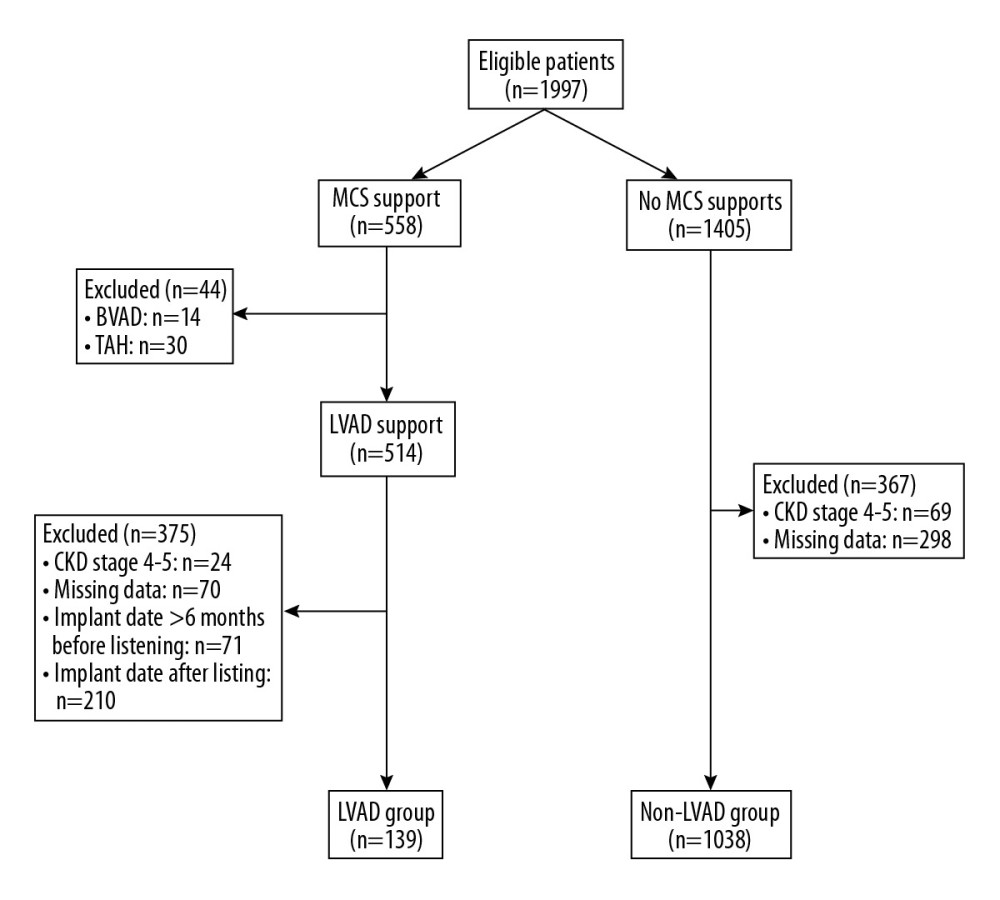 Figure 1. Flow chart of included and excluded studies. MCS – mechanical circulatory support; BVAD – biventricular assist device; TAH – total artificial heart; LVAD – left ventricular assist device.
Figure 1. Flow chart of included and excluded studies. MCS – mechanical circulatory support; BVAD – biventricular assist device; TAH – total artificial heart; LVAD – left ventricular assist device. 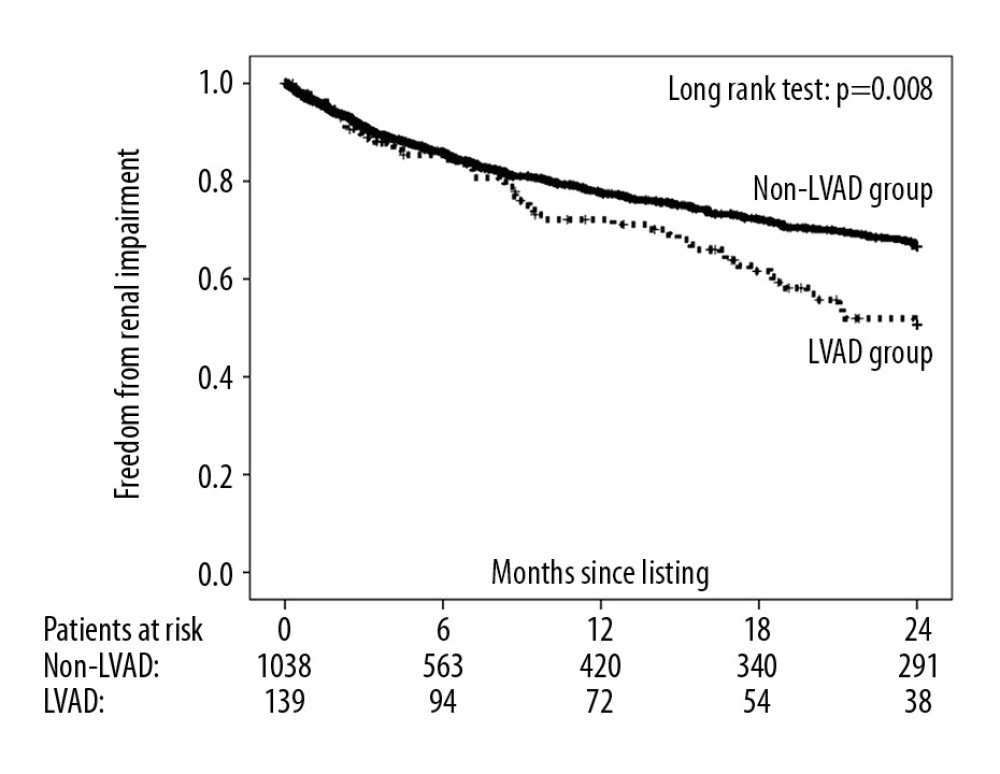 Figure 2. Two-year freedom from renal impairment after listing for a heart transplant by study group.
Figure 2. Two-year freedom from renal impairment after listing for a heart transplant by study group. 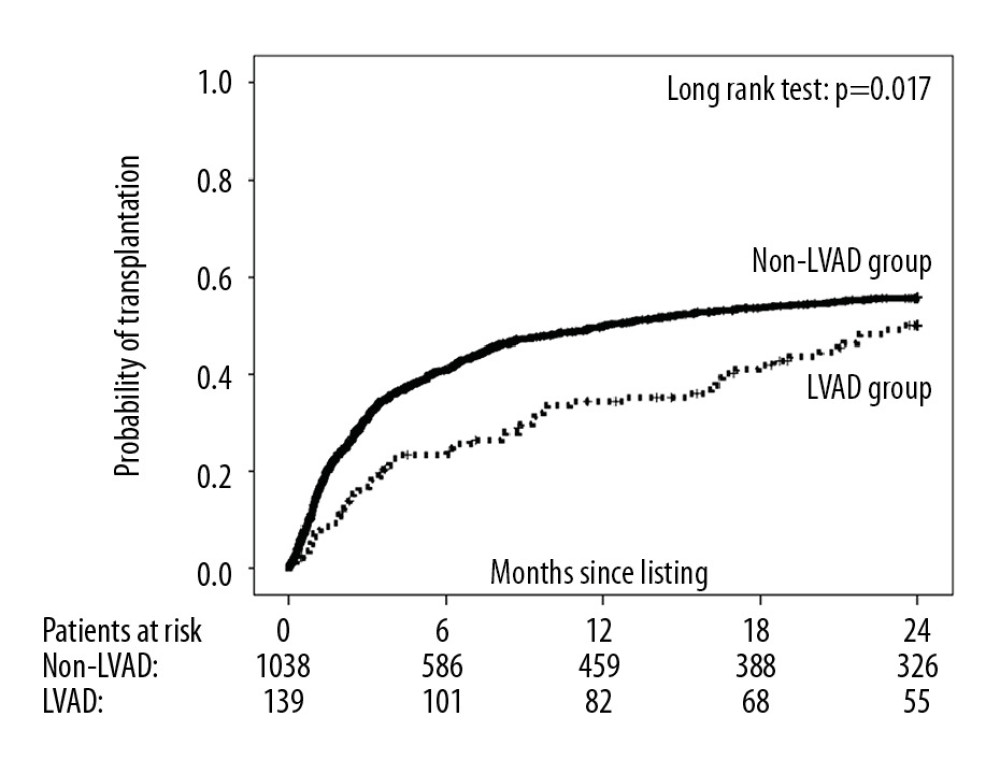 Figure 3. Two-year probability of transplantation after listing for a heart transplant by study group.
Figure 3. Two-year probability of transplantation after listing for a heart transplant by study group. 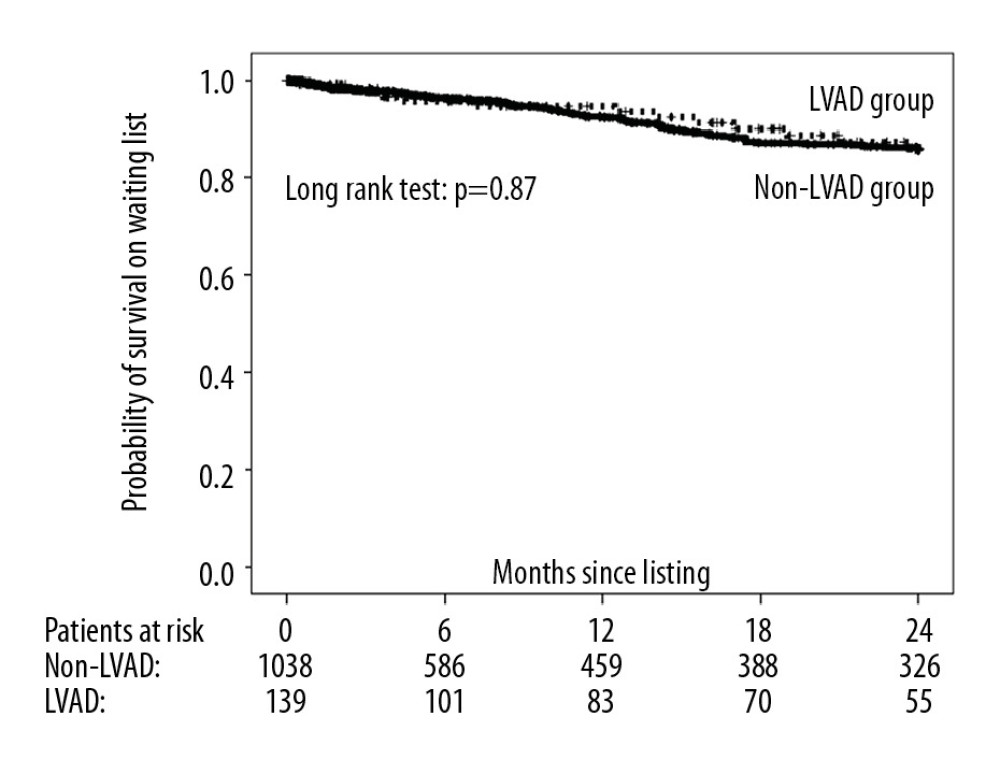 Figure 4. Two-year survival after listing for a heart transplant by study group.
Figure 4. Two-year survival after listing for a heart transplant by study group. 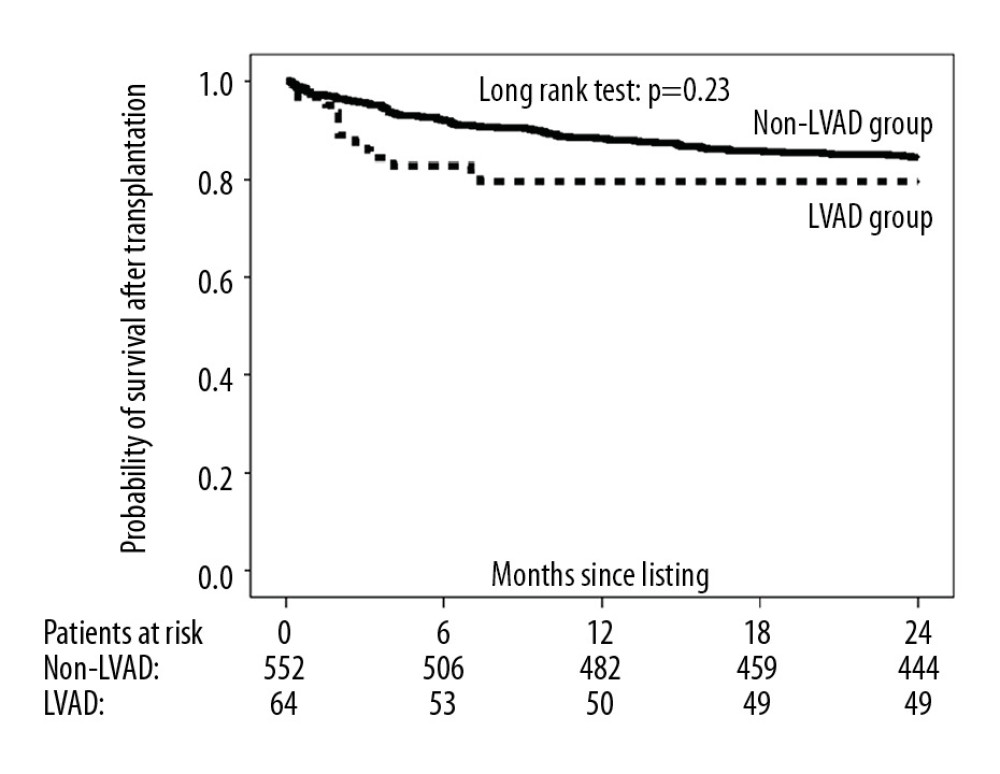 Figure 5. One-year survival after heart transplantation by study group.
Figure 5. One-year survival after heart transplantation by study group. References
1. Roger VL, Epidemiology of heart failure: Circ Res, 2013; 113; 646-59
2. Costanzo MR, Mills RM, Wynne J, Characteristics of “stage D” heart failure: Insights from the Acute Decompensated Heart Failure National Registry Longitudinal Module (ADHERE LM): Am Heart J, 2008; 155; 339-47
3. Hunt SA, Taking heart: Cardiac transplantation past, present, and future: N Engl J Med, 2006; 355; 231-35
4. Uriel N, Jorde UP, Woo Pak S, Impact of long term left ventricular assist device therapy on donor allocation in cardiac transplantation: J Heart Lung Transplant, 2013; 32; 188-95
5. Ammirati E, Oliva F, Cannata A, Current indications for heart transplantation and left ventricular assist device: A practical point of view: Eur J Intern Med, 2014; 25; 422-29
6. Kirklin JK, Naftel DC, Pagani FD, Seventh INTERMACS annual report: 15,000 patients and counting: J Heart Lung Transplant, 2015; 34; 1495-504
7. Costanzo MR, The cardiorenal syndrome in heart failure: Heart Fail Clin, 2020; 16; 81-97
8. Damman K, Valente MA, Voors AA, Renal impairment, worsening renal function, and outcome in patients with heart failure: An updated meta-analysis: Eur Heart J, 2014; 35; 455-69
9. Al-Ahmad A, Rand WM, Manjunath G, Reduced kidney function and anemia as risk factors for mortality in patients with left ventricular dysfunction: J Am Coll Cardiol, 2001; 38; 955-62
10. Hillege HL, Nitsch D, Pfeffer MA, Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM) Investigators: Renal function as a predictor of outcome in a broad spectrum of patients with heart failure: Circulation, 2006; 113; 671-78
11. Ljajikj E, Zittermann A, Koster A, Risk factors for adverse outcomes after left ventricular assist device implantation and extracorporeal cardiopulmonary resuscitation: Int J Artif Organs, 2019; 42; 207-11
12. Mehra MR, Kobashigawa J, Starling R, Listing criteria for heart transplantation: International Society for Heart and Lung Transplantation guidelines for the care of cardiac transplant candidates – 2006: J Heart Lung Transplant, 2006; 25; 1024-42
13. Roehm B, Vest AR, Weiner DE, Left ventricular assist devices, kidney disease, and dialysis: Am J Kidney Dis, 2017; 71; 257-66
14. Quader M, Goodreau AM, Johnson RM, Impact of renal function recovery utilizing left ventricular assist device support: J Card Surg, 2020; 35; 100-7
15. Brisco MA, Kimmel SE, Coca SG, Prevalence and prognostic importance of changes in renal function after mechanical circulatory support: Circ Heart Fail, 2014; 7; 68-75
16. Hoppe D, Schreiber HL, Richtlinien zur Organtransplantation gemäß §16 Transplantationsgesetz: Dtsch Arztebl, 2000; 97; 396-411 [in German]
17. Bundesärztekammer: Richtlinien für die Wartelistenführung und Organvermittlung zur Herz- und Herz-Lungen-Transplantation vom 12 09, 2013 http://www.bundesaerztekammer.de/fileadmin/user_upload/downloads/Herz-Lunge_09122013.pdf
18. Oz MC, Rose EA, Levin HR, Selection criteria for placement of left ventricular assist devices: Am Heart J, 1995; 129; 173-77
19. Williams MR, Oz MC, Indications and patient selection for mechanical ventricular assistance: Ann Thorac Surg, 2001; 71(3 Suppl); S86-91
20. Lin J, Knight EL, Hogan ML, Singh AK, A comparison of prediction equations for estimating glomerular filtration rate in adults without kidney disease: J Am Soc Nephrol, 2003; 14; 2573-80
21. Ricklefs M, Heimeshoff J, Hanke JS, The influence of less invasive ventricular assist device implantation on renal function: J Thorac Dis, 2018; 10(Suppl 15); S1737-42
22. Yalcin YC, Muslem R, Veen KM, Impact of continuous flow left ventricular assist device therapy on chronic kidney disease: A longitudinal multicenter study: J Card Fail, 2020; 26; 333-41
23. Kamboj M, Kazory A, Left ventricular assist device and the kidney: Getting to the heart of the matter: Blood Purif, 2019; 48; 289-98
24. Kazory A, Ronco C, Are we barking up the wrong tree? Rise in serum creatinine and heart failure: Blood Purif, 2019; 48; 193-95
25. Pinsino A, Mondellini GM, Royzman EA, Cystatin C-versus creatinine-based assessment of renal function and prediction of early outcomes among patients with a left ventricular assist device: Circ Heart Fail, 2020; 13(1); e006326
26. Scheld K, Zittermann A, Heer M, Nitrogen metabolism and bone metabolism markers in healthy adults during 16 weeks of bed rest: Clin Chem, 2001; 47; 1688-95
27. Jonsson A, Viklund I, Jonsson A, Comparison of creatinine-based methods for estimating glomerular filtration rate in patients with heart failure: ESC Heart Fail, 2020; 7(3); 1150-60
28. Yalcin YC, Bunge JJH, Guven G, Acute kidney injury following left ventricular assist device implantation: Contemporary insights and future perspectives: J Heart Lung Transplant, 2019; 38; 797-805
29. Go AS, Chertow GM, Fan D, Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization: N Engl J Med, 2004; 351; 1296-305
30. Bansal N, Hailpern SM, Katz R, Outcomes associated with left ventricular assist devices among recipients with and without end-stage renal disease: JAMA Intern Med, 2018; 178; 204-9
31. Walther CP, Niu J, Winkelmayer WC, Implantable ventricular assist device use and outcomes in people with end-stage renal disease: J Am Heart Assoc, 2018; 7; e008664
32. Kumar A, Howard A, Thomas CP, Estimated glomerular filtration rate at transplant listing and other predictors of post-heart transplant mortality and the development of ESRD: Transplantation, 2020 [Online ahead of print]
33. Kolsrud O, Karason K, Holmberg E, Renal function and outcome after heart transplantation: J Thorac Cardiovasc Surg, 2018; 155; 1593-604
34. Jawitz OK, Fudim M, Raman V, Renal outcomes in patients bridged to heart transplant with a left ventricular assist device: Ann Thorac Surg, 2020; 110(2); 567-74
35. Rose EA, Gelijns AC, Moskowitz AJ, Long-term use of a left ventricular assist device for end-stage heart failure: N Engl J Med, 2001; 345; 1435-43
36. Sunavsky J, Zittermann A, Oezpeker C, One-year clinical outcome in end-stage heart failure: comparison of “high urgent” listing for heart transplantation with mechanical circulatory support implantation: Thorac Cardiovasc Surg, 2015; 63; 647-52
37. Smits JM, de Vries E, De Pauw M, Is it time for a cardiac allocation score? First results from the Eurotransplant pilot study on a survival benefit-based heart allocation: J Heart Lung Transplant, 2013; 32; 873-80
38. Spaderna H, Zittermann A, Reichenspurner H, Role of depression and social isolation at time of waitlisting for survival 8 years after heart transplantation: J Am Heart Assoc, 2017; 6; e007016
Figures
 Figure 1. Flow chart of included and excluded studies. MCS – mechanical circulatory support; BVAD – biventricular assist device; TAH – total artificial heart; LVAD – left ventricular assist device.
Figure 1. Flow chart of included and excluded studies. MCS – mechanical circulatory support; BVAD – biventricular assist device; TAH – total artificial heart; LVAD – left ventricular assist device. Figure 2. Two-year freedom from renal impairment after listing for a heart transplant by study group.
Figure 2. Two-year freedom from renal impairment after listing for a heart transplant by study group. Figure 3. Two-year probability of transplantation after listing for a heart transplant by study group.
Figure 3. Two-year probability of transplantation after listing for a heart transplant by study group. Figure 4. Two-year survival after listing for a heart transplant by study group.
Figure 4. Two-year survival after listing for a heart transplant by study group. Figure 5. One-year survival after heart transplantation by study group.
Figure 5. One-year survival after heart transplantation by study group. Tables
In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588