09 August 2022: Case Report
Extrapulmonary Manifestations of SARS-CoV-2: A Report of 3 Cases and a Literature Review
Aleksandra Barbachowska1EF, Jolanta Gozdowska1AE*, Dorota Piotrowska-Kownacka2C, Monika Sawicka2D, Dominika Dęborska-Materkowska1A, Ewa Skolimowska1F, Magdalena Durlik1ADOI: 10.12659/AOT.935874
Ann Transplant 2022; 27:e935874
Abstract
BACKGROUND: COVID-19 disease, caused by the SARS-CoV-2 virus, has been one of the greatest challenges in modern medicine. It is mostly known to affect the pulmonary system, leading to pneumonia and acute respiratory distress syndrome, but there is a growing body of evidence of extrapulmonary manifestations of COVID-19 disease.
CASE REPORT: This article presents 3 cases of various extrapulmonary symptoms of COVID-19 disease and a literature review of similar clinical cases. Two patients had a medical history of living-donor kidney transplantation, and 1 patient was a kidney donor. We present symptoms, diagnostic processes, laboratory and imaging results, and treatment approach. Patient 1 was 29-year-old woman with new-onset diabetes mellitus due to SARS-CoV-2, which required temporary insulin treatment. Patient 2 was a 34-year-old man with fever, chronic fatigue, back pain, and abdominal pain. Imagining showed acalculous cholecystitis, epiploic appendagitis of the right colic flexure, and inflammation of pericardial fat pad in the left cardiophrenic angle. Coagulopathy due to COVID-19 was the most probable cause of the described processes. Therapeutic doses of low-molecular-weight heparin were administered. Patient 3 was a 68-year-old male kidney donor who had painless, nodular, reddening lesions on both shins, accompanied by itching on both shins and recurrent fever. The diagnosis of erythema nodosum during COVID-19 was made. After treatment with low-molecular-weight heparin, significant decreases of symptoms were observed.
CONCLUSIONS: We conclude that SARS-CoV-2 infection can have a varied course and can involve other systems and organs. Physicians should be aware of possible extrapulmonary symptoms associated with infection with this virus. Correct diagnosis is a prerequisite for proper treatment and prevention of unexpected complications.
Keywords: COVID-19, SARS-CoV-2, Transplantation, Extrapulmonary Manifestations, Kidney Transplant, Adult, Aged, Female, Heparin, Low-Molecular-Weight, Humans, Male, SARS-CoV-2
Background
The SARS-CoV-2 virus (Severe Acute Respiratory Syndrome Coronavirus 2), the cause of COVID-19 disease (Coronavirus disease 19), has turned out to be one of the greatest challenges in modern medicine. First described in December 2019 in Wuhan (China), it rapidly spread worldwide, leading to the declaration of a pandemic in March 2020 [1]. To date, over 178 million cases have been reported, including 3.8 million deaths [2].
COVID-19 disease spreads mostly via aerosol transmission of respiratory droplets and is known to cause pulmonary diseases such as pneumonia and acute respiratory distress syndrome (ARDS) [3]. There is a growing body of evidence that SARS-CoV-2 infection can also cause extrapulmonary manifestations, disturbing the function of many organs, including kidneys, liver, vascular endothelial cells, and the gastrointestinal tract [3]. This correlates with the expression of cell receptor of angiotensin-converting enzyme 2 (ACE2) in these organs. The attachment of SARS-CoV-2 to the receptor, enable the virus to enter the cell and cause infection [4]. Another substantial process causing by the virus is decreasing the number (down-regulation) of ACE2 receptors. As a consequence, it leads to excessive, out-of-control release of a various of inflammatory cytokines (a cytokine storm) through dysregulation of the renin-angiotensin-aldosterone system (RAAS), attenuation of Mas receptor (ACE2/MasR axis), increase activation of [des-Arg9]-bradykinin, and activation of the complement system C5a and C5b-9 components [5], which are directly associated with the presented clinical symptoms and course of inflammation. In this article, we present 3 cases of extrapulmonary manifestations of COVID 19, including reported symptoms, diagnosis, and administered treatments.
Among the patients under the care of the Clinic of Transplantation Medicine, Nephrology and Internal Diseases during the development of the COVID-19 pandemic (in 2020–2021), we observed patients with atypical clinical symptoms preceded by SARS-CoV-2 infection.
Based on the medical history and the results of additional tests, we linked the occurrence of these symptoms with the prevailing pandemic. We present 3 clinical cases of extrapulmonary manifestation of SARS-CoV-2 infection and discuss the diagnostic methods and treatments administered.
Case Reports
CASE 1:
A 29-year-old woman after a kidney transplantation from a living donor (mother) in 2013, due to end-stage kidney disease in the course of IgA nephropathy, with stable parameters of the graft (creatinine 1.42 mg/dl, eGFR 43.7 ml/min, 1.73 m2), was admitted to the hospital with symptoms of weakness, cough, and the elevated heart rate, starting 2 weeks before. The patient denied having dyspnea, chest pain, and fever.
Weight loss, polydipsia, and polyuria were not observed. Laboratory tests revealed: glycemia 618 mg/dL, HbA1C 7.6%, hyponatremia 128 mmol/L, creatinine 1.8 mg/dl, CRP 24.2 mg/L, and WBC 13 000 cells/uL (Table 1). Computed tomography (CT) of the chest revealed unilateral, multifocal ground-glass opacities in the right upper lobe and segment 6 of the right lower lobe. Postinflammatory changes were found in the left lung. Nasopharyngeal swab reverse transcriptase-PCR (RT-PCR) tested positive for SARS-CoV-2.
Pneumonia was diagnosed due to COVID-19 disease and new-onset diabetes mellitus. Low-flow oxygen supplementation, antibiotic therapy (ceftriaxone), steroid therapy, and anticoagulant therapy were administered. Mycophenolate mofetil was temporarily discontinued, and only 2 immunosuppressants (prednisone, tacrolimus) were continued.
Intensive insulin therapy through an infusion pump was administered. After stabilization of vital parameters, recovering from pneumonia, normalization of inflammatory blood markers, and lowering glycemia levels, the patient was discharged from the hospital, with a prescription of a fast-acting insulin analog (NovoRapid) in scheme 10-12-10 units s.c. and insulin isofan (Gensulin N) 6 units s.c. in the evening. Furthermore, the patient was referred to the Diabetes Clinic for additional examinations of diabetes mellitus. During hospitalization, anti-islet cells (ICA) and anti-GAD antibodies were tested in the blood–both tests were negative. Insulin and C-peptide levels were normal (93.3 pmol/L and 0.811 nmol/L, respectively). HBA1C was 7.2%. Over time, slow improvement of glycemia was observed, so that insulin doses were steadily reduced. Two months after receiving a positive SARS-CoV-2 PCR test result, due to satisfactory glucose levels, the decision was made to treat with only diet. Control HBA1C was 6.0%. Daily glucose levels have remained normal.
CASE 2:
A 34-year-old man, with past medical history of living-donor kidney transplantation (mother) in 2017 due to end-stage kidney disease in course of IgA nephropathy, was admitted to the hospital with recurrent fever up to 39.5°C lasting for 3 weeks, chronic fatigue, back pain, bloating, nausea, pain in upper quadrant of the abdomen, and loss of appetite.
The beginning of the fever was accompanied by loss of smell and taste. The patient neglected symptoms of upper respiratory system infection (including cough and dyspnea). RT-PCR for SARS-CoV-2 test was performed several times; the first result was inconclusive and the next ones were negative.
At the beginning, ambulatory treatment with antibiotics (amoxicillin with azithromycin, further amoxicillin with clavulanate acid) was administered, but no resolution of symptoms was observed.
During hospital admission, the patient was in average general condition, and laboratory tests revealed: leukocytosis 15 G/L, with lymphopenia 0.86 G/L, granulocytosis >13 G/L, CRP 197 mg/L, D-dimers >4500 ng/mL, pro-BNP >2900 pg/mL, INR 1.4 (no history of anticoagulants intake), fibrinogen 6.34g/L, creatinine 2.82 mg/dL. Slightly reduced CRP (159 mg/L), D-dimers (3969 ng/mL), and significant thrombocytosis (604 G/L) were observed during the next few days.
A chest CT revealed typical COVID-19 findings in the lungs. Considering the pandemic, characteristic clinical signs and symptoms, as well as incorrect results of performed tests, the assumption of recent SARS-CoV-2 infection was made and proven by highly elevated levels of IgM and IgG antibodies. In the following days, the patient reported new symptoms of chest pain and persistent pain in both knees. An infection of unknown origin related to post-COVID-19 disease was taken into consideration, which would also explain the prolonged duration of the presented symptoms. Due to the initial diagnosis, mycophenolate mofetil was temporarily discontinued and the 2 immunosuppressants (prednisone and tacrolimus) were continued.
Blood and urine cultures were performed numerous times, including aerobic as well as anaerobic bacteria and fungi: Aspergillus, Candida, and Cryptococcus. Viral load of CMV and EBV was checked as well. All test results were negative.
Due to elevated inflammation markers and refractory fever, broad empiric antibiotic therapy with ceftriaxone and levofloxacin was begun. Because of no improvement of clinical condition, meropenem with vancomycin were applied (still empiric), then sulfamethoxazole with trimethoprim, and finally imipenem with linezolid.
Simultaneously, the origins of the infection were sought and new imaging tests were performed. Echocardiography excluded endocarditis. Repeated chest and abdominal CT revealed acute acalculous cholecystitis with presence of pericholecystitis and increased density and edema of surrounding tissues (Figure 1). Additionally, increased density of fatty tissue around the liver hilum was observed. In the chest, basal bilateral single, streaky atelectasis and fibrous changes were noticed, as well as slight pericardial effusion. All abnormalities have been observed in follow-up examinations.
Surgical consultation was performed, in which no direct recommendation for cholecystectomy was ascertained, and further observation was advised. The next performed imaging test was essential for diagnosis and treatment. After cholangio-MRI, coagulopathy in COVID-19 was suspected as the source of the previously described abnormalities: acalculous cholecystitis, epiploic appendagitis of right colic flexure, and inflammation of pericardial fat pad in the left cardiophrenic angle was diagnosed (Figures 2–4).
Due to the suspicion of a thrombotic process, the dose of low-molecular-weight heparin was increased from prophylactic to therapeutic, under anti-Xa control. At the same time, antibiotics were discontinued due to their ineffectiveness. As a result of the treatment, the symptoms gradually disappeared and the clinical condition improved. Laboratory tests revealed decrease of D-dimers levels <600 ng/ml and CRP 73 mg/L. White blood cells and thrombocyte dysfunctions returned to normal (WBC 8.16 G/L; PLT 328 G/L). The patient was discharged from the hospital at the end of December with satisfying tests results. One week later, during ambulatory check-up, morphology and kidney function parameters had returned to normal levels (Table 2).
CASE 3:
A 68-year-old patient after left nephrectomy in 2007 (living kidney donor) with stable parameters of the right kidney (creatinine 0.72 mg/dL, eGFR 108 ml/min/1.73 m2) was admitted to the department with erythematous changes on both shins and recurrent fever up to 38°Celsius. In April 2021 he was hospitalized due to COVID-19 pneumonia (RT-PCR tested positive for SARS-CoV-2). In HRCT performed during a previous hospitalization, 40% of the lungs were affected by disease. The patient received treatment with reconvalescent plasma and empiric antibiotic therapy (ceftazidime). A few days after discharge from the hospital, the fever had returned. Additionally, painless, nodular, reddening lesions on both shins, accompanied by itching, were observed, and were extending and hardening.
On an outpatient basis, empiric antibiotic therapy (azithromycin) and anticoagulant therapy (low-molecular-weight heparin in treatment doses 0.6 ml SC) were administered.
Slight regression of symptoms, including itching, nodular expansion, and fever episodes, was achieved. During the recent hospitalization, anticoagulant treatment was continued. Due to normal inflammation parameters and negative blood and urine cultures, azithromycin was discontinued. Additionally, laboratory tests showed elevated D-dimers levels >1300 ng/mL and thrombocytosis 645 G/L (Table 3). During diagnosis, anti-HCV antibodies were positive, but the connection between HCV infection and the presenting symptoms was excluded. Thereafter, a negative HCV RNA PCR result was obtained.
A follow-up chest CT revealed changes typical for past COVID-19 infection: ground-glass opacities with consolidations and minor reticular pattern linear opacifications in both lungs. USG doppler of lower-extremity veins excluded thrombosis. The projection of nodal lesions showed obliterated subcutaneous fatty tissue, with increased echogenicity, dimly separated from healthy tissue. After Dermatological consultation, erythema nodosum during COVID-19 infection was diagnosed. Over the following days of hospitalization, improvement of clinical symptoms (resolution of fever and significant regression of nodal lesions) was observed. Laboratory tests showed gradual normalization of D-dimer and thrombocyte levels. During an outpatient follow-up visit, the patient’s condition was good, he did not report any complaints, and the skin changes disappeared. Laboratory test results were normal.
Discussion
HEMOPOETIC DISORDERS:
In all presented cases, morphology abnormalities such as leukocytosis, lymphopenia, neutrophilia, and thrombocytopenia were observed. Recent studies showed that 67–90% of patients presented with lymphopenia. Decreased CD 4+ T cells and CD 8+ T cells and leukocytosis with thrombocytopenia are negative prognostic marker [3]. This correlates with clinical manifestations presented in our cases, especially Case 2. The possible pathophysiology might be direct cytotoxic action of the virus related to ACE2-dependent or ACE2-independent entry into lymphocytes, apoptosis-mediated lymphocyte depletion, and inhibition effects of lactic acid on lymphocyte proliferation [3]. In Case 2, laboratory results seemed to correspond with the general course of inflammation. Meticulous check-up is essential to monitor the results of treatment.
COAGULOPATHY:
Coagulopathy commonly presents as elevated D-dimer and fibrinogen, but also as prolonged prothrombin time and partial thromboplastin time. Highly elevated D-dimer and its prolonged increased levels has been linked with worse mortality among COVID-19 patients [3].
A connection between COVID-19 and thrombotic complications has been observed since the early stages of the pandemic, including arterial and venous thrombosis, catheter-related thrombosis, and cytokine-releasing syndrome [3]. Our Case 2, since admission to the hospital, had significantly elevated D-dimer and fibrinogen levels. Most common places of thrombosis have been excluded through standard radiograph diagnostic. The diagnosis of inflammation of omental appendices shows that thrombosis can affect almost every part of the human body. In this case, there was no massive thrombotic process and no major vessels were clogged, but laboratory test results were disproportionately high. Although empiric use of higher-than-routine prophylactic doses without proven diagnosis has been discussed, standard-dose prophylaxis should be considered in hospitalized patients without evidence of thrombosis [3].
ACUTE ACALCULOUS CHOLECYSTITIS:
Acute acalculous cholecystitis is a rarely reported extrapulmonary manifestation of COVID-19, generally caused by hypomobility of the gallbladder in response to increased intraluminal pressures evolving in inflammation, ischemia, and necrosis [9]. During severe COVID-19, prolonged parenteral nutrition and mechanical ventilation might be the cause, but in Case 2, the course of disease could be described as mild. The patient was not mechanically ventilated and he did not receive parenteral nutrition. The probable cause of this complication was an ischemic process due to thrombosis. This hypothesis is supported by elevated D-dimer and fibrinogen levels, but also the fact that there was thrombosis in other locations. What needs to be emphasized is the lack of clinical manifestations of cholecystitis. In addition, no diagnostic endoscopy has been performed, as multi-society guidelines recommend require urgent reasons [3].
DIABETES MELLITUS:
Case 1 had new-onset diabetes mellitus type 1. Although most research has focused on correlations between diabetes and severity of SARS-CoV-2 infection, some studies presented early onset of disease due to COVID-19 infection. Possibly, the virus can enter pancreatic islets B cells and cause reversible damage presented by transient hyperglycemia and even ketoacidosis [6]. In the presented case, during diagnosis, common antibodies leading to DM1 became negative. Furthermore, after 3 months, the patient was able to stop insulin therapy and control her glycemia only through diet.
DERMATOLOGICAL MANIFESTATIONS:
During the COVID-19 pandemic, different dermatologic manifestations have been reported, mostly erythromatous rash, urticaria, chickenpox-like vesicles, livedoid, and necrotic lesions [3]. A study of 88 patients from Italy showed that up to 20.4% of patients presented with cutaneous manifestations [11]. In a systematic review by Cinforti, erythema nodosum (EN) was also described as a rare clinical demonstration [8].
EN is commonly seen during infections, sarcoidosis, and neoplastic or inflammatory diseases. Up to 55% of cases are idiopathic [7]. It is crucial to exclude those feasible causes, including drug exposure, during diagnosis. The exact pathophysiology is unknown, although hypersensitivity response to SARS-CoV-2 RNA, cytokine-release syndrome, deposition of microthrombi, and vasculitis are possible mechanisms [3]. Nonetheless, treatment with local glucocorticosteroids has been effective.
Conclusions
We report 3 cases of extrapulmonary manifestations of SARS-CoV-2. The literature describing different courses of disease is consistently expanding; therefore, clinicians should be aware of various, sometimes life-threatening manifestations of COVID-19, so the diagnosis is made quickly and adequate treatment is provided immediately.
Figures
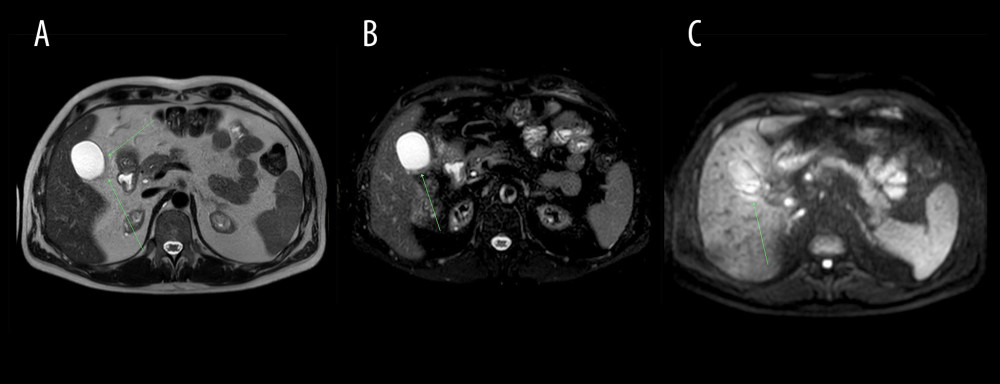 Figure 1. Abdominal CT axial images at the gallbladder level (arrows). (A) Non-contrast CT (performed on Nov 25, 2020) presented normal gallbladder, (B) Contrast enhanced CT on Dec 2, 2020 indicated slightly enhanced and thickened gallbladder wall, pericholecystic fluid, and inflammatory fat stranding (arrow), (C) Non-contrast CT revealed partial resolution of inflammatory process on Dec 9, 2020.
Figure 1. Abdominal CT axial images at the gallbladder level (arrows). (A) Non-contrast CT (performed on Nov 25, 2020) presented normal gallbladder, (B) Contrast enhanced CT on Dec 2, 2020 indicated slightly enhanced and thickened gallbladder wall, pericholecystic fluid, and inflammatory fat stranding (arrow), (C) Non-contrast CT revealed partial resolution of inflammatory process on Dec 9, 2020. 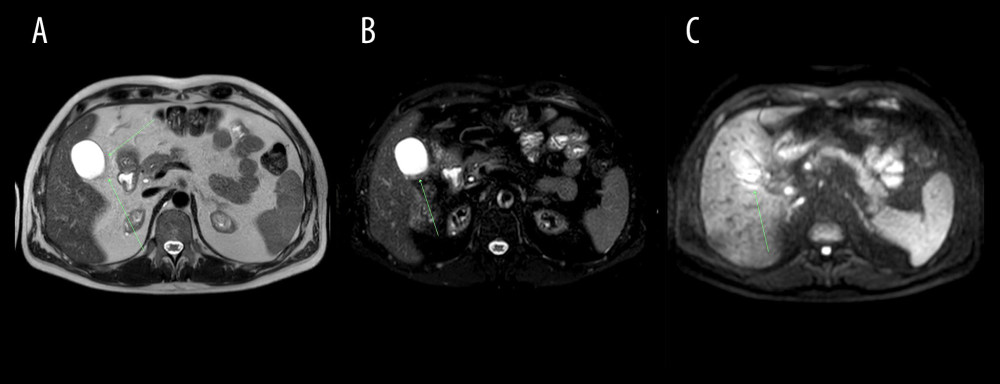 Figure 2. Abdominal MRI obtained on December 10, 2020. (A) T2-weighted, (B) T2 SPAIR, (C) DWI; axial images at the gallbladder level. Gallbladder surrounded by inflamed adipose tissue (arrow) – low signal intensity on T2-weighted and T2 SPAIR images and increased signal intensity on diffusion-weighted image.
Figure 2. Abdominal MRI obtained on December 10, 2020. (A) T2-weighted, (B) T2 SPAIR, (C) DWI; axial images at the gallbladder level. Gallbladder surrounded by inflamed adipose tissue (arrow) – low signal intensity on T2-weighted and T2 SPAIR images and increased signal intensity on diffusion-weighted image. 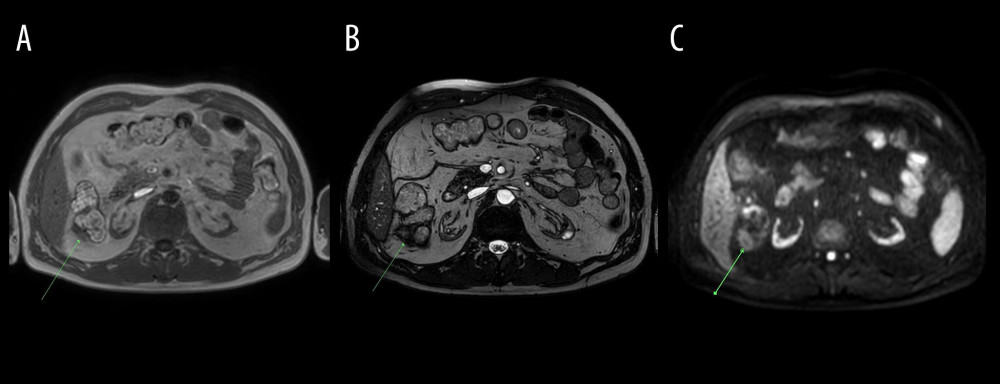 Figure 3. Abdominal MRI obtained on December 10, 2020. (A) mDIXON (water), (B) BTFE, (C) DWI axial images at the level of the right colic flexure. Epiploic appendagitis and inflamed attached adipose tissue (arrows): low signal intensity on T1-weighted (mDIXON water) as well as on BTFE images and increased signal intensity on DWI image.
Figure 3. Abdominal MRI obtained on December 10, 2020. (A) mDIXON (water), (B) BTFE, (C) DWI axial images at the level of the right colic flexure. Epiploic appendagitis and inflamed attached adipose tissue (arrows): low signal intensity on T1-weighted (mDIXON water) as well as on BTFE images and increased signal intensity on DWI image. 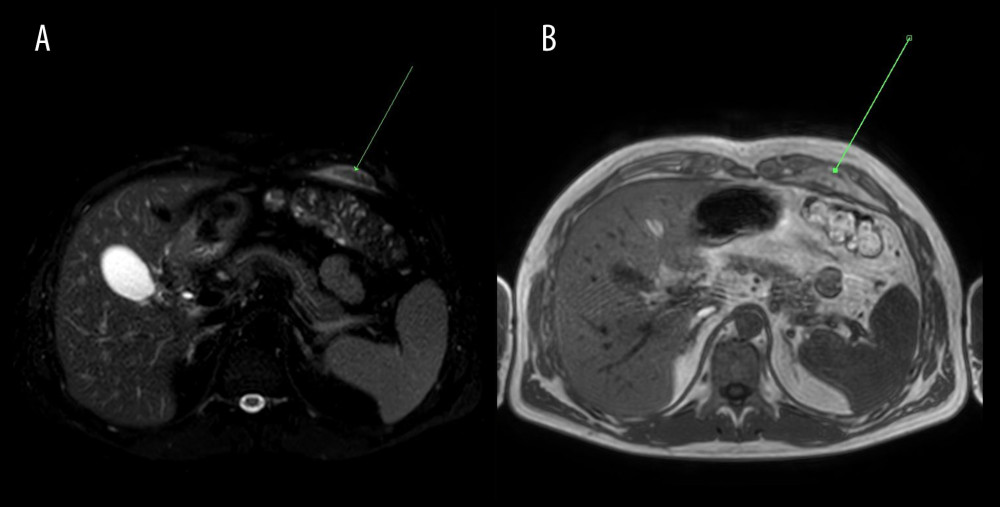 Figure 4. Abdominal MRI obtained on December 10, 2020. (A) T2 SPAIR, (B) mDixon water, (C) DWI axial images at the level of pericardial fat. Inflammation of pericardial fat pad in the left cardiophrenic angle – increased signal intensity on T2 SPAIR as well as on DWI images and decreased signal intensity on T1-weighted (mDIXON water) image.
Figure 4. Abdominal MRI obtained on December 10, 2020. (A) T2 SPAIR, (B) mDixon water, (C) DWI axial images at the level of pericardial fat. Inflammation of pericardial fat pad in the left cardiophrenic angle – increased signal intensity on T2 SPAIR as well as on DWI images and decreased signal intensity on T1-weighted (mDIXON water) image. Tables
Table 1. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 1).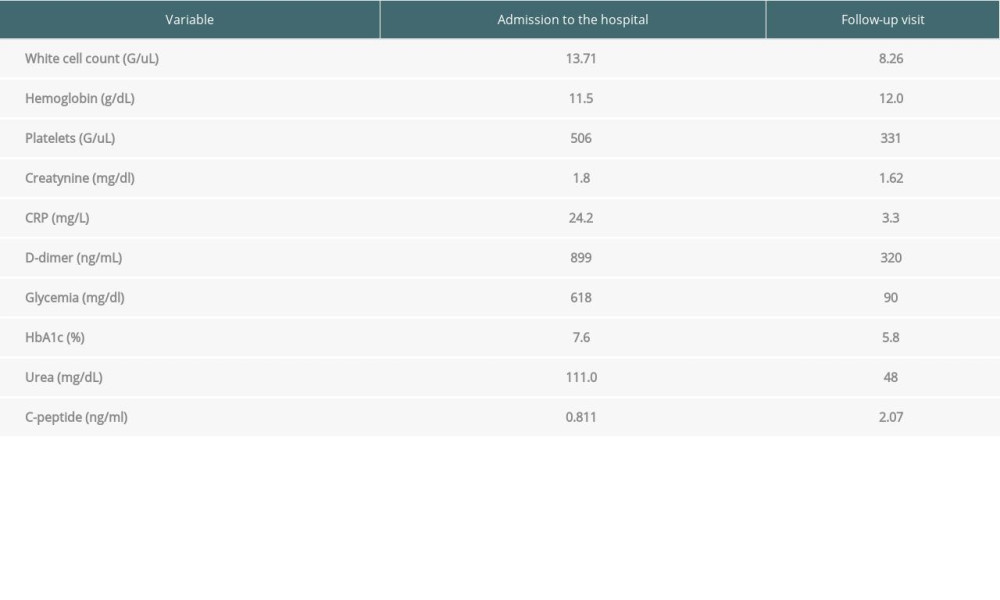 Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2).
Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2).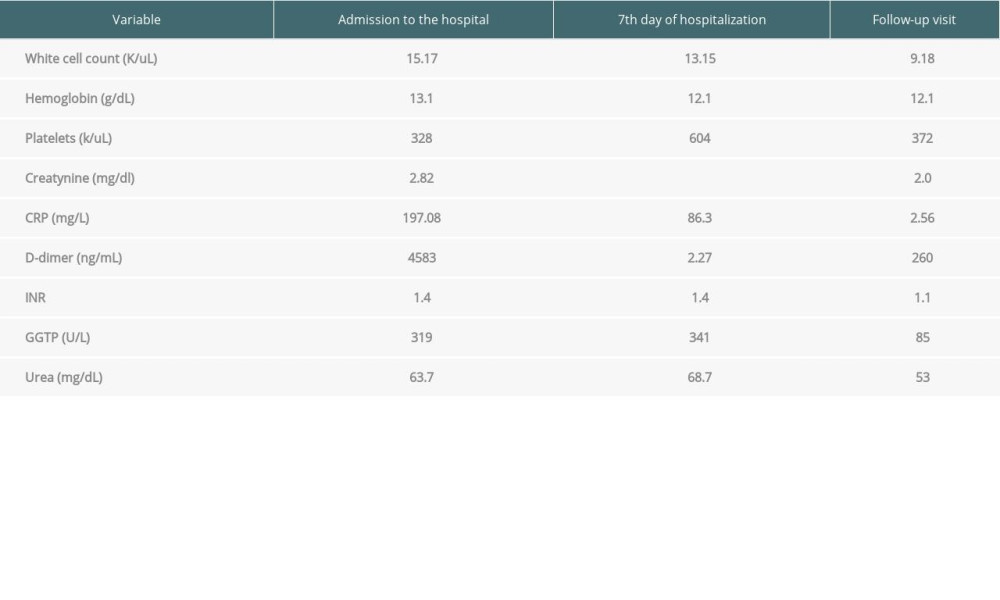 Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3).
Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3).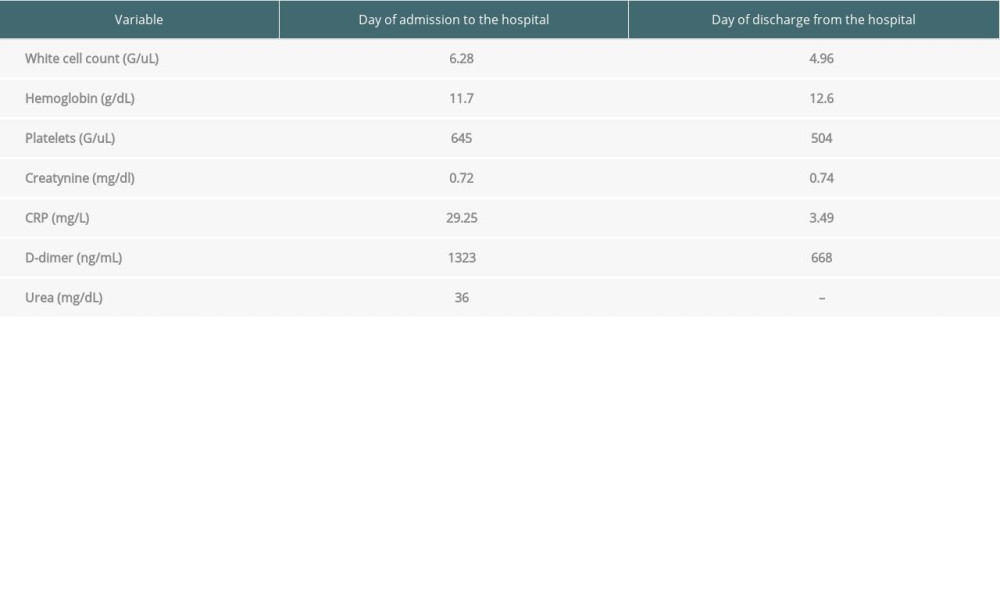
References
2. 07 06, 2021 https://www.who.int/emergencies/diseases/novel-coronavirus-2019
3. Gupta A, Madhavan MV, Sehgal K, Extrapulmonary manifestations of COVID-19: Nat Med, 2020; 26(7); 1017-32
4. Zhou P, Yang XL, Wang XG, A pneumonia outbreak associated with a new coronavirus of probable bat origin: Nature, 2020; 579(7798); 270-73
5. Mahmudpour M, Roozbeh J, Keshavarz M, COVID-19 cytokine storm: The anger of inflammation: Cytokine, 2020; 133; 155151
6. Boddu SK, Aurangebadkar G, Kuchay MS, New onset diabetes, type 1 diabetes and COIVD-19: Diabetes Metab Syndr, 2020; 14(6); 2211-17
7. Blake T, Manahan M, Rodins K, Erythema nodosum – a review of an uncommon panniculitis: Dermatol Online J, 2014; 20; 22376
8. Conforti C, Dianzani C, Agozzino M, Cutaneous manifestations in confirmed COVID-19 patients: A systematic review: Biology (Basel) 5, 2020; 9(12); 449
9. Balmadrid B, Recent advances in management of acalculous cholecystitis: F1000Research, 2018; 7(F1000 Faculty Rev); 1660
10. Johnson KD, Harris C, Cain JK, Pulmonary and extra-pulmonary clinical manifestations of COVID-19: Front Med (Lausanne), 2020; 7; 526
11. Recalcati S, Cutaneous manifestations in COVID-19: A first perspective: J Eur Acad Dermatol Venereol, 2020; 34; e212-13
Figures
 Figure 1. Abdominal CT axial images at the gallbladder level (arrows). (A) Non-contrast CT (performed on Nov 25, 2020) presented normal gallbladder, (B) Contrast enhanced CT on Dec 2, 2020 indicated slightly enhanced and thickened gallbladder wall, pericholecystic fluid, and inflammatory fat stranding (arrow), (C) Non-contrast CT revealed partial resolution of inflammatory process on Dec 9, 2020.
Figure 1. Abdominal CT axial images at the gallbladder level (arrows). (A) Non-contrast CT (performed on Nov 25, 2020) presented normal gallbladder, (B) Contrast enhanced CT on Dec 2, 2020 indicated slightly enhanced and thickened gallbladder wall, pericholecystic fluid, and inflammatory fat stranding (arrow), (C) Non-contrast CT revealed partial resolution of inflammatory process on Dec 9, 2020. Figure 2. Abdominal MRI obtained on December 10, 2020. (A) T2-weighted, (B) T2 SPAIR, (C) DWI; axial images at the gallbladder level. Gallbladder surrounded by inflamed adipose tissue (arrow) – low signal intensity on T2-weighted and T2 SPAIR images and increased signal intensity on diffusion-weighted image.
Figure 2. Abdominal MRI obtained on December 10, 2020. (A) T2-weighted, (B) T2 SPAIR, (C) DWI; axial images at the gallbladder level. Gallbladder surrounded by inflamed adipose tissue (arrow) – low signal intensity on T2-weighted and T2 SPAIR images and increased signal intensity on diffusion-weighted image. Figure 3. Abdominal MRI obtained on December 10, 2020. (A) mDIXON (water), (B) BTFE, (C) DWI axial images at the level of the right colic flexure. Epiploic appendagitis and inflamed attached adipose tissue (arrows): low signal intensity on T1-weighted (mDIXON water) as well as on BTFE images and increased signal intensity on DWI image.
Figure 3. Abdominal MRI obtained on December 10, 2020. (A) mDIXON (water), (B) BTFE, (C) DWI axial images at the level of the right colic flexure. Epiploic appendagitis and inflamed attached adipose tissue (arrows): low signal intensity on T1-weighted (mDIXON water) as well as on BTFE images and increased signal intensity on DWI image. Figure 4. Abdominal MRI obtained on December 10, 2020. (A) T2 SPAIR, (B) mDixon water, (C) DWI axial images at the level of pericardial fat. Inflammation of pericardial fat pad in the left cardiophrenic angle – increased signal intensity on T2 SPAIR as well as on DWI images and decreased signal intensity on T1-weighted (mDIXON water) image.
Figure 4. Abdominal MRI obtained on December 10, 2020. (A) T2 SPAIR, (B) mDixon water, (C) DWI axial images at the level of pericardial fat. Inflammation of pericardial fat pad in the left cardiophrenic angle – increased signal intensity on T2 SPAIR as well as on DWI images and decreased signal intensity on T1-weighted (mDIXON water) image. Tables
 Table 1. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 1).
Table 1. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 1). Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2).
Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2). Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3).
Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3). Table 1. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 1).
Table 1. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 1). Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2).
Table 2. Laboratory results from admission to the hospital, after 7 days of hospitalization, and from follow-up visit. (Case presentation 2). Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3).
Table 3. Laboratory results from admission to the hospital and from follow-up visit. (Case presentation 3). In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








