05 January 2022: Original Paper
Reconstruction of Separated Intrahepatic Ducts Using the Cystic and Common Hepatic Ducts in Right-Lobe Living-Donor Liver Transplantation: Experiences of Surgical Techniques, Biliary Complications, and Outcomes in a Single Institution
Po-Jung Hsu1ABCDEF, Hao-Chien Hung12ABCDEF, Ching-Sung Lee3A, Kuang-Tse Pan4A, Jin-Chiao Lee32A, Yu-Chao Wang32A, Chih-Hsien Cheng32A, Tsung-Han Wu32A, Chen-Fang Lee32A, Hong-Shiue Chou32A, Kun-Ming Chan32A, Wei-Chen Lee32A, Ting-Jung Wu12ACDEF*DOI: 10.12659/AOT.934459
Ann Transplant 2022; 27:e934459
Abstract
BACKGROUND: Duct-to-duct biliary reconstruction has been increasingly used in living-donor liver transplantation. Information regarding dual duct-to-duct biliary anastomoses is limited. We present the largest case series to date on the use of the cystic and common hepatic ducts as dual-ductal anastomosis, along with long-term follow-up results.
MATERIAL AND METHODS: In this study, 740 patients underwent right-lobe living-donor liver transplantation; 56 of them were documented as dual-ductal anastomoses. We analyzed recipient and donor characteristics, surgical procedures, appearance of biliary complications, corresponding interventions, and long-term biliary outcomes.
RESULTS: Cystic and common hepatic ducts were utilized in 56 cases of dual-ductal biliary reconstruction, which we categorized into 2 types: A (78.6%), in which the right anterior intrahepatic duct was anastomosed to the common hepatic duct and the right posterior intrahepatic duct to the cystic duct; and B (21.4%), which was the reverse of A. After a median follow-up period of 46.4 months, 23 patients (41.1%) experienced complications, including biliary leakage and biliary stricture. However, after aggressive intervention (patent biliary anastomosis in most of them), 50 of 56 patients (89.3%) had patent biliary anastomosis and restored normal liver function at the end of follow-up. A small graft (graft-to-recipient weight ratio <0.9%) was the only predictor of biliary complications after multivariate analysis.
CONCLUSIONS: Dual-ductal biliary reconstruction in adult right-lobe living-donor liver transplantation is challenging but feasible. Our findings support the use of the cystic duct for reconstruction in selected patients. Good long-term results can be achieved with adequate management of patients with biliary complications.
Keywords: Biliary Tract, Cystic Duct, Living Donors, Adult, Anastomosis, Surgical, Bile Ducts, Hepatic Duct, Common, Humans, Liver, Liver Transplantation
Background
Duct-to-duct anastomosis during bile duct reconstruction in living-donor liver transplantation (LDLT) was first introduced by Wachs and has gained popularity over the years [1–3]. Compared with Roux-en-Y hepaticojejunostomy, duct-to-duct anastomosis preserves the anatomy, especially the sphincter of Oddi, which can prevent ascending cholangitis by acting as a barrier to reduce enteric reflux. Preservation of the natural physiological bilioenteric route also makes endoscopic evaluation and management of biliary complications feasible. Furthermore, biliary reconstruction is less time-consuming than hepaticojejunostomy. It also does not require the creation of intestinal openings, thereby reducing the risk of intra-abdominal contamination [4]. However, anatomic variants of the biliary tract are common (24% to 44.2%) [5–9]. Atypical biliary anatomy of the graft in right-lobe LDLT leads to multiple bile duct anastomoses, with an incidence of up to 35% [10]. For grafts with 2 or more separated bile duct orifices, which are not rare, an alternative approach of reconstruction should be considered. If 2 orifices in the right hepatic duct are close to each other, a single anastomosis might be possible with or without ductoplasty. In contrast, double anastomoses are mandatory when the orifices are separated by some distance. The cystic duct could be an option provided it is in a healthy condition. Previous studies presented a limited number of cases utilizing cystic ducts for dual biliary reconstruction [4,11,12]. Here, we present what is, to the best of our knowledge, the largest case series of double ductal anastomosis using the cystic duct and common hepatic duct (CHD) without a biliary stent.
Material and Methods
PATIENTS AND DATA COLLECTION:
We retrospectively reviewed a total of 740 patients who underwent right-lobe LDLT at Chang-Gung Memorial Hospital, Linkou Medical Center, Taiwan, between January 2004 and December 2018. After excluding pediatric cases and patients with intraoperative biliary stents, we finally included 56 patients who underwent dual duct-to-duct anastomosis utilizing the recipient’s cystic duct and CHD. Clinical data, including donor and recipient demographics, indications for LDLT, surgical procedural details, subsequent biliary outcome, biliary complication-free time, associated intervention to treat biliary complications, and survival outcomes, were studied. Cold ischemia time is defined as the time between putting the donor liver into cold storage solution and removing the organ from this solution. Warm ischemia time represents the time the graft liver remains at body temperature after removal from cold storage solution but before its blood supply has been restored. The time for dual duct-to-duct reconstruction is referred to as the period between the beginning and end of biliary anastomoses.
The study was approved by the Institutional Review Board of Chang-Gung Memorial Hospital (approval no. 202001325B0). The requirement for informed consent was waived because of the retrospective nature of the study.
DEFINITION OF BILIARY COMPLICATION AND MANAGEMENT PRINCIPLE:
Biliary complications, including biliary leakage and stricture, were specifically and thoroughly reviewed from the medical and radiological records of each recipient. Biliary leakage was defined as in a previous study [13]: drained fluid with increased bilirubin on or after postoperative day 3 or radiologic evidence of biloma and the need for intervention or relaparotomy due to bile-associated peritonitis. A biliary stricture was suspected when there was an increase in postoperative liver enzyme and bilirubin concentrations and biliary tree dilation noted on sonography or computed tomography during follow-up; it was diagnosed by identifying an abnormal narrowing or discontinuity near the previous anastomotic site using either endoscopic retrograde cholangiography (ERC) or percutaneous transhepatic cholangiography (PTC). To manage biliary strictures, we performed endoscopic balloon dilatation of the stricture site during the first ERC, and varying numbers of endoscopic retrograde biliary drainage (ERBD) stents were inserted as needed to maintain an adequate diameter after expansion. In addition, PTC and PTC drainage (PTCD) was reserved for difficult cases in which ERC failed, such as complete stricture or failed biliary cannulation. Further Rendezvous procedures could be considered to convert external drainage, PTCD, to internal drainage, ERBD [14]. The durations between transplantation, intervention, and treatment outcomes were reviewed.
PREOPERATIVE MAGNETIC RESONANCE CHOLANGIOPANCREATOGRAPHY FOR DONORS:
To identify biliary abnormalities in living liver donors, we routinely performed preoperative magnetic resonance cholangiopancreatography (MRCP) for biliary imaging. The arrangement for MRCP was determined on preoperative evaluation by a group of radiologists and transplant surgeons who held meetings especially centered on the anatomic aspects of surgery. MRCP-displayed biliary anatomy of the donor was identified and classified [15] (Supplementary Figure 1). The most common classification was type 1 with normal variation. Triple confluence of the right anterior and posterior intrahepatic ducts (IHDs) and left IHD were classified as type 2. Right anterior and posterior IHDs each draining into the left IHD were referred to as types 3a and 3b, respectively. In type 4a, the right anterior IHD drains into the CHD, and in type 4b, the right posterior IHD drains into the CHD.
SURGICAL TECHNIQUE:
We started hilar dissection and mobilization of the right lobe of the liver for donor hepatectomy. Caution was taken to avoid over-dissection where the right hepatic artery comes close to the bile duct. This was recommended to prevent devascularization of the donor bile duct and subsequent ischemic strictures. A Cavitron ultrasonic aspirator was preferred for hepatic parenchymal transection. If an anomaly existed or an unexpected situation occurred, donor and recipient surgeons discussed whether modification of the surgical plan was necessary. We did not routinely perform intraoperative cholangiography before cutting the right IHDs from the biliary tree in cases of donor hepatectomy. For recipient hepatectomy, a piggyback technique with preservation of the retrohepatic vena cava was our surgical convention [16]. In addition, we did not routinely use pump-driven venous-venous bypass or portal-caval decompression procedures. Furthermore, we focused on keeping the blood supply to the bile duct safe from damage (by minimally dissecting between the right hepatic artery and bile duct) to ensure a direct duct-to-duct biliary anastomosis. One of the many precautions we took was to ensure an unobstructed venous outflow by using the recipient’s right hepatic vein for a proper and natural position. A graft of middle hepatic vein reconstruction using a cryopreserved cadaveric iliac vein graft was considered when necessary. Adequate shortening could be essential to eliminate redundancy between the anastomosed hepatic veins. An end-to-end anastomosis between the donor’s right portal vein and the recipient’s portal trunk was performed, followed by arterial reconstruction through microsurgery by a specialist plastic surgeon. An intraoperative flowmetry test after anastomosis was required to ensure patency of the vascular anastomosis.
In this study, all enrolled patients had dual duct-to-duct anastomoses without a biliary stent. We confirmed the adequacy of blood supply by cutting the edge of the cystic duct before proceeding, we also paid particular attention to accurate coaptation, gentle duct handling, avoidance of crushing or mechanical trauma of any form, avoidance of using ligatures for hemostasis near bile ducts, avoidance of thermal injury by electrocautery, shortening ducts if required, tensionless anastomosis, moderate dilatation of the cyst duct using metallic probes, loupe magnification, and avoiding the strangulation effect of excessive knot tying. Biliary anastomosis was performed in the following steps (Figure 1): first, polydioxanone 6-0 interrupted sutures were used through ductal edges on both sides, starting from the least accessible part, usually at the medial side with limited space. Second, a couple of stitches were created in the form of long stay sutures (both remained untied), then the knot in the first stitch was secured (this was easily done by moving the second stitch in the opposite direction). We proceeded laterally and posteriorly until the posterior part was completed. We performed superior or medial side anastomosis first, followed by inferior or lateral side anastomosis. Finally, the anterior part of the ductal anastomosis was completed by tying knots at 0.8 to 1.0-mm intervals, which were sutured into the edges of ducts but remained untied until the end for a clearer view. All knots were kept outside the anastomosis after the procedure. In our technique, no stents or external drainage tubes were used.
In this study, reconstruction of the biliary system using the cystic duct and CHD of the recipient were classified into 2 types. Type A was defined as the right anterior IHD anastomosed to the CHD and the right posterior IHD to the cystic duct (Figure 2). Type B was defined as the right posterior IHD anastomosed to the CHD and the right anterior IHD to the cystic duct (Figure 3). The choice of the type of reconstruction was based on the exact axis of the graft, appropriate length and tension of biliary reconstruction, and free space between the hepatic artery and bile ducts. Finally, 3 surgical Jackson-Pratt drains were left in the left subphrenic, right subphrenic, and subhepatic regions to allow subsequent monitoring.
STATISTICAL ANALYSIS:
Median values with ranges and numbers (percentages) were used for continuous and categorical variables, respectively. Kaplan-Meier analysis was used to assess biliary complication-free survival and overall survival, and differences between subgroups were analyzed using the log-rank test. Risk factors recognized in univariate analysis with a
Results
PATIENT CHARACTERISTICS:
Patient characteristics are shown in Table 1. The median age of the recipients was 53.9 (range, 32.4–70.2) years, and the median MELD score was 15.5 (range, 7.0–40.0). The median graft-to-recipient weight ratio (GRWR) was 0.90, ranging from 0.51 to 1.49. In our study, 24 patients (42.9%) had hepatocellular carcinoma. A strong association with hepatitis was observed, and there were 31 (55.4%) and 14 (25%) patients with hepatitis B and C infections, respectively. Notably, 13 (23.2%) patients received an ABO-incompatible liver transplant. The average warm and cold ischemia times were 36.6±8.5 and 63.0±44.1 min, respectively. The median time spent on dual-ductal biliary reconstruction was 71.0 min (range, 22.0 to 150.0 min). Types A and B biliary reconstruction were performed in 44 and 12 patients, respectively. The median follow-up time was 46.4 (range, 0.1 to 178.0) months, and the cumulative 1-year and 5-year overall survival rates were 85.6% and 68.8%, respectively. In this study, there were no cases of primary non-function or vascular complications. During the follow-up period, 6 patients had acute rejection, resulting in 1 death. By the end of follow-up, 16 patients had died. All mortalities occurred due to infection-related severe sepsis, except in 4 patients: 1 died of acute-onset subdural hematoma, 1 of acute rejection, and 2 of post-transplant viral hepatitis C-related graft failure.
BILIARY COMPLICATIONS:
Subsequent biliary complications after right-lobe LDLT with dual biliary anastomosis developed in 23 (41.1%) patients. The incidence of biliary complications declined with the maturation of the technique and accumulation of experience: 17 of 35 patients (48.6%) developed biliary complications in the first 10-year period, and 6 of 21 patients (28.6%) in the second period (2015–2018). Overall, 8 (14.3%) patients had biliary leakage and 19 (33.9%) had biliary strictures in this study. The median time between transplant and biliary complications was 12.4 months (range, 0.1–165.7 months). Univariate analysis for perioperative risk of biliary complications is shown in Table 2, including MELD scores of >20 (P=0.044, odds ratio [OR] 3.462, 95% confidence interval [CI] 1.03–11.60) and GRWR <0.9% (P=0.017, OR 4, 95% CI 1.28–12.47). Independent risk identified in multivariate analysis revealed that a GRWR of <0.9% (P=0.042, OR 3.503, 95% CI 1.05–11.73) was significantly correlated with biliary complication occurrence. A lower GRWR indicated an inferior biliary complication-free survival (P=0.038; Figure 4A). However, the presence or absence of biliary complications did not influence overall survival after transplantation (P=0.360; Figure 4B).
MANAGEMENT FOR BILIARY COMPLICATIONS:
The management of biliary complications is shown in detail in Figure 5. With regards to the management of biliary leakage (n=8), 4 patients needed additional echography- or computer tomographic-guided percutaneous pigtail drainage of bilomas, while 3 of them had the drainage successfully performed without subsequent anastomosis stricture. The other half with biliary leakage (4 of 8, 50%) subsequently developed biliary stricture and needed further ERBD intervention. Among the 19 patients who presented with biliary stricture, 18 received ERC with or without ERBD, and the first ERC attempts failed in 8 of them, and additional PTCD was required. Five of 8 patients with PTCD eventually had ERBD successfully inserted as internal drainage by the Rendezvous procedure and had the PTCD external drainage removed. Most patients (14 of 18, 77.8%) became stricture-free after ERBD with or without PTCD as a bridge. The ERBD intervention failed in 1 patient who experienced a biliary complication due to a non-anastomosis stricture. The patient eventually died of graft failure and could not undergo re-transplantation. Because cholangiography showed multiple biliary stenoses beyond the anastomosis, we suspected that this patient had persistent ischemic cholangitis due to chronic antibody-mediated rejection caused by ABO blood type incompatibility.
The median duration of biliary complications was 10.4 months (range, 0.2–31.4 months). At the time of follow-up, 50 (89.3%) patients were free from biliary complications. The other 6 patients were unable to recover from biliary complications and eventually died during follow-up. The leading cause of death was advanced sepsis (n=5), and we also had a case of liver failure due to hepatitis C reactivation. Notably, 4 of these 6 (66.7%) patients had LDLT under ABO incompatibility circumstances; however, ABO blood type incompatibility was not documented as a significant perioperative risk factor for biliary complications in this 56-patient case series (
MRCP FOR DONOR EVALUATION:
In this study, grafts with 2 bile duct orifices were obtained from donors with biliary anatomy type 4b (n=37, 66.1%), type 4a (n=6, 10.7%), types 1 and 2 (both n=5, 8.9%), and type 3b (n=3, 5.4%). There was no significant association between the different biliary anatomic types and the incidence of biliary complications (
Discussion
Biliary complications remain one of the most common morbidities following liver transplantation. The incidence of biliary complications ranges from 5.3% to 43% in liver transplant recipients [3,17–20]. Great diversities were prevalent in different eras, and the reported incidence of biliary stricture was up to 42.8% with a biliary leakage rate of 26.7% at an earlier period [21]. Furthermore, the decreasing occurrence of biliary leakage and stricture has been reported in later studies [20,22]. Biliary complications are more likely to occur in LDLT than in deceased-donor liver transplantation [19]. Approximately two-thirds of all biliary complications occur early, within the first 3 months of liver transplantation. The exact cause of biliary complications remains to be elucidated, although many reports note different factors, such as technical differences, degree of tissue ischemia, immunological factors, anatomical variations, combined infections, age disparity, ABO incompatibility, cytomegalovirus infection, cross-match, chronic ductopenic rejection, and immunosuppressive drugs [23].
In the present study, 33.9% of patients developed biliary strictures, and 14.3% had bile leaks after right-lobe LDLT. The incidence of overall biliary complications was estimated to be 41.1%, which is higher than that reported in previous studies. Currently, there is no satisfactory explanation for why the presence of multiple bile ducts in the liver allograft tends to lead to the development of biliary complications [24]. Reconstruction of small and multiple bile ducts is technically challenging and gives rise to a greater likelihood of leakage or further stenosis. In addition, it seems that most biliary complications develop early and can be considered to be related to surgical techniques rather than to arterial insufficiency.
Surgical details regarding biliary reconstruction have been refined over time and include minimal dissection to preserve adequate peri-choledochal fat and blood supply to the bile duct, avoidance of electrocautery-associated thermal injury, prevention of inadvertent suture under loupe magnification using reassurance with probe exploration, and separation of every stitch first at the anterior wall before knotting. The use of a higher-order biliary radical technique for reconstruction can be associated with potentially impaired arterial perfusion, which contributes to biliary complications [25]. We believe that all technical details are of great significance in preventing biliary complications. Our biliary complication rate decreased to 28.6% during the last 4 years compared with in the first decade (48.6%), and this is suggestive of technical maturation. In the present study, only 8 of 56 patients (14.2%) developed biliary leakage and half of them spontaneously healed without subsequent biliary stricture, which suggests good preservation of the peribiliary blood supply and reliable surgical technique in biliary anastomosis. In this study, GRWR of <0.9% was the only independent risk factor for predicting the occurrence of biliary complications in the multivariate analysis. GRWR of <0.9% indicates a small graft in the right-lobe LDLT. The axis of the biliary tree between the IHDs of the graft and native common bile duct (CBD) of the recipient will change after transplantation during liver regeneration. A smaller graft might cause a sharper angle of the axis between the IHD of the graft and the native CBD of the recipient (as shown in Figures 2B and 3B) compared with the larger graft, especially in the case of dual biliary anastomosis (2 weak points in dual anastomosis have a higher chance of developing biliary stricture). The sharp angle of biliary anastomosis caused by liver regeneration is a possible reason for delayed biliary stenosis. In addition, there is no difference between both types of biliary reconstruction in terms of the incidence of biliary complications.
The management of biliary complications after living-donor liver transplantation requires multidisciplinary approaches involving transplant surgeons, gastroenterologists, and interventional radiologists. Endoscopic balloon dilation, internal stenting, and percutaneous procedures are effective. Hisatue et al reported that 13 of 14 patients with biliary stricture after duct-to-duct anastomosis were successfully treated with internal stenting [26]. In the present study, 14 of 18 (77.8%) patients with biliary strictures after ERBD were eventually free from biliary complications and stent placement at the end of the follow-up period. Although a relatively higher incidence of biliary complications was observed after utilizing the cystic duct for dual biliary duct reconstruction, most cases after adequate management achieved a complication- and stent-free status at the end of follow-up. Preserving patent biliary tracts from leaks or strictures is essential for post-transplant care, and a natural anatomical route enables an endoscopic approach. Therefore, it is of great advantage to use the cystic duct for biliary reconstruction in grafts with separate biliary ducts. However, it was not a significant factor in determining the 5-year survival rate of patients with or without biliary complications.
Atypical biliary anatomy variations in donors increase the complexity of biliary reconstruction. The transection plane of the right hepatic duct is designed based on concerns regarding the integrity of the left hepatic duct and of yielding a single orifice of the right hepatic duct. Preoperative donor MRCP assists in donor selection and surgical planning. The presence of biliary anatomical variants, other than type 1, increases biliary complications [27]. In all our dual-duct biliary reconstructions, the most common biliary anatomy type was 4b (66.1%), in which the right posterior HD drains into the CHD. However, only 6.5% of the general population has biliary anatomy type 4b [5]. It is reasonable to have a discrepant result from the most general biliary anatomy that 2 separate bile duct orifices, which are supposed to be present through planning the parenchymal transection line. Surprisingly, types 1 and 2 accounted for 8.9% of the dual ducts. We believe that the greater the narrowing of the primary to secondary length of the right IHD displays, the lesser the chance of transect deviation separating bile orifices. The sharp angle between the confluence of the right and left HDs can play a role [28]. Conversely, because we did not perform intraoperative cholangiography, the line of biliary-tree cutting could have been erroneous, which could explain why some patients with normal biliary anatomy (types 1 and 2, both n=5) had grafts with 2 biliary orifices (the line of biliary-tree cutting did not pass through the conjunction point but was further to the right side). In other words, when preoperative MRCP in the donor shows the possibility of dual bile orifices, utilization of the cystic duct should be adopted during surgical planning, and extra attention should be paid to preserving the cystic duct of healthy recipients.
The use of an external drainage tube or stent across the anastomosis remains controversial. In previous studies, Asonuma [12] reported 5 cases of dual-ductal reconstruction, using 2 stents for each, in which the cystic duct was used for 1 of 2 anastomoses. All stents were removed between 17 and 37 weeks. Kasahara [22] reported leakage in only 6% and biliary stricture in 17.3% of 266 patients who underwent biliary reconstruction using an external stent. However, excellent results have been demonstrated after duct-to-duct anastomosis in right-lobe living-donor liver transplantation without biliary drainage [29]. One study reported that biliary stenting predisposes to biliary complications [30]. In our center, we do not routinely use external drainage tubes or stents across anastomosis for the following reasons: signs of sepsis develop in some patients when tubes are removed, which could be caused by infection related to foreign bodies or lack of an anti-reflux barrier; the need to keep the tubes for a long time, limiting patient mobility; and the tubes can also be accidentally dislodged and cause damages. In selected cases, it not only shortens anastomosis time but also physiologically fits natural biliary anatomy to decrease reflux infection compared with Roux-en-Y hepaticojejunostomy. In situations involving 2 separated intrahepatic ducts, utilizing the cystic and CHD in dual-ductal biliary reconstruction is a rational choice. However, multiple anastomoses of small-caliber ducts, identification of possible biliary variations, and precise execution of the surgical plan are challenges that transplant surgeons face.
Conclusions
In conclusion, utilization of the cystic duct in dual-ductal reconstruction is technically feasible in selected patients. Endoscopic or interventional percutaneous procedures are effective in dealing with biliary complications, and good biliary and survival outcomes can be achieved with adequate management.
Figures
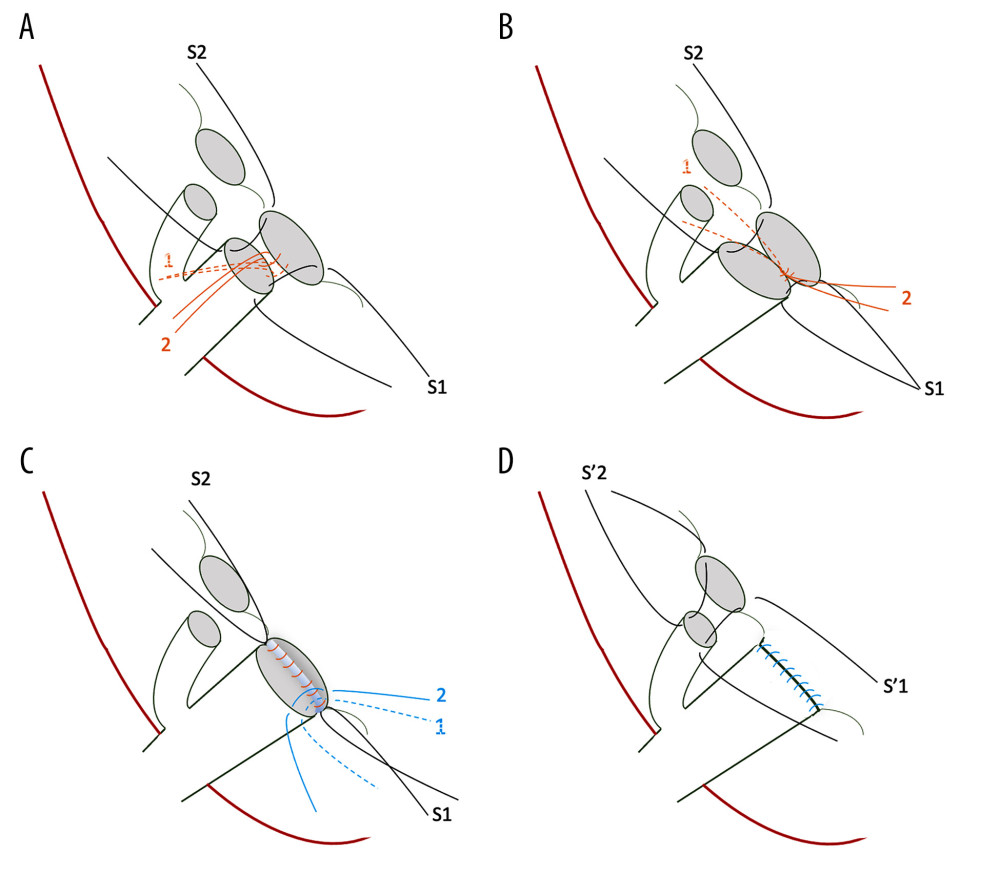 Figure 1. Steps of the dual biliary duct reconstruction, demonstrated as the anastomosis of the right posterior intrahepatic duct (IHD) to the common hepatic duct (CHD)/anastomosis of the right anterior IHD to the cystic duct: (A) S1 and S2 represent 6-0 interrupted sutures through ductal edges on both sides, serving as long stay sutures. Reconstruction is initiated at the posterior wall of the bile duct at the medial side, as shown by the orange lines. (B) The second stitch (orange line 2) is moved in the opposite direction to create a working space and then the knot is secured in the first stitch (orange line 1). Proceed laterally and posteriorly stitch by stitch until the posterior part is completed. Care should be taken to ensure to tie the knots outside the bile duct lumen. (C) After the posterior wall is completed, start the anterior wall from the superior or medial side, and finish by tying knots, which are sutured into the edges of ducts but remain untied until the end for a clearer view. (D) Start the right anterior IHD-cystic duct anastomosis using stay long sutures (S1 and S2) and follow the aforementioned steps.
Figure 1. Steps of the dual biliary duct reconstruction, demonstrated as the anastomosis of the right posterior intrahepatic duct (IHD) to the common hepatic duct (CHD)/anastomosis of the right anterior IHD to the cystic duct: (A) S1 and S2 represent 6-0 interrupted sutures through ductal edges on both sides, serving as long stay sutures. Reconstruction is initiated at the posterior wall of the bile duct at the medial side, as shown by the orange lines. (B) The second stitch (orange line 2) is moved in the opposite direction to create a working space and then the knot is secured in the first stitch (orange line 1). Proceed laterally and posteriorly stitch by stitch until the posterior part is completed. Care should be taken to ensure to tie the knots outside the bile duct lumen. (C) After the posterior wall is completed, start the anterior wall from the superior or medial side, and finish by tying knots, which are sutured into the edges of ducts but remain untied until the end for a clearer view. (D) Start the right anterior IHD-cystic duct anastomosis using stay long sutures (S1 and S2) and follow the aforementioned steps. 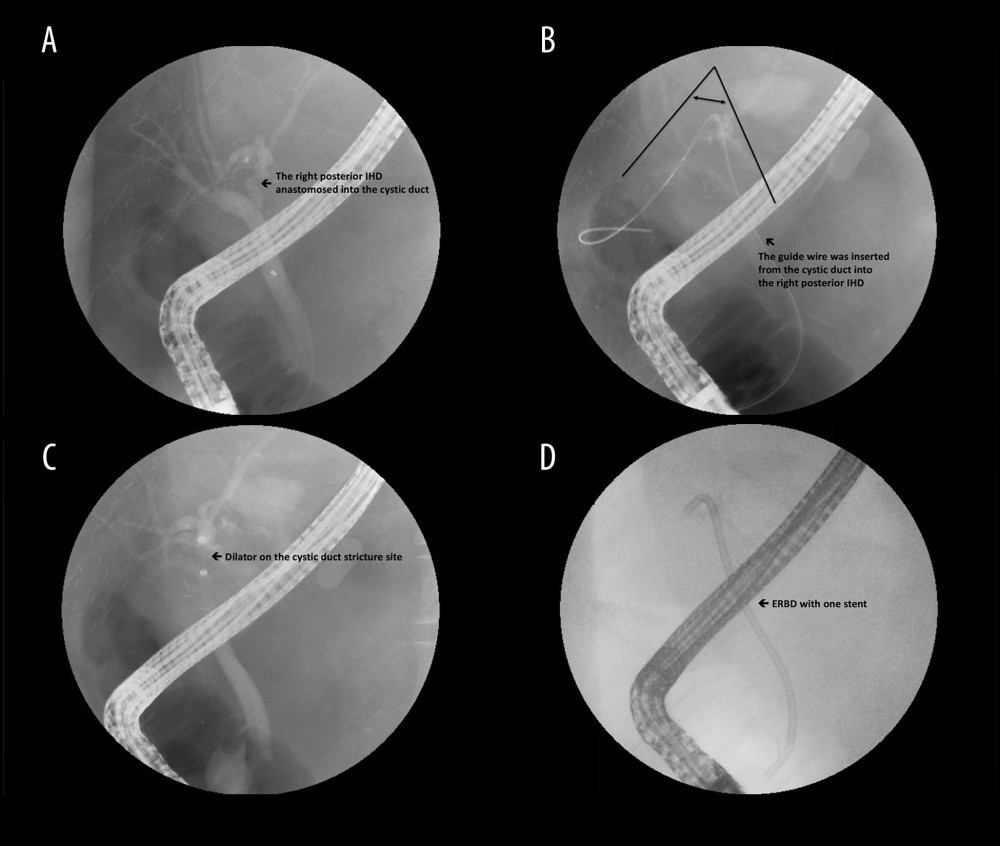 Figure 2. Steps of type A reconstruction: (A) right anterior intrahepatic duct (IHD) is anastomosed to the common hepatic duct (CHD) and right posterior IHD to the cystic duct; (B) guide wire is inserted through the cystic duct into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) dilator is placed in the anastomosed cystic duct stricture; (D) endoscopic retrograde biliary drainage with 1 stent for stricture of cystic duct anastomosis.
Figure 2. Steps of type A reconstruction: (A) right anterior intrahepatic duct (IHD) is anastomosed to the common hepatic duct (CHD) and right posterior IHD to the cystic duct; (B) guide wire is inserted through the cystic duct into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) dilator is placed in the anastomosed cystic duct stricture; (D) endoscopic retrograde biliary drainage with 1 stent for stricture of cystic duct anastomosis. 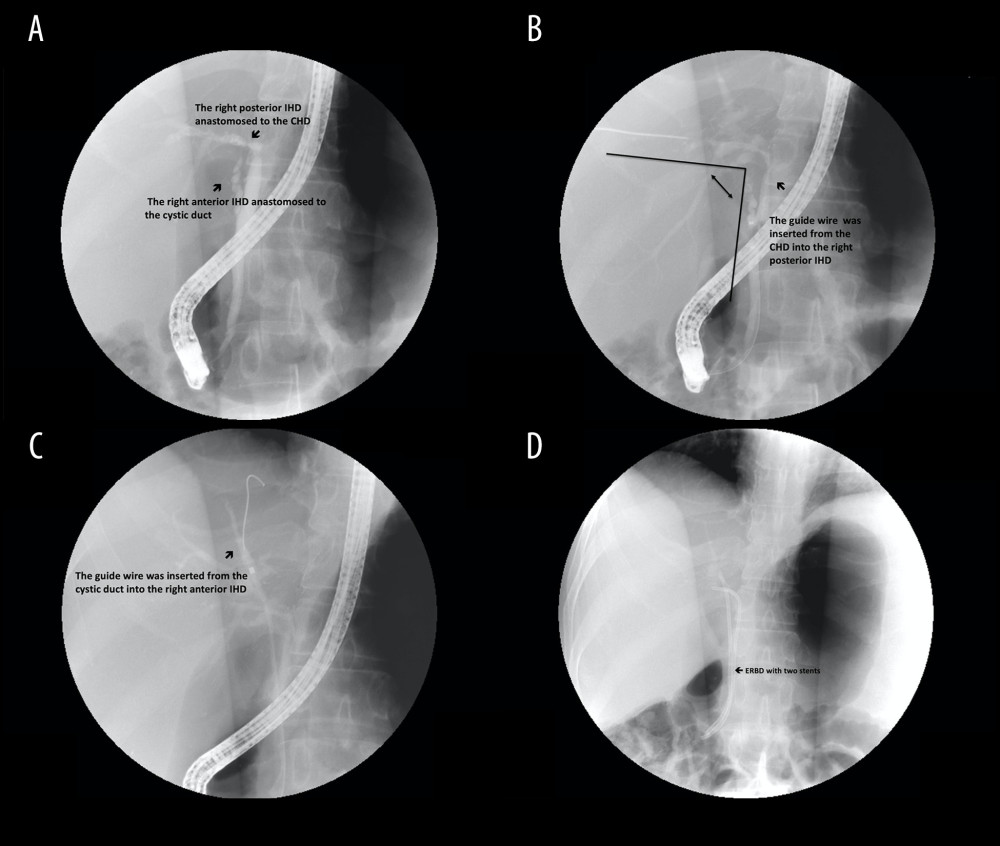 Figure 3. Steps of type B reconstruction: (A) right posterior intrahepatic duct (IHD) anastomosed to the common hepatic duct (CHD), and right anterior IHD anastomosed to cystic duct; (B) guide wire is inserted through the CHD into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) guide wire is inserted through the cystic duct into the right anterior IHD; (D) endoscopic retrograde biliary drainage with 2 stents in the strictures of dual anastomosis.
Figure 3. Steps of type B reconstruction: (A) right posterior intrahepatic duct (IHD) anastomosed to the common hepatic duct (CHD), and right anterior IHD anastomosed to cystic duct; (B) guide wire is inserted through the CHD into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) guide wire is inserted through the cystic duct into the right anterior IHD; (D) endoscopic retrograde biliary drainage with 2 stents in the strictures of dual anastomosis. 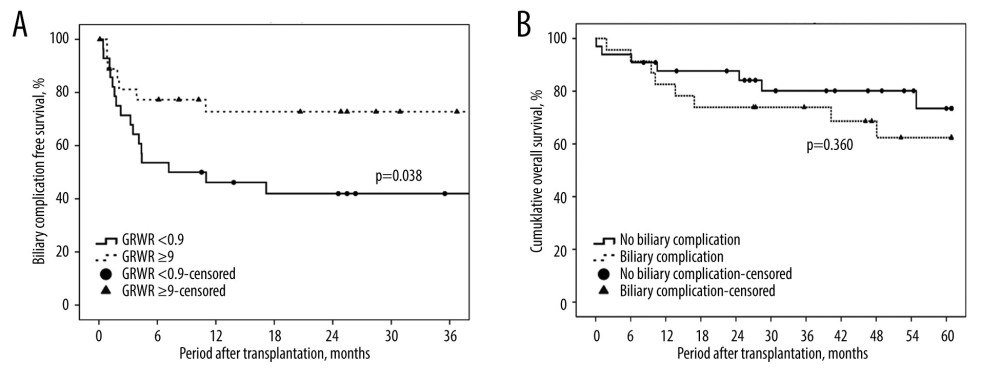 Figure 4. (A) Kaplan-Meier method for biliary complication-free survival according to different graft-to-recipient weight ratios (GRWR). The biliary complication-free survival rate was worse among the GRWR <0.9 group. This figure demonstrates only the 3-year follow-up because no biliary complication events were noted after 3 years. (B) Kaplan-Meier method for overall survival and biliary complications. No significant difference was found between groups with and without biliary complications.
Figure 4. (A) Kaplan-Meier method for biliary complication-free survival according to different graft-to-recipient weight ratios (GRWR). The biliary complication-free survival rate was worse among the GRWR <0.9 group. This figure demonstrates only the 3-year follow-up because no biliary complication events were noted after 3 years. (B) Kaplan-Meier method for overall survival and biliary complications. No significant difference was found between groups with and without biliary complications. 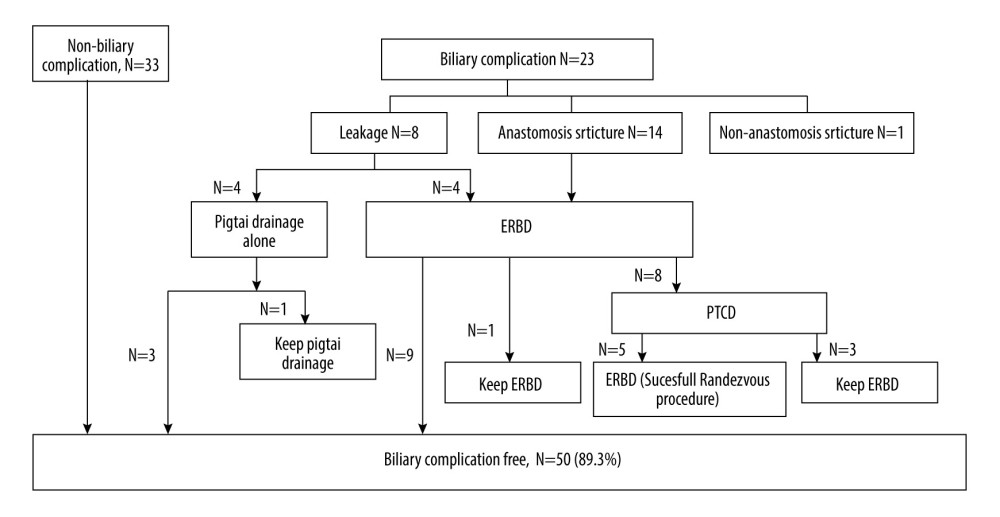 Figure 5. Flow chart of management of biliary complications. ERBD – endoscopic retrograde biliary drainage; PTCD – percutaneous transhepatic cholangiography and drainage.
Figure 5. Flow chart of management of biliary complications. ERBD – endoscopic retrograde biliary drainage; PTCD – percutaneous transhepatic cholangiography and drainage. References
1. Wachs ME, Bak TE, Karrer FM, Adult living donor liver transplantation using a right hepatic lobe: Transplantation, 1998; 66; 1313-16
2. Ishiko T, Egawa H, Kasahara M, Duct-to-duct biliary reconstruction in living donor liver transplantation utilizing right lobe graft: Ann Surg, 2002; 236; 235-40
3. Hong SY, Hu XG, Lee HY, Longterm analysis of biliary complications after duct-to-duct biliary reconstruction in living donor liver transplantations: Liver Transpl, 2018; 24; 1050-61
4. Suh KS, Choi SH, Yi NJ, Biliary reconstruction using the cystic duct in right lobe living donor liver transplantation: J Am Coll Surg, 2004; 199; 661-64
5. Varotti G, Gondolesi GE, Goldman J, Anatomic variations in right liver living donors: J Am Coll Surg, 2004; 198(4); 577-82
6. Yaprak O, Demirbas T, Duran C, Living donor liver hilar variations: Surgical approaches and implications: Hepatobiliary Pancreat Dis Int, 2011; 10(5); 474-79
7. Puente SG, Bannura GC, Radiological anatomy of the biliary tract: Variations and congenital abnormalities: World J Surg, 1983; 7(2); 271-76
8. Mariolis-Sapsakos T, Kalles V, Papatheodorou K, Anatomic variations of the right hepatic duct: Results and surgical implications from a cadaveric study: Anat Res Int, 2012; 2012; 838179
9. Choi JW, Kim TK, Kim KW, Anatomic variation in intrahepatic bile ducts: An analysis of intraoperative cholangiograms in 300 consecutive donors for living donor liver transplantation: Korean J Radiol, 2003; 4(2); 85-90
10. Nakamura T, Tanaka K, Kiuchi T, Anatomical variations and surgical strategies in right lobe living donor liver transplantation: lessons from 120 cases: Transplantation, 2002; 73; 1896-903
11. Kadry Z, Cintorino D, Foglieni CS, Fung J, The pitfall of the cystic duct biliary anastomosis in right lobe living donor liver transplantation: Liver Transpl, 2004; 10; 1549-50
12. Asonuma K, Okajima H, Ueno M, Feasibility of using the cystic duct for biliary reconstruction in right-lobe living donor liver transplantation: Liver Transpl, 2005; 11; 1431-34
13. Koch M, Garden OJ, Padbury R, Bile leakage after hepatobiliary and pancreatic surgery: A definition and grading of severity by the International Study Group of Liver Surgery: Surgery, 2011; 149; 680-88
14. Robertson DA, Ayres R, Hacking CN, Experience with a combined percutaneous and endoscopic approach to stent insertion in malignant obstructive jaundice: Lancet, 1987; 2; 1449-52
15. Busuttil R, Klintmalm G: Transplantation of the liver, 2005; 681, Philadelphia, Elsevier Saunders
16. Jovine E, Mazziotti A, Grazi GL, Piggy-back versus conventional technique in liver transplantation: Report of a randomized trial: Transpl Int, 1997; 10; 109-12
17. Chok KS, Lo CM, Biliary complications in right lobe living donor liver transplantation: Hepatol Int, 2016; 10; 553-58
18. Senter-Zapata M, Khan AS, Subramanian T, Patient and graft survival: Biliary complications after liver transplantation: J Am Coll Surg, 2018; 226; 484-94
19. Freise CE, Gillespie BW, Koffron AJ, Recipient morbidity after living and deceased donor liver transplantation: findings from the A2ALL Retrospective Cohort Study: Am J Transplant, 2008; 8; 2569-79
20. Wang SF, Huang ZY, Chen XP, Biliary complications after living donor liver transplantation: Liver Transpl, 2011; 17; 1127-36
21. Greif F, Bronsther OL, Van Thiel DH, The incidence, timing, and management of biliary tract complications after orthotopic liver transplantation: Ann Surg, 1994; 219; 40-45
22. Kasahara M, Egawa H, Takada Y, Biliary reconstruction in right lobe living-donor liver transplantation: Comparison of different techniques in 321 recipients: Ann surg, 2006; 243; 559-66
23. Neuhaus P, Pascher A, Technical problems: Transplantation of the liver, 2005; 929-47, Philadelphia, Elsevier Saunders
24. Salvalaggio PR, Whitington PF, Alonso EM, Superina RA, Presence of multiple bile ducts in the liver graft increases the incidence of biliary complications in pediatric liver transplantation: Liver Transpl, 2005; 11; 161-66
25. Baker TB, Zimmerman MA, Goodrich NP, Biliary reconstructive techniques and associated anatomic variants in adult living donor liver transplantations: The adult-to-adult living donor liver transplantation cohort study experience: Liver Transpl, 2017; 23; 1519-30
26. Hisatsune H, Yazumi S, Egawa H, Endoscopic management of biliary strictures after duct-to-duct biliary reconstruction in right-lobe living-donor liver transplantation: Transplantation, 2003; 76; 810-15
27. Kashyap R, Bozorgzadeh A, Abt P, Stratifying risk of biliary complications in adult living donor liver transplantation by magnetic resonance cholangiography: Transplantation, 2008; 85; 1569-72
28. Ragab A, Lopez-Soler RI, Oto A, Testa G, Correlation between 3D-MRCP and intra-operative findings in right liver donors: Hepatobiliary Surg Nutr, 2013; 2; 7-13
29. Liu CL, Lo CM, Chan SC, Fan ST, Safety of duct-to-duct biliary reconstruction in right-lobe live-donor liver transplantation without biliary drainage: Transplantation, 2004; 77; 726-32
30. Qian YB, Liu CL, Lo CM, Fan ST, Risk factors for biliary complications after liver transplantation: Arch Surg, 2004; 139; 1101-5
Figures
 Figure 1. Steps of the dual biliary duct reconstruction, demonstrated as the anastomosis of the right posterior intrahepatic duct (IHD) to the common hepatic duct (CHD)/anastomosis of the right anterior IHD to the cystic duct: (A) S1 and S2 represent 6-0 interrupted sutures through ductal edges on both sides, serving as long stay sutures. Reconstruction is initiated at the posterior wall of the bile duct at the medial side, as shown by the orange lines. (B) The second stitch (orange line 2) is moved in the opposite direction to create a working space and then the knot is secured in the first stitch (orange line 1). Proceed laterally and posteriorly stitch by stitch until the posterior part is completed. Care should be taken to ensure to tie the knots outside the bile duct lumen. (C) After the posterior wall is completed, start the anterior wall from the superior or medial side, and finish by tying knots, which are sutured into the edges of ducts but remain untied until the end for a clearer view. (D) Start the right anterior IHD-cystic duct anastomosis using stay long sutures (S1 and S2) and follow the aforementioned steps.
Figure 1. Steps of the dual biliary duct reconstruction, demonstrated as the anastomosis of the right posterior intrahepatic duct (IHD) to the common hepatic duct (CHD)/anastomosis of the right anterior IHD to the cystic duct: (A) S1 and S2 represent 6-0 interrupted sutures through ductal edges on both sides, serving as long stay sutures. Reconstruction is initiated at the posterior wall of the bile duct at the medial side, as shown by the orange lines. (B) The second stitch (orange line 2) is moved in the opposite direction to create a working space and then the knot is secured in the first stitch (orange line 1). Proceed laterally and posteriorly stitch by stitch until the posterior part is completed. Care should be taken to ensure to tie the knots outside the bile duct lumen. (C) After the posterior wall is completed, start the anterior wall from the superior or medial side, and finish by tying knots, which are sutured into the edges of ducts but remain untied until the end for a clearer view. (D) Start the right anterior IHD-cystic duct anastomosis using stay long sutures (S1 and S2) and follow the aforementioned steps. Figure 2. Steps of type A reconstruction: (A) right anterior intrahepatic duct (IHD) is anastomosed to the common hepatic duct (CHD) and right posterior IHD to the cystic duct; (B) guide wire is inserted through the cystic duct into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) dilator is placed in the anastomosed cystic duct stricture; (D) endoscopic retrograde biliary drainage with 1 stent for stricture of cystic duct anastomosis.
Figure 2. Steps of type A reconstruction: (A) right anterior intrahepatic duct (IHD) is anastomosed to the common hepatic duct (CHD) and right posterior IHD to the cystic duct; (B) guide wire is inserted through the cystic duct into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) dilator is placed in the anastomosed cystic duct stricture; (D) endoscopic retrograde biliary drainage with 1 stent for stricture of cystic duct anastomosis. Figure 3. Steps of type B reconstruction: (A) right posterior intrahepatic duct (IHD) anastomosed to the common hepatic duct (CHD), and right anterior IHD anastomosed to cystic duct; (B) guide wire is inserted through the CHD into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) guide wire is inserted through the cystic duct into the right anterior IHD; (D) endoscopic retrograde biliary drainage with 2 stents in the strictures of dual anastomosis.
Figure 3. Steps of type B reconstruction: (A) right posterior intrahepatic duct (IHD) anastomosed to the common hepatic duct (CHD), and right anterior IHD anastomosed to cystic duct; (B) guide wire is inserted through the CHD into the right posterior IHD, showing the sharp angle of biliary axis between the IHD of the graft and the native CBD of the recipient; (C) guide wire is inserted through the cystic duct into the right anterior IHD; (D) endoscopic retrograde biliary drainage with 2 stents in the strictures of dual anastomosis. Figure 4. (A) Kaplan-Meier method for biliary complication-free survival according to different graft-to-recipient weight ratios (GRWR). The biliary complication-free survival rate was worse among the GRWR <0.9 group. This figure demonstrates only the 3-year follow-up because no biliary complication events were noted after 3 years. (B) Kaplan-Meier method for overall survival and biliary complications. No significant difference was found between groups with and without biliary complications.
Figure 4. (A) Kaplan-Meier method for biliary complication-free survival according to different graft-to-recipient weight ratios (GRWR). The biliary complication-free survival rate was worse among the GRWR <0.9 group. This figure demonstrates only the 3-year follow-up because no biliary complication events were noted after 3 years. (B) Kaplan-Meier method for overall survival and biliary complications. No significant difference was found between groups with and without biliary complications. Figure 5. Flow chart of management of biliary complications. ERBD – endoscopic retrograde biliary drainage; PTCD – percutaneous transhepatic cholangiography and drainage.
Figure 5. Flow chart of management of biliary complications. ERBD – endoscopic retrograde biliary drainage; PTCD – percutaneous transhepatic cholangiography and drainage. Tables
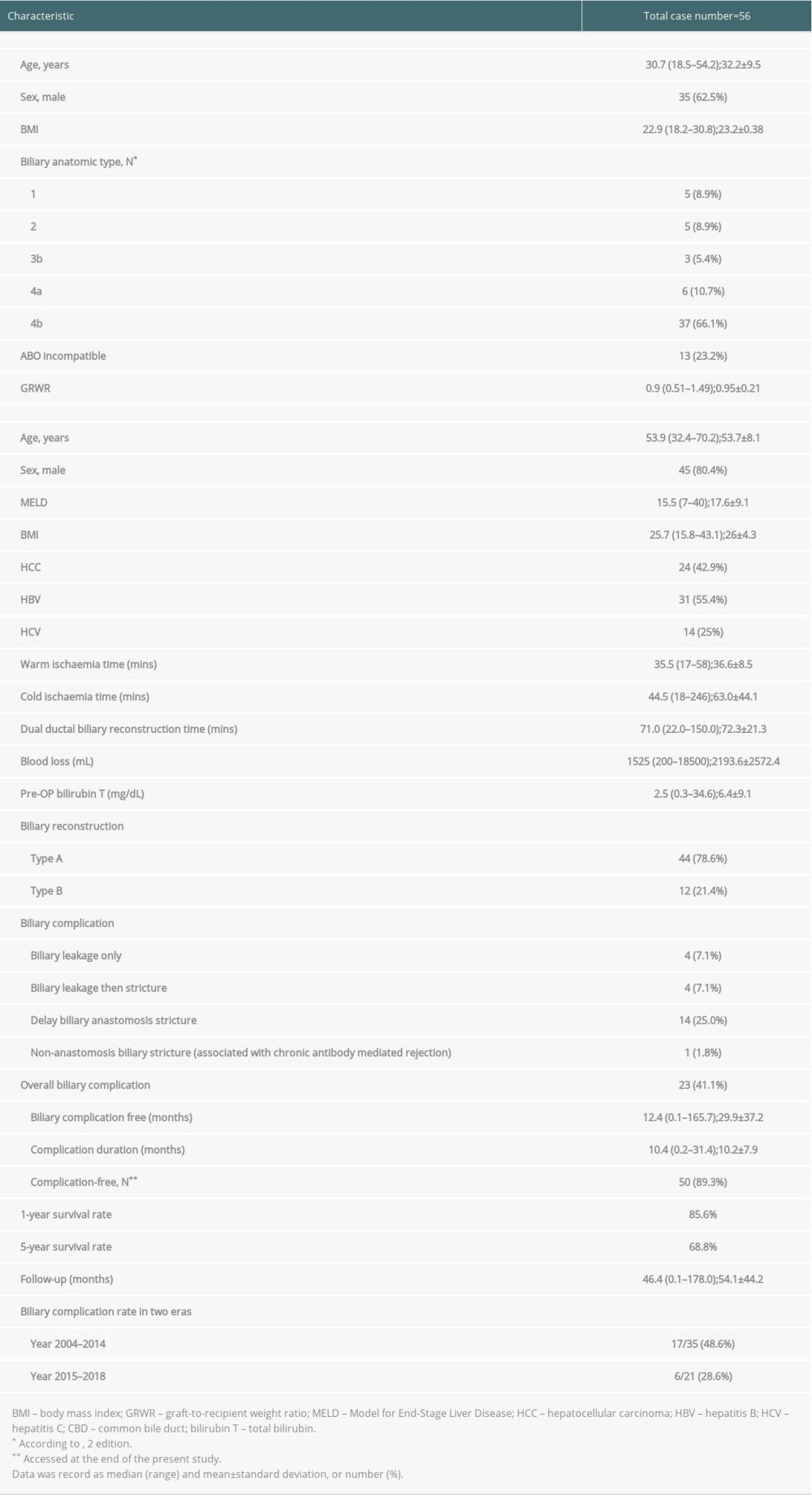 Table 1. Characteristics of enrolled patients after right-lobe living-donor liver transplantation with dual-ductal biliary reconstruction.
Table 1. Characteristics of enrolled patients after right-lobe living-donor liver transplantation with dual-ductal biliary reconstruction.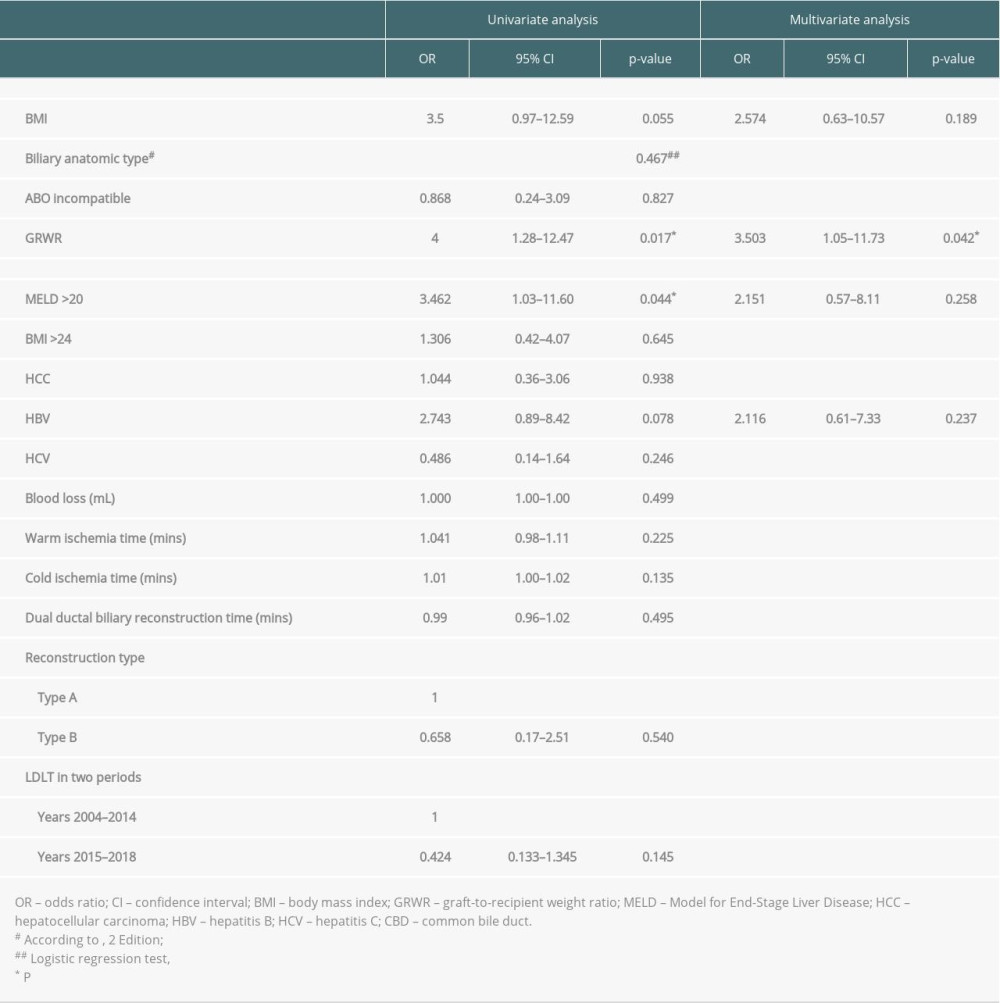 Table 2. Risks of developing biliary complications in the univariate and multivariate analyses.
Table 2. Risks of developing biliary complications in the univariate and multivariate analyses. Table 1. Characteristics of enrolled patients after right-lobe living-donor liver transplantation with dual-ductal biliary reconstruction.
Table 1. Characteristics of enrolled patients after right-lobe living-donor liver transplantation with dual-ductal biliary reconstruction. Table 2. Risks of developing biliary complications in the univariate and multivariate analyses.
Table 2. Risks of developing biliary complications in the univariate and multivariate analyses. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








