23 October 2020: Original Paper
Transplant Radical Nephrectomy and Transplant Radical Nephroureterectomy for Renal Cancer: Postoperative and Survival Outcomes
Reza Nabavizadeh1ABCDEF*, Anika A. Noorali2ABCDEF, Salima S. Makhani2ABCDEF, Gordon Hong3ABC, Sarah Holzman4CDE, Dattatraya H. Patil1CDE, Frances Y. Kim1BCF, Paul L. Tso5ADE, Nicole A. Turgeon6DEF, Kenneth Ogan17ADEF, Viraj A. Master17ADEFDOI: 10.12659/AOT.925865
Ann Transplant 2020; 25:e925865
Abstract
BACKGROUND: The treatment of complex tumors in non-functioning renal transplants requiring surgical extirpation is challenging. Here, we report the largest series of patients who underwent transplant radical nephrectomy for renal cell carcinoma (RCC) and transplant radical nephroureterectomy for urothelial cell carcinoma (UCC) in their transplanted kidneys.
MATERIAL AND METHODS: From 2004 to 2018, 10 patients underwent transplant radical nephrectomy (7 patients) and nephroureterectomy (3 patients). Retrospective analyses, in terms of complications, oncological recurrence, and survival, of peri-operative and long-term outcomes, were performed.
RESULTS: Out of the 10 patients, 7 had RCC and 3 had UCC. No intraoperative mortality occurred. Three patients presented with Clavien-Dindo grade IIIa or greater within 30 days of surgery. Two patients died within 60 days of surgery, both due to vascular events: one due to myocardial infarction and one due to stroke. Two other patients died: one after 2.9 years, due to myocardial infarction, and the other after 6 years, due to unknown reasons. At the 7-year follow-up, there was a 60% overall survival rate. For all patients, average survival post-nephrectomy was approximately 4.5 years, including the 6 living patients and 4 deceased patients. Importantly, there was no observed cancer recurrence.
CONCLUSIONS: This study reports outcomes of the largest series of transplant radical nephrectomy and nephroureterectomy for malignancies of renal allografts. In the optimized setting, extirpative surgeries appear safe, with favorable long-term oncological and survival outcomes.
Keywords: Kidney Neoplasms, Nephrectomy, nephroureterectomy, Ureteral Neoplasms
Background
Genitourinary cancers account for approximately 15% of all tumors in transplant recipients, and a large portion of these cases are renal cancers [1,2]. Pathologically, the majority of renal cancers are renal cell carcinomas (RCC), followed by urothelial cell carcinomas (UCC). Transplant recipients have a 100-fold increased risk of developing RCC compared to the general population [1]. Development of RCC is more common in the native kidney compared to the allografts [2–7]. UCC arises from the urothelium of the collecting system and can be seen in the kidney (minor and major calyxes and renal pelvis), ureter, or both [8–10]. For UCC tumors, transplant radical nephroureterectomy, and for RCC tumors, transplant radical nephrectomy, are considered the standards of care for patients with failed allografts on dialysis or patients who are not candidates for nephron-sparing surgery. Despite the clinical relevance, there is scant data on short-term and long-term outcomes of transplant radical nephrectomy and nephroureterectomy. Many surgeons consider the surgeries challenging and even question whether the procedures are safe in immunosuppressed patients. Here, we report the outcomes of transplant radical nephrectomy and nephroureterectomy in our cohort of post-transplant patients with allograft cancer.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
This study retrospectively evaluated a case series of patients who underwent transplant radical nephrectomy and nephroureterectomy for malignancy of their renal allograft between 2004 and 2018. Patient data were collected using a combination of Institution Kidney Cancer Database, Natural Language search of electronic charts, and Social Security Death Index (SSDI). Inclusion criteria consisted of patients at our institution who underwent transplant radical nephrectomy and nephroureterectomy for kidney cancer. The surgery was performed in patients who met one or more of the following criteria: (i) dialysis at time of cancer diagnosis, (ii) tumor size and location that precluded partial nephrectomy, (iii) presence of multifocal disease on renal allograft, or (iv) urothelial cell carcinoma arising from the collecting system of the transplanted kidney. Exclusion criteria consisted of patients undergoing surgeries for non-oncological purposes. The Institutional Review Board (IRB) at our institution approved this study.
PATIENT DATA:
The following clinical and demographic variables were collected: race, sex, age of donor, donor type, age of patient at time of surgery, time from transplantation until diagnosis of malignancy, end-stage renal disease (ESRD) as the primary diagnosis, and tumor characteristics (pathological findings, tumor size, stage, type, presence of multifocality and grade). History of prior transplants, multi-organ transplantations, hypertension, smoking, and obesity were also recorded. Intraoperative variables included the following: surgery duration, blood loss, length of stay, and postoperative complications per Clavien-Dindo Classification protocol [11,12].
DIAGNOSIS AND TREATMENT:
Each tumor was identified with various radiographic techniques: ultrasound, computed tomography (CT) and/or magnetic resonance imaging (MRI) followed by confirmation by CT-guided biopsy or endoscopic biopsy via ureteroscopy for urothelial carcinomas. None of the patients had radiographic evidence of metastasis. Image-guided biopsy is not routinely required for kidney cancer diagnosis in native kidneys. Renal biopsy, historically, did not gain much attention due to concern for biopsy tract seeding. However, recent studies have shown that with evolved techniques, this risk is no longer a concern. Therefore, whenever the diagnosis is not clear, a biopsy might provide additional information to both the clinician and the patient. In cases of tumors in a transplanted kidney, a preoperative biopsy can confirm the diagnosis before proceeding with a potentially complex surgical procedure.
Prior to surgery, patients received preoperative cardiology clearance if deemed necessary by the surgeon. All surgeries were performed as a multidisciplinary surgical collaboration between the transplant surgery and urology services.
SURGICAL TECHNIQUE:
Transplant radical nephrectomy was undertaken through a longitudinal midline or Gibson incision. The allografts were exposed via a trans-peritoneal approach, allowing for visualization and access to the external iliac vein and artery. The posterior peritoneum overlying the great vessels was opened, and the vena cava and aorta were identified for proximal vascular control, as were the distal iliac vessels. The transplant renal unit was reflected medially from the sidewall, along with all of the surrounding fibrofatty tissue. Importantly, intraoperative ultrasonography with Doppler was performed to trace the position of the vessels. Using Doppler ultrasonography is extremely helpful in these cases, as the desmoplastic reaction after transplant often precludes direct vessel visualization.
The superior aspect of the kidney was dissected off the psoas muscle and the perinephric fat removed. The peritoneum was incised down to the level of the bladder, and that allowed for medial mobilization. The lower pole of the kidney was reflected upwards, allowing for inferior exposure of the iliac vessels, and the vessels were traced to the renal pedicle. Care was taken to differentiate the blood supply to the lower extremity from the renal pedicle. The absence of normal anatomic planes made differentiating the blood supply the most challenging component of the transplant nephrectomy. The hilum was divided with an endovascular stapler. In the 3 patients with UCC, in addition to performing transplant radical nephrectomy, the entire ureter and bladder cuff were also removed.
OUTCOMES OF INTEREST:
The primary outcome of interest was overall survival, defined as the time from the date of surgery to the date of last follow-up or death. A Kaplan-Meier plot was generated to evaluate overall survival of patients over a 7-year follow-up period. Secondary outcomes were postoperative complications using the Clavien-Dindo criteria [11], length of stay (LOS), and 30-day hospital readmissions. Data were collected from medical records, Social Security National Index (SSNDI), and follow-up phone calls.
Results
PATIENT CHARACTERISTICS:
Between 2004 and 2018, at our institution, 10 patients underwent transplant radical nephrectomy and nephroureterectomy for kidney cancer. Seven out of the 10 patients were male, with average age of 54.6 years (range, 44 to 63). Patient characteristics are presented in Table 1. The most common cause of ESRD requiring transplantation was glomerulonephritis. All 10 patients had a history of hypertension, and 5 patients had a history of smoking. Three patients had a previously failed renal transplant. The mean number of years from the time of kidney transplant to surgery was 15 years, ranging from 8 to 25 years (Table 2).
All patients had received immunosuppression with a calcineurin-inhibitor-based regimen. Seven of the 10 patients (70%) were on dialysis at the time of cancer diagnosis. All 10 patients were placed on hemodialysis after transplant radical nephrectomy; one patient subsequently underwent peritoneal catheter placement. Seven out of 10 patients (70%) were symptomatic, with the most common chief complaint of painless gross hematuria. In the preoperative setting, MRI was the most common form of imaging utilized to detect the tumor.
INTRAOPERATIVE OUTCOMES:
Intra- and postoperative data are displayed in Table 3. The average surgery duration and mean blood loss of all our patients who either underwent transplant radical nephrectomy or nephrouretectomy was 165 min and 460 mL, respectively. One patient (Case 4) had intraoperative surgical complications of the external iliac artery and vein requiring repair by vascular surgery, which resulted in 336 min of operative time and 700 mL of blood loss. The average surgery duration and mean blood loss for other patients (excluding Case 4) was 126 min and 183 mL, respectively.
For the 3 patients who underwent transplant radical nephroureterectomy for UCC, operative time (and blood loss) was 150 min in Case 9 (800 mL), 173 min in Case 3 (500 mL), and 336 min in Case 1 (700 mL). The patient in Case 1 underwent bilateral native nephroureterectomy, cystoprostatectomy, and retroperitoneal lymph node dissection in addition to the transplant radical nephroureterectomy, with intraoperative complications requiring a general surgical consult for splenic injury. Furthermore, this patient was admitted to the intensive care unit (ICU) and subsequently died at 35 days after surgery. Of note, the patient in Case 1 had high-grade papillary UCC with invasion to the lamina propria of the bladder and no invasion into the muscularis with chromosomal analysis, indicating the transplanted kidney as the source. Therefore, it was decided to perform a transplant radical nephrectomy in addition to a cystoprostatectomy. The patient had non-functioning bilateral native kidneys that were still producing urine. Thus, it was decided to remove the bilateral native kidneys as opposed to performing urinary diversion.
POSTOPERATIVE OUTCOMES:
Mean length of stay for all the patients was 6 days. Three patients presented with Clavien-Dindo grade IIIa or greater within 30 days of surgery. One patient had hospital readmission within 30 days of surgery. To date, all living patients are without evidence of kidney cancer recurrence, confirmed by regular cross-sectional imaging follow-up. Profiles of each patient are provided in Table 3.
On surgical specimen pathology, 5 patients had papillary RCC, 3 patients had UCC, and 2 patients had Acquired Cystic Kidney Disease Associated Carcinoma (ACDAC) (Figure 1). All 3 UCC patients had high-grade and invasive disease. While the location of the UCC tumors was frequently at the ureteral neocystotomy site, the locations of the RCC tumors varied. Nine of the 10 patients (90%) had tumors confined to the organ, and multifocal tumors were present in 5 patients. Tumor characteristics are presented in Table 4.
SURVIVAL OUTCOMES:
A Kaplan-Meier curve in Figure 2 represents the overall survival of all patients. Long-term survival was determined, and a 7-year follow-up was available for all patients, with the exception of 2 patients (Cases 1 and 10) who died within 60 days. At the 7-year follow-up, there was a 60% overall survival rate. Overall survival was approximately 4.5 years, including 6 living patients and the 4 deceased patients. With the exception of the 2 patients who died within 60 days, the remaining 8 patients survived at least 5.6 years after nephrectomy. Despite the death of 2 additional patients in the 7-year follow-up period, there are 6 patients still alive, with an average survival of 6.1 years. None of those 6 patients have experienced recurrence of disease.
DECEASED PATIENTS:
Among the 4 deceased patients, the average survival was 2.3 years. All deceased patients were white males and recipients of deceased donor (DD) allografts, with an average age of 53.8 years at the time of surgery. On final pathology, 3 of the 4 deceased patients had multifocal disease.
CASE 1:
A 37-year-old man received a kidney from a 37-year-old DD donor. Eight years after transplantation, the recipient underwent bilateral native nephroureterectomy, cystoprostatectomy, and retroperitoneal lymph node dissection, in addition to the transplant radical nephroureterectomy for high-grade papillary UCC with invasion to the lamina propria of the bladder and no invasion into the muscularis UCC. Despite the complicated surgery, he had an unremarkable initial postoperative course. However, 35 days after surgery, he experienced a myocardial infarction, resulting in his death.
CASE 5:
A 35-year-old man with ESRD secondary to immunoglobulin A (IgA) nephropathy received a kidney from a 30-year-old DD donor. The recipient underwent a laparoscopic nephrectomy of his right native kidney, as well as a left transplant radical nephrectomy of his allograft 11 years after transplantation, due to multifocal papillary RCC. He presented with multiple postoperative complications, including acute blood loss anemia, admission to the ICU for pneumonia, and
CASE 7:
A 51-year-old man received a transplant from an 18-year-old DD. The recipient underwent transplant radical nephrectomy 12 years after transplantation, due to papillary RCC, and died 6.1 years after nephrectomy. The cause of death was unknown.
CASE 10:
A 63-year-old man received a renal transplant from a DD in 1992. The recipient underwent transplant radical nephrectomy 25 years after transplantation, because of ACDAC, and died within 49 days after nephrectomy because of stroke.
LIVING PATIENTS:
Two living patients had complications greater than Clavien-Dindo grade IIIa. Case 4 had an intraoperative external iliac artery and a vein injury that required transfusion and ICU care in the postoperative period. Case 6 was noted to have a non-ST elevation myocardial infarction (NSTEMI) and multilobar pneumonia on postoperative day 4. Among the 6 living patients, there was no evidence of recurrence of kidney cancer at the average follow-up of 6.1 years (range, 1.9 to 10.3 years).
Discussion
Despite the possible benefits expected from routine non-oncologic transplant nephrectomy, routine excision is not the standard, because it is a technically demanding procedure [13,14]. Even without the presence of malignancy, transplant radical nephrectomy of a failed allograft is associated with improvement in hematological, biochemical, and clinical parameters, such as erythropoietin (EPO) resistance index, serum levels of albumin, prealbumin, ferritin, fibrinogen, c-reactive protein (CRP), and erythrocyte sedimentation rate (ESR) [15,16]. Studies have shown that the removal of the patient’s transplanted kidney at the time the patient has progressed to requiring dialysis is independently associated with increased survival [17,18]. An important consideration is that the removal of the transplanted kidney can be detrimental to the outcome of subsequent transplantations, by virtue of the increased level of antibodies to mismatched antigens, increased rate of primary non-function, and delayed graft function [15,19]. Many of the studies reporting these outcomes were retrospective. Moreover, with univariate analysis of small numbers of patients, conclusions drawn from such studies should be made with caution.
Additionally, these concerns are not clinically relevant in the case of renal allograft malignancies because those patients nevertheless would not be candidates for subsequent transplantation without transplant radical nephrectomy or nephrouretectomy. Systemic therapies are often more challenging for malignancies of renal allograft compared to kidney cancers in native functioning kidneys, and often are not ideal treatment options [16]. Extirpative surgery for malignancies of the renal allograft can benefit these patients in various ways by removing both the malignancy and the failed allograft.
Immunomodulation and immunosuppression induced to allow viability of the renal allograft can provide a more favorable niche for the malignant cells to emerge and thrive [2,10,20,21]. In renal transplant patients, allograft malignancies in the immunosuppressed state are often more aggressive [22]. Despite the clinical relevance, little is known about outcomes of transplant radical nephrectomy performed for allograft malignancies [15,18,23–25]. Tables 5 and 6 summarize the published literature on transplant radical nephrectomy performed for RCC and transplant radical nephroureterectomy for UCC [16,26–31]. While the existing literature supports nephron-sparing surgery, when indicated, to preserve renal allograft function and avoid dialysis, limited data informs clinicians regarding outcomes of transplant radical nephrectomy or nephroureterectomy in patients with either an already failed allograft or, when nephron-sparing surgery is not feasible, because of the complexity of the cancer, determined by the tumor size, location, grade and multifocality [5,32–34]. Transplant radical nephrectomy with the excision of the ureter and bladder cuff is the optimal approach for patients with UCC, regardless of the tumor size, location, or feasibility of nephron-sparing surgery [8–10,35,36]. In our cohort, 7 patients had failed transplants and were on dialysis, and 3 had tumors not suitable for nephron-sparing surgery.
To the best of our knowledge, this study is the largest series to date to evaluate outcomes of transplant radical nephrectomy or nephroureterectomy. In our series, 2 patients died within 60 days of surgery; one because of myocardial infarction and one due to stroke. Within the time frame of a 7-year follow-up, 2 other patients died: one after 2.9 years, because of myocardial infarction, and another after 6.1 years, for unknown reason. Patients who are considered for these surgeries should be counselled regarding peri-, intra-, and postoperative risks, especially considering the fact that this patient pool usually has multiple comorbidities such as history of ESRD, immunosuppression, hypertension, and vasculopathy, which increases their risk for peri- and postoperative morbidity and mortality.
Although transplant radical nephrectomy and nephroureterectomy are challenging surgeries, a prepared multidisciplinary surgical collaboration can achieve favorable surgical and oncological outcomes and minimize the risk of complications. In our series there was one instance of vascular injury and one instance of splenic injury occurred, and both received successful repair by vascular and general surgery teams. No intraoperative mortality occurred. Most of the patients in our cohort had an unremarkable postoperative course and no recurrence of cancer was observed on surveillance follow-up.
In this series, only 3 of the 10 patients (30%) presented with complications greater than Clavien-Dindo grade IIIa within 30 days of surgery. Two of these patients (Cases 4 and 6) had significant comorbidities, including obesity, hypertension, and smoking. Despite these risk factors, both patients are alive today, approximately 10- and 6-years after nephrectomy, respectively, with unremarkable follow-up. The long-term outcomes of this study are promising and suggest that, in capable hands, transplant radical nephrectomy and nephroureterectomy are safe and feasible surgeries. Although this study includes the largest cohort of patients who underwent transplant radical nephrectomy and nephroureterectomy, the small number of patients imposes limitations on the study’s conclusions. These surgeries, moreover, were accomplished with a multidisciplinary team at a tertiary academic center, which is a limitation in terms of external validity, if the same surgery is attempted in a different practice setting. More research on this topic, perhaps with larger numbers of patients and from different institutions, may be necessary to further investigate the outcomes.
Conclusions
This study reports the outcome of the largest case series to date on transplant radical nephrectomy and nephroureterectomy for malignancies of renal allografts. These extirpative surgeries, in the optimized setting, appear to be safe, with favorable long-term oncological and survival outcomes.
Figures
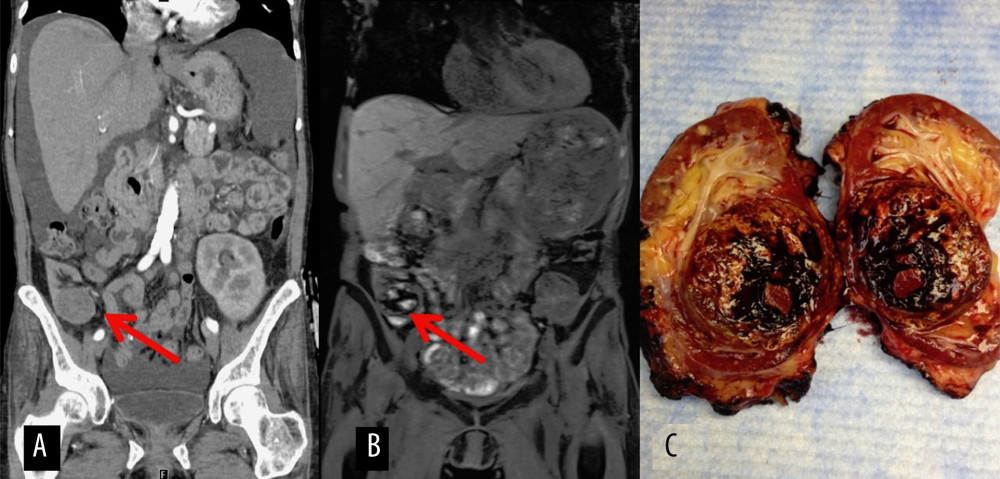 Figure 1. Preoperative imaging of Case 2 with acquired cystic disease-associated RCC and gross specimen. (A) CT abdomen and pelvis from 3 years prior to transplant radical nephrectomy showing a cyst in the right transplant renal unit. (B) Preoperative MRI in 2013, renal mass in transplant unit marked by arrow. (C) Gross specimen from Case 2.
Figure 1. Preoperative imaging of Case 2 with acquired cystic disease-associated RCC and gross specimen. (A) CT abdomen and pelvis from 3 years prior to transplant radical nephrectomy showing a cyst in the right transplant renal unit. (B) Preoperative MRI in 2013, renal mass in transplant unit marked by arrow. (C) Gross specimen from Case 2. 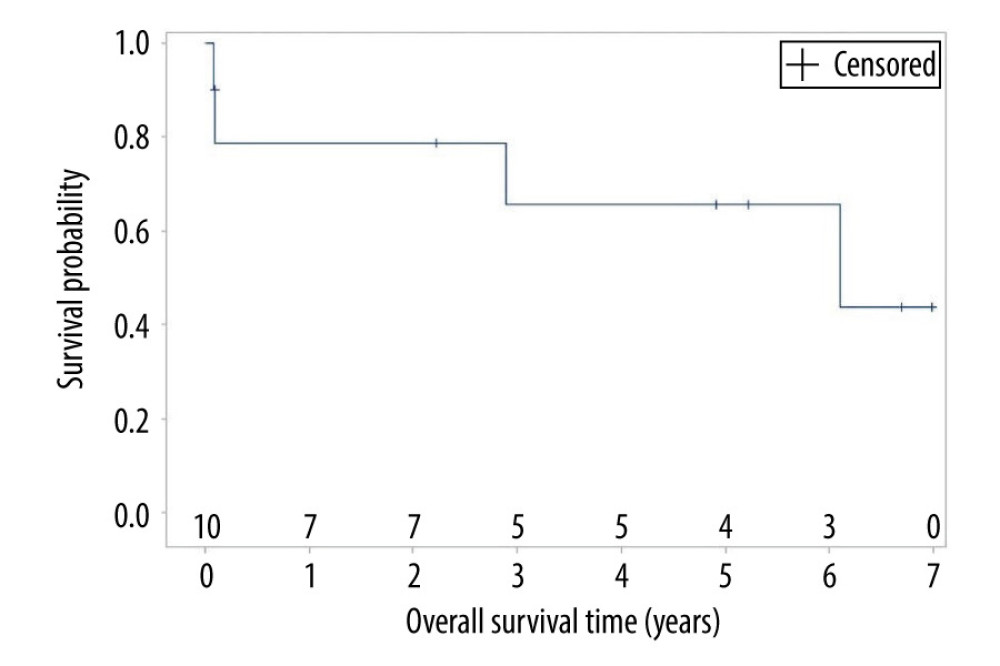 Figure 2. Overall survival among transplant radical nephrectomy and nephroureterectomy patients. (+) Censored defined as lost to follow-up or death. Overall Survival (OS) time defined as follow-up period of 7 years with 4 patient deaths.
Figure 2. Overall survival among transplant radical nephrectomy and nephroureterectomy patients. (+) Censored defined as lost to follow-up or death. Overall Survival (OS) time defined as follow-up period of 7 years with 4 patient deaths. Tables
Table 1. Patient characteristics cohort demographic.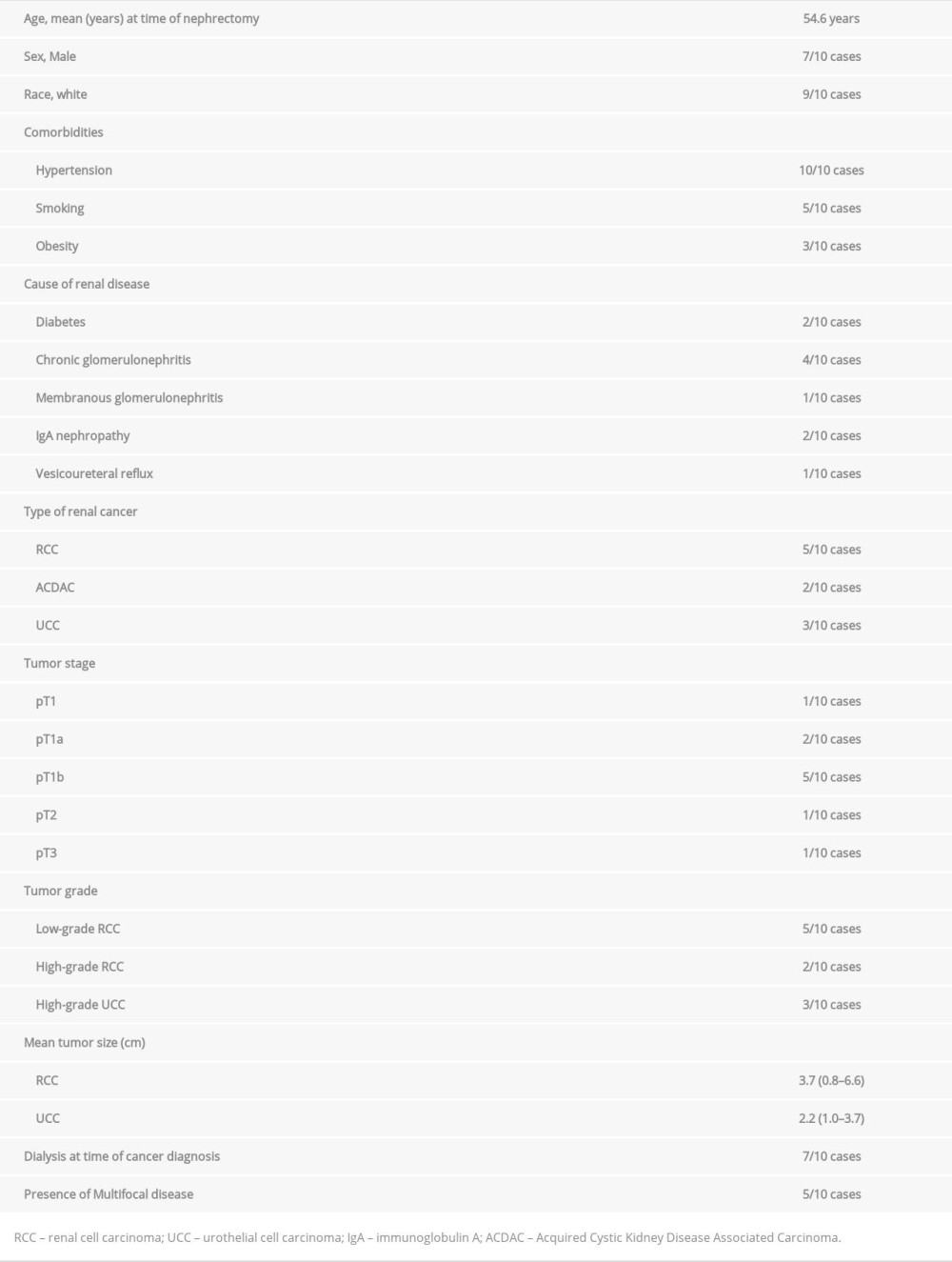 Table 2. Transplant characteristics.
Table 2. Transplant characteristics. Table 3. Intra- and postoperative characteristics.
Table 3. Intra- and postoperative characteristics.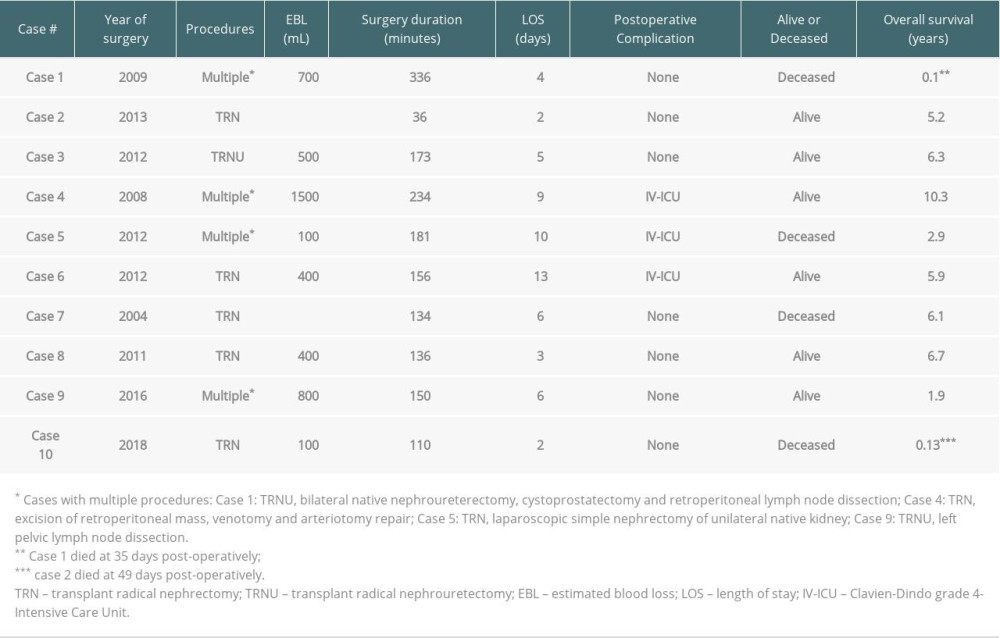 Table 4. Tumor characteristics.
Table 4. Tumor characteristics.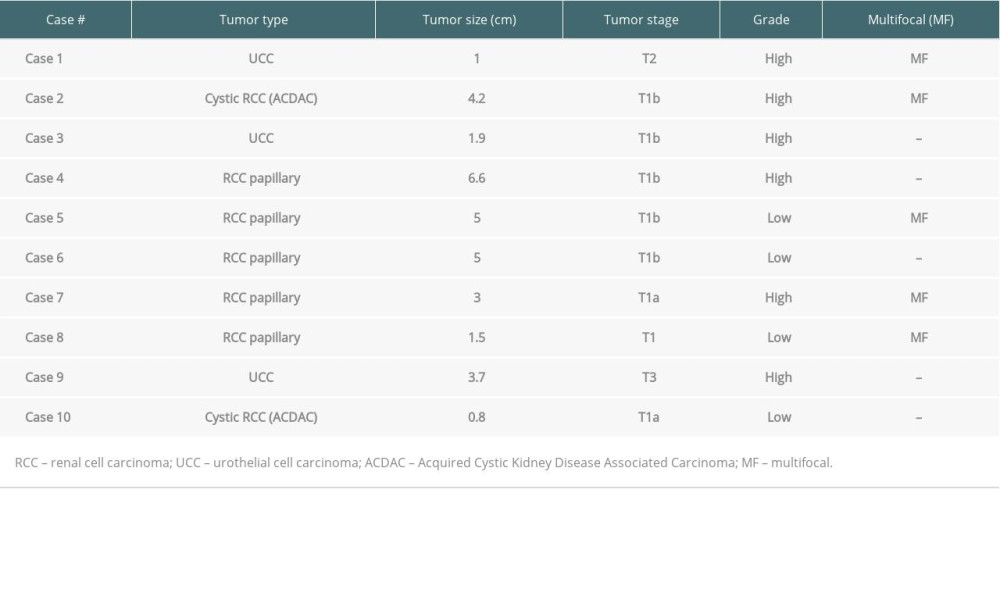 Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy.
Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy.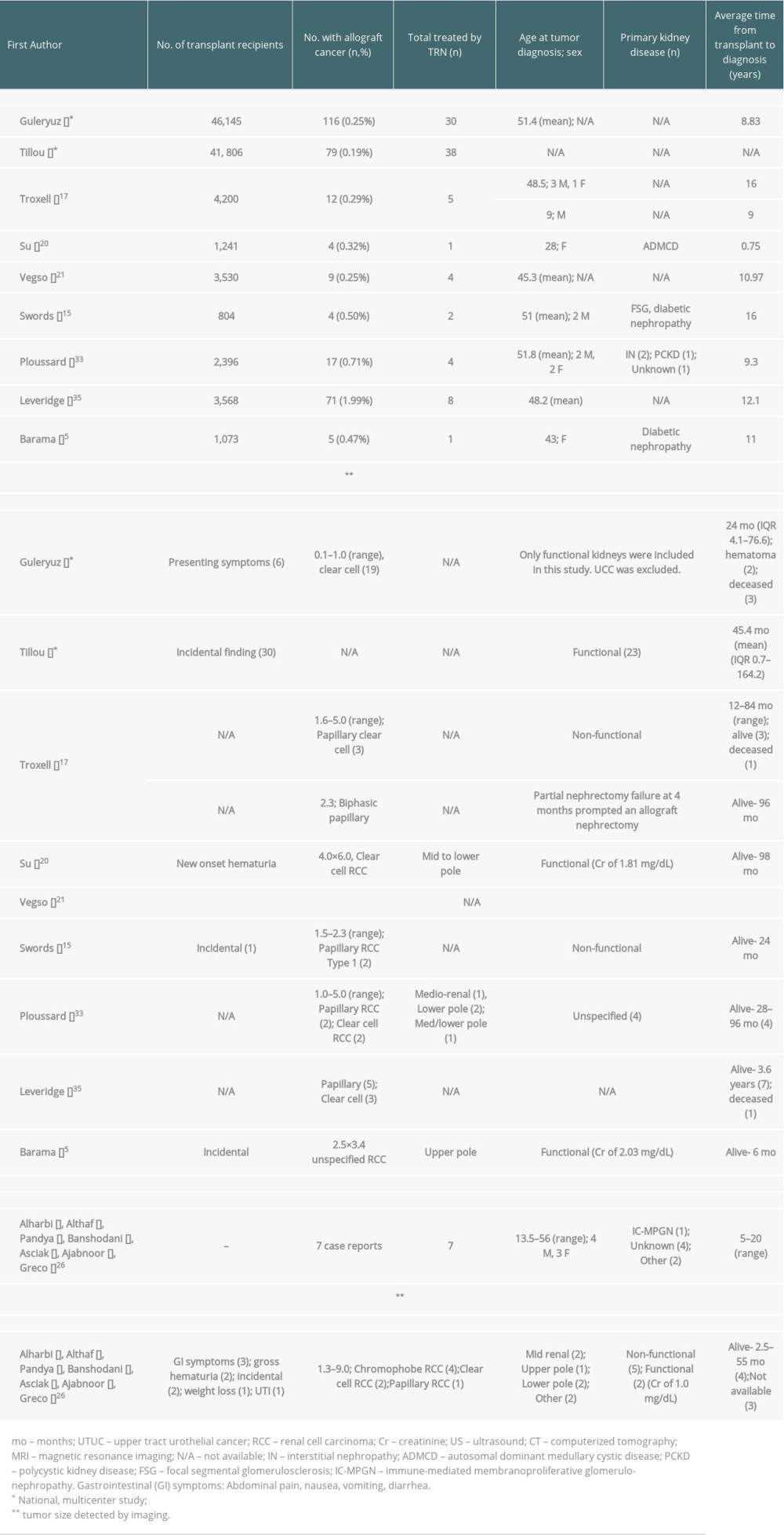 Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy.
Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy.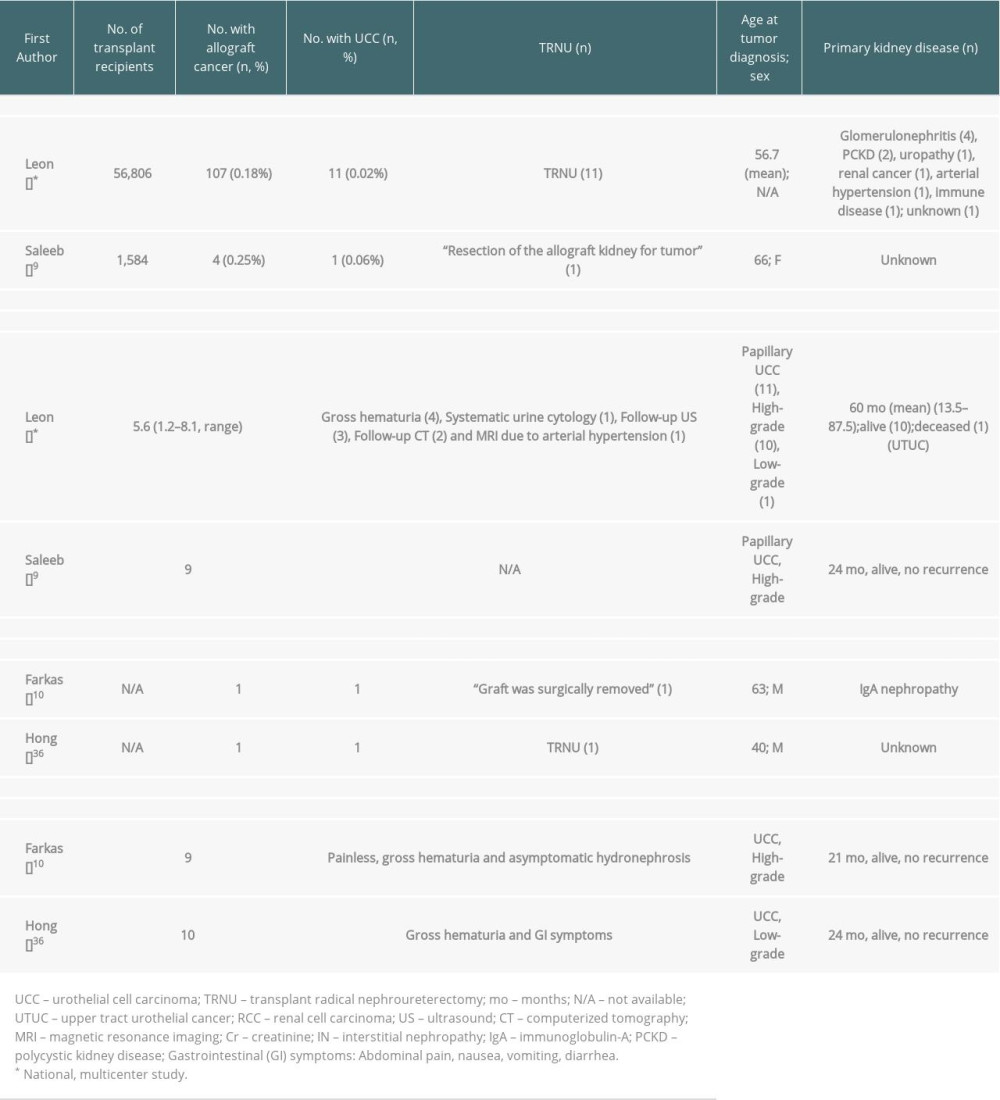
References
1. Moudouni SM, Lakmichi A, Tligui M, Renal cell carcinoma of native kidney in renal transplant recipients: BJU Int, 2006; 98(2); 298-302
2. Penn I, Sarcomas in organ allograft recipients: Transplantation, 1995; 60(12); 1485-91
3. Gunji Y, Sakamoto K, Yamada K, Successful surgical treatment of renal cell carcinoma in a transplanted kidney from a cadaveric donor: Report of a case: Surg Today, 2001; 31(4); 374-77
4. Ianhez LE, Lucon M, Nahas WC, Renal cell carcinoma in renal transplant patients: Urology, 2007; 69(3); 462-64
5. Barama A, St-Louis G, Nicolet V, Renal cell carcinoma in kidney allografts: A case series from a single center: Am J Transplant, 2005; 5(12); 3015-18
6. Tyden G, Tollemar J, Bolinder J, Combined pancreas and kidney transplantation improves survival in patients with end-stage diabetic nephropathy: Clin Transplant, 2000; 14(5); 505-8
7. Neuzillet Y, Lay F, Luccioni A: Cancer, 2005; 103(2); 251-57
8. Leon G, Szabla N, Boissier R, Kidney graft urothelial carcinoma: Results from a multicentric retrospective national study: Urology, 2020; 135; 101-5
9. Saleeb R, Faragalla H, Yousef G, Malignancies arising in allograft kidneys, with a first reported translocation RCC post-transplantation: A case series: Pathol Res Pract, 2015; 211(8); 584-87
10. Farkas ÁZ, Török S, Kovács JB: Transplant Proc, 2019; 51(4); 1281-85
11. Clavien PA, Barkun J, de Oliveira ML, The Clavien-Dindo classification of surgical complications: Five-year experience: Ann Surg, 2009; 250(2); 187-96
12. Dindo D, Clavien P-A, What is a surgical complication?: World J Surg, 2008; 32(6); 939-41
13. Ucar AR, Demir E, Mehmel SS, Transplant patients with failing renal allografts: Exp Clin Transplant, 2018; 16(1); 4-8
14. Guleryuz K, Doerfler A, Codas R, A national study of kidney graft tumor treatments: Toward ablative therapy: Surgery, 2016; 160(1); 237-44
15. Swords DC, Al-Geizawi SM, Farney AC, Treatment options for renal cell carcinoma in renal allografts: A case series from a single institution: Clin Transplant, 2013; 27(2); E199-E205
16. Asciak R, Buttigieg J, Buhagiar L: CEN Case Rep, 2016; 5(1); 99-102
17. Troxell ML, Higgins JP, Renal cell carcinoma in kidney allografts: Histologic types, including biphasic papillary carcinoma: Hum Pathol, 2016; 57; 28-36
18. Lee HH, Choi KH, Yang SC, Han WK, Renal cell carcinoma in kidney transplant recipients and dialysis patients: Korean J Urol, 2012; 53(4); 229-33
19. Tillou X, Doerfler A, Collon S: Am J Transplant, 2012; 12(12); 3308-15
20. Su MZ, Campbell NA, Lau HM, Management of renal masses in transplant allografts at an Australian kidney-pancreas transplant unit: Transplantation, 2014; 97(6); 654-59
21. Végső G, Toronyi É, Deák PÁ, Detection and management of renal cell carcinoma in the renal allograft: Int Urol Nephrol, 2013; 45(1); 93-98
22. Barrett WL, First MR, Aron BS, Penn I, Clinical course of malignancies in renal transplant recipients: Cancer, 1993; 72(7); 2186-89
23. Adami J, Gäbel H, Lindelöf B, Cancer risk following organ transplantation: A nationwide cohort study in Sweden: Br J Cancer, 2003; 89(7); 1221-27
24. Akoh JA, Transplant nephrectomy: World J Transplant, 2011; 1(1); 4-12
25. Ariyarathenam A, Bamford A, Akoh JA, Transplant nephrectomy – A single-center experience: Saudi J Kidney Dis Transpl, 2015; 26(6); 1108-12
26. Alharbi A, Al Turki MS, Aloudah N, Alsaad KO, Incidental eosinophilic chromophobe renal cell carcinoma in renal allograft: Case Rep Transplant, 2017; 2017 4232474
27. Althaf MM, Al-Sunaid MS, Abdelsalam MS, Chromophobe renal cell carcinoma occurring in the renal allograft of a transplant recipient presenting with weight loss: Saudi J Kidney Dis Transpl, 2016; 27(1); 139
28. Pandya V, Sutariya H: Int J Organ Transplant Med, 2016; 7(1); 51-56
29. Banshodani M, Kawanishi H, Marubayashi S: Case Rep Transplant, 2015; 2015 679262
30. Ajabnoor R, Al-Sisi E, Zafer G, Al-Maghrabi J, Chromophobe renal cell carcinoma in renal allograft: Case report: Life Sci J, 2014; 11(7); 9-14
31. Greco AJ, Baluarte JH, Meyers KE, Chromophobe renal cell carcinoma in a pediatric living-related kidney transplant recipient: Am J Kidney Dis, 2005; 45(6); e105-8
32. Vasisth G, Kapoor A, Piercey K, Lambe S, Renal cell carcinoma in renal allograft: Case series and review of literature: Urol Ann, 2018; 10(2); 229-32
33. Ploussard G, Chambade D, Meria P: BJU Int, 2012; 109(2); 195-99
34. Tsui K-H, van Ophoven A, Shvarts O, Belldegrun A, Nephron-sparing surgery for renal cell carcinoma: Rev Urol, 1999; 1(4); 216-25
35. Leveridge M, Musquera M, Evans A, Renal cell carcinoma in the native and allograft kidneys of renal transplant recipients: J Urol, 2011; 186(1); 219-23
36. Hong YA, Hwang HS, Sul HJ, Transitional cell carcinoma involving graft kidney in a kidney transplant recipient: A case report: BMC Nephrol, 2017; 18(1); 299
Figures
 Figure 1. Preoperative imaging of Case 2 with acquired cystic disease-associated RCC and gross specimen. (A) CT abdomen and pelvis from 3 years prior to transplant radical nephrectomy showing a cyst in the right transplant renal unit. (B) Preoperative MRI in 2013, renal mass in transplant unit marked by arrow. (C) Gross specimen from Case 2.
Figure 1. Preoperative imaging of Case 2 with acquired cystic disease-associated RCC and gross specimen. (A) CT abdomen and pelvis from 3 years prior to transplant radical nephrectomy showing a cyst in the right transplant renal unit. (B) Preoperative MRI in 2013, renal mass in transplant unit marked by arrow. (C) Gross specimen from Case 2. Figure 2. Overall survival among transplant radical nephrectomy and nephroureterectomy patients. (+) Censored defined as lost to follow-up or death. Overall Survival (OS) time defined as follow-up period of 7 years with 4 patient deaths.
Figure 2. Overall survival among transplant radical nephrectomy and nephroureterectomy patients. (+) Censored defined as lost to follow-up or death. Overall Survival (OS) time defined as follow-up period of 7 years with 4 patient deaths. Tables
 Table 1. Patient characteristics cohort demographic.
Table 1. Patient characteristics cohort demographic. Table 2. Transplant characteristics.
Table 2. Transplant characteristics. Table 3. Intra- and postoperative characteristics.
Table 3. Intra- and postoperative characteristics. Table 4. Tumor characteristics.
Table 4. Tumor characteristics. Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy.
Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy. Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy.
Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy. Table 1. Patient characteristics cohort demographic.
Table 1. Patient characteristics cohort demographic. Table 2. Transplant characteristics.
Table 2. Transplant characteristics. Table 3. Intra- and postoperative characteristics.
Table 3. Intra- and postoperative characteristics. Table 4. Tumor characteristics.
Table 4. Tumor characteristics. Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy.
Table 5. Literature review of renal allograft RCC treated by transplant radical nephrectomy. Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy.
Table 6. Literature review of renal allograft UCC treated by transplant radical nephroureterectomy. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








