06 September 2022: Original Paper
Leucine-Rich Alpha-2-Glycoprotein (LRG-1) as a Potential Kidney Injury Marker in Kidney Transplant Recipients
Anna Popova123ACDE*, Aiga Vasiļvolfa134ABCDEF, Kārlis Rācenis12ACD, Renārs Erts5C, Baiba Šlisere2BC, Anna Jana Saulīte12EF, Ieva ZiedinaDOI: 10.12659/AOT.936751
Ann Transplant 2022; 27:e936751
Abstract
BACKGROUND: Kidney transplantation is the treatment of choice for most patients with end-stage renal disease. To improve patient and transplant survival, non-invasive diagnostic methods for different pathologies are important. Leucine-rich alpha-2-glycoprotein (LRG-1) is an innovative biomarker that is elevated in cases of angiogenesis, inflammation, and kidney injury. However, there are limited data about the diagnostic role of LRG-1 in kidney transplant recipients. The aim of this study was to evaluate the association between serum LRG-1, urine LRG-1, and kidney transplant function and injury.
MATERIAL AND METHODS: We enrolled 35 kidney transplant recipients in the study. LRG-1 in the serum and urine was detected using ELISA. We evaluated the correlation of serum and urine LRG-1 with traditional serum and urine kidney injury markers.
RESULTS: A higher level of serum LRG-1 correlates with a higher level of urine LRG-1. Serum LRG-1 has a positive correlation with transplant age, serum urea, serum creatinine, serum cystatin C, proteinuria, and fractional excretion of sodium (FENa) and a negative correlation with hemoglobin and estimated glomerular filtration rate (eGFR). Urine LRG-1 has a positive correlation with serum cystatin C, proteinuria, and urine neutrophil gelatinase-associated lipocalin (NGAL).
CONCLUSIONS: Higher levels of serum and urine LRG-1 are associated with kidney transplant injury and functional deterioration. Thus, LRG-1 might be also as a biomarker for tubular dysfunction in patients after kidney transplantation.
Keywords: Kidney Function Tests, Kidney Transplantation, LRG1 Protein, Human, cystatin C, Glycoproteins, Humans, Kidney, Leucine, Proteinuria
Background
For most patients with end-stage renal disease, kidney transplantation is the treatment of choice because it significantly improves survival and quality of life compared to dialysis [1]. Leucine-rich alpha-2-glycoprotein-1 (LRG-1) could be a potential non-invasive diagnostic tool for kidney transplant recipients. Improving the health of recipients and maintaining graft function not only improves the quality of patient’s lives and survival but also reduces the need for re-transplantation and increases the number of organs available for transplantation [2].
LRG-1, which was first isolated from human serum by Haupt and Baudner in 1977 [3], is a protein that is expressed during blood cell-granulocyte differentiation [4]. LRG-1 consists of 347 amino acids [5,6] and has a molecular weight of 50 kDa [5]. Based on mRNA analysis, LRG-1 is thought to be normally produced in liver cells and neutrophils [4], but it may be expressed in a variety of cells in the presence of inflammatory cytokines (interleukin-6, interleukin-1b), lipopolysaccharides and tumor necrosis factor-alpha (TNF-α) [3,6]. LRG-1 levels increase in inflammatory processes and infectious diseases [3,7–10], malignancy [11–16], autoimmune diseases such as rheumatoid arthritis [10], inflammatory bowel disease [16], systemic lupus erythematosus [17], and the pathogenesis of diabetic nephropathy [5].
LRG-1 has been described as a biomarker of acute kidney injury [6] and tubular dysfunction [5,6]. Terada et al showed that serum and urine LRG-1 levels were increased in a mouse model of acute kidney injury [6]. In that study, immunohistological examination showed that LRG was mainly expressed in renal tubular cells [6]. Lee and co-authors also studied a mouse model and observed that urine LRG-1 was expressed in proximal, distal tubules and collecting ducts. Data suggest that LRG-1 could be used as a marker of tubular damage in various kidney diseases [5].
There are few studies on the role of LRG-1 in patients undergoing renal replacement therapy [18,19]. In a study by Glorieux and co-authors, plasma LRG-1 levels were 2-fold higher in patients with end-stage renal disease compared with chronic kidney disease (CKD) stages 2 and 3 [18]. Serum LRG-1 has been shown to have a negative correlation with GFR [18]. Thus, LRG-1 could be a potential marker for CKD [18].
Banon-Maneus and co-authors studied the presence of various proteomes in urine and concluded that urine LRG-1 can be used as a biomarker to assess the severity of interstitial fibrosis and tubular atrophy in patients after kidney transplantation [19].
Few studies have described the diagnostic role of LRG-1 in kidney transplant recipients. Therefore, the aim of the present study was to evaluate the association between this biomarker and the function and injury of kidney transplants.
Material and Methods
This study was performed according to the ethical principles of the Declaration of Helsinki and approved by the Clinical Research Ethics Committee of the Development Society of Pauls Stradiņš Clinical University Hospital, approval number 280520-18L. The patients provided written informed consent prior to study participation.
The study, conducted in spring 2020 at Pauls Stradiņš Clinical University Hospital, included 35 patients over 18 years old with a stable stage functioning kidney transplant. The study did not include patients with active infections, autoimmune diseases or diabetes mellitus, patients who had had a kidney transplantation in the last 12 months, or pregnant women. Following criteria despite kidney function may influence LRG-1 levels.
Baseline demographics and clinical characteristics were obtained from patient and medical records. We collected peripheral blood and urine samples. Urine and blood examination was performed 7.7 (±6.9) years after transplantation (median time – 6 years [1 to 25]). To assess the association between LRG-1 and kidney function, we used the correlation with serum creatinine, eGFR and cystatin C. Clinical chemistry tests (eg, creatinine, urea, sodium) were evaluated on a Siemens Atellica analyzer. GFR was estimated according to the MDRD (Modification of Diet in Renal Disease) formula. NGAL was measured using chemiluminescent microparticle immunoassay. Correlation with urine NGAL and FENa was performed to obtain association between LRG-1 and tubular dysfunction. Proteinuria was detected using the protein-to-creatinine ratio.
The obtained data were processed with Microsoft Office Excel, IBM SPSS Statistics and R-Studio programs. Parameters are reported as median values with interquartile ranges, and the frequency of results as a percentage. Given that the obtained data did not correspond to the normal distribution, the Spearman method was used to determine correlations between variables. The correlation is considered weak if the correlation coefficient (r) is less than 0.25, moderate if r=0.25–0.75, and strong if r >0.75. Age adjustment was used for data analysis. To estimate urine LRG-1 as a biomarker for proteinuria detection, we used receiver operating characteristic (ROC) curve.
Results
PATIENT DEMOGRAPHIC AND CLINICAL CHARACTERISTICS:
There were 35 patients with a functioning kidney graft enrolled in this study. Their median age was 57 years, and 60% of patients were male. The median transplant age was 6.0 (IQR 2.0–10.0) years. The patient median serum creatinine was 123 (IQR 108–176) μmol/L, and the eGFR was 47.1 (IQR 33.7–60.0) mL/min/1.73 m2. The most common maintenance immunosuppressive regimen was therapy with tacrolimus, mycophenolate, and prednisolone (48.6% of patients). Patient characteristics are listed in Table 1.
SERUM AND URINE LRG-1 LEVELS IN KIDNEY TRANSPLANT RECIPIENTS:
The mean concentration of serum LRG-1 was 51.07±19.81 μg/ml, median – 47.1 μg/mL, and the mean concentration of urine LRG-1 was 0.17±0.19 μg/ml, median – 0.09 μg/mL. After adjustment for patient’s age, there was a positive correlation between serum and urine LRG-1 (r=0.41; 95% CI: 0.09–0.66; p=0.01).
Linear regression analysis was conducted with urine LRG-1 as dependent variable. Every 1 ug/ml increase in urine LRG-1 was associated with a 53-units increase in serum LRG-1. Linear regression accounted for R2=25% of the variance of the serum LRG-1, as shown in Figure 1.
SERUM AND URINE LRG-1 CORRELATION WITH OTHER QUANTITATIVE DATA:
After adjustment for patient’s age, serum LRG-1 had a positive correlation with transplant age (r=0.52; 95% CI: 0.26–0.71; p<0.01), serum urea (r=0.59; 95% CI: 0.23–0.68; p<0.01), serum creatinine (r=0.33; 95% CI: 0.01–0.59; p=0.04), serum cystatin C (r=0.42; 95% CI: 0.08–0.67; p=0.01), serum phosphorus (r=0.48; 95% CI: 0.14–0.72; p<0.01), proteinuria (r=0.35; 95% CI: 0.02–0.61; p=0.03), and FENa (r=0.35; 95% CI: 0.01–0.62; p=0.04). Serum LRG-1 had a negative correlation with hemoglobin (r=−0.37; 95% CI: −0.66 – −0.01; p=0.01) and eGFR (r=−0.35; 95% CI: −0.01 – −0.61; p=0.03). The results are shown in Table 2.
Urine LRG-1 had a positive correlation with serum cystatin C (r=0.39; 95% CI: 0.02–0.67; p=0.03), parathyroid hormone (PTH) (r=0.5; 95% CI: 0.19–0.72; p<0.01), proteinuria (r=0.59; 95% CI: 0.01–0.3; p<0.01) and urine NGAL (r=0.44; 95% CI: 0.13–0.67; p<0.01) (shown in Table 2).
The chance line on the ROC curve means the same probability of detecting healthy/sick using the urine LRG-1 biomarker as the area of the ROC curve AUC=0.5. The diagnostic accuracy of urine LRG− 1 for the diagnosis of proteinuria >0.5 mg/g was moderate (AUC, 0.77; 95% CI: 0.60–0.94) (Figure 2). This means there is a 77% chance by increase of LRG-1 to distinguish between patients with or without proteinuria.
The ROC curve-derived optimal cut-off values for urine LRG-1 was 0.1 ug/ml. The use of these cut-off values resulted in sensitivity (Se), specificity (Sp), positive predictive value (PPV), and negative predictive value (NPV) of 79%, 70%, 65%, and 82%, respectively, for urine LRG-1 (Table 3). This refers to the higher probability of a positive test (79%) than the probability of a negative test (70%).
Discussion
Higher LRG-1 levels are observed in various disorders, such as malignancy and autoimmune and inflammatory diseases [3,7–10]. Data in the literature also show that LRG-1 levels are elevated in cases of acute or chronic kidney injury. In our study, the main aim was to investigate LRG-1 as a biomarker for kidney injury. The study did not include patients with infection, diabetes, or autoimmune diseases because these conditions per se can increase serum or urine LRG-1 levels. Presence of infection was checked by clinical and laboratory examination. Patients did not have any symptoms such as fever, pain, or cough, and none of them had any remarkable changes in laboratory results (eg, elevated c-reactive protein, white blood cell count). Urinalysis and urine culture were sent for analysis to exclude urinary tract infection. Eleven (31%) patients had asymptomatic bacteriuria with positive urine culture. There were no significant changes in serum or urine LRG1 in patients with or without asymptomatic bacteriuria. BK virus nephropathy was excluded in patients who underwent kidney graft biopsy and was not considered by nephrologist evaluation in those who did not have a biopsy.
This study is one of the few to explore LRG-1 as a potential biomarker for kidney injury, particularly in a kidney transplant recipient population.
There is no defined reference interval for serum and urine LRG-1. Weivoda et al analyzed patients with different infections and autoimmune diseases, and the normal serum LRG-1 level was defined as up to 50 μg/mL based on a control group of 20 patients [20]. In a study of systemic lupus erythematosus, Ahn et al found that a serum LRG-1 level of more than 45.7 ng/mL could indicate disease activity [17]. The reported norms differ significantly since the 2 studies used different units of measurement. In this study, mean serum LRG-1 was 51.07±19.81 μg/mL. In comparison, in a study by Yang et al, which included 169 hemodialysis patients, the mean serum LRG-1 was higher (67.73±15.10 μg/mL) [21]. This result indirectly suggests that patients with lower kidney function have higher serum LRG-1.
There was a positive correlation between serum and urine LRG-1. Similar data have been described in type 2 diabetes mellitus patients who were studied over a 3-year period to investigate the correlation of LRG-1 with albuminuria progression. The researchers found that higher serum LRG-1 correlated with higher urine LRG-1 and indicated the progression of albuminuria [22].
We determined that a higher serum LRG-1 level had a moderate positive correlation with the level of serum urea, serum creatinine, and serum cystatin C and a moderate negative correlation with eGFR. These correlations show that serum LRG-1 is associated with decreased kidney function of the renal transplant. Glorieux et al also found a negative correlation between serum LRG-1 and eGFR [18].
In our study, serum LRG-1 had a negative correlation with the level of hemoglobin, which may indirectly indicate worse kidney transplant function because decreased renal function is associated with erythropoietin deficiency and secondary anemia [23]. In addition, serum LRG-1 had a positive correlation with a higher level of phosphorus. This might be related to a decreased transplant function, tubular injury, and secondary hyperparathyroidism [23]. Yang et al found a negative correlation between serum LRG-1 and hemoglobin in hemodialysis patients, but they did not find a correlation between serum LRG-1 and bone mineral disorders [21].
We found a positive correlation between serum LRG-1 and transplant age. A longer time after kidney transplantation is associated with decreased GFR levels due to chronic transplant dysfunction [24]. Banon-Maneus et al studied different proteomes in urine. They found that urine LRG-1 may be used as a marker in diagnosing chronic graft dysfunction because it correlates with the severity of interstitial fibrosis and tubular atrophy [19].
In our study, urine LRG-1 had a positive correlation with proteinuria and urine NGAL. Other studies suggest that plasma and urine LRG-1 levels are increased in cases of proteinuria and tubular damage [5,22]. In a study on a mouse model of induced acute kidney injury, Lee et al found that urine LRG-1 was expressed in the renal tubular epithelium and could be used as a tubular damage marker [5]. Urine NGAL is produced by tubular epithelial cells during renal damage [25], and the correlation of these 2 markers (urine LRG-1 and urine NGAL) in our study indirectly suggests tubular damage.
This is a pilot study with the relatively small study cohort. Similar research needs to be performed in a larger cohort to obtain more precise correlations.
Conclusions
Our study demonstrates the potential role of serum and urine LRG-1 as a kidney injury marker in kidney transplant recipients. It correlates with other tubular injury markers (NGAL) and functional deterioration. Further research regarding LRG-1 in a larger post-transplant patient cohort with subsequent transplant biopsies is warranted.
Figures
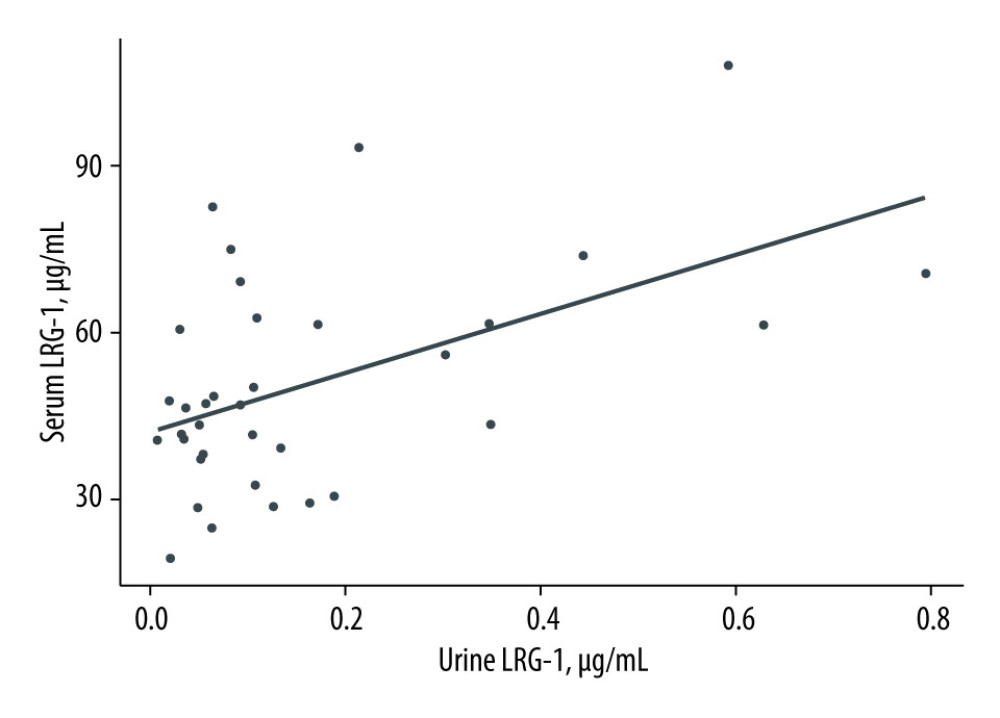 Figure 1. Linear regression analysis between serum and urine LRG-1. Every 1 ug/ml increase in urine LRG-1 was associated with a 53-units increase in serum LRG-1. Linear regression accounted for R2=25% of the variance of the serum LRG-1. Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria).
Figure 1. Linear regression analysis between serum and urine LRG-1. Every 1 ug/ml increase in urine LRG-1 was associated with a 53-units increase in serum LRG-1. Linear regression accounted for R2=25% of the variance of the serum LRG-1. Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria). 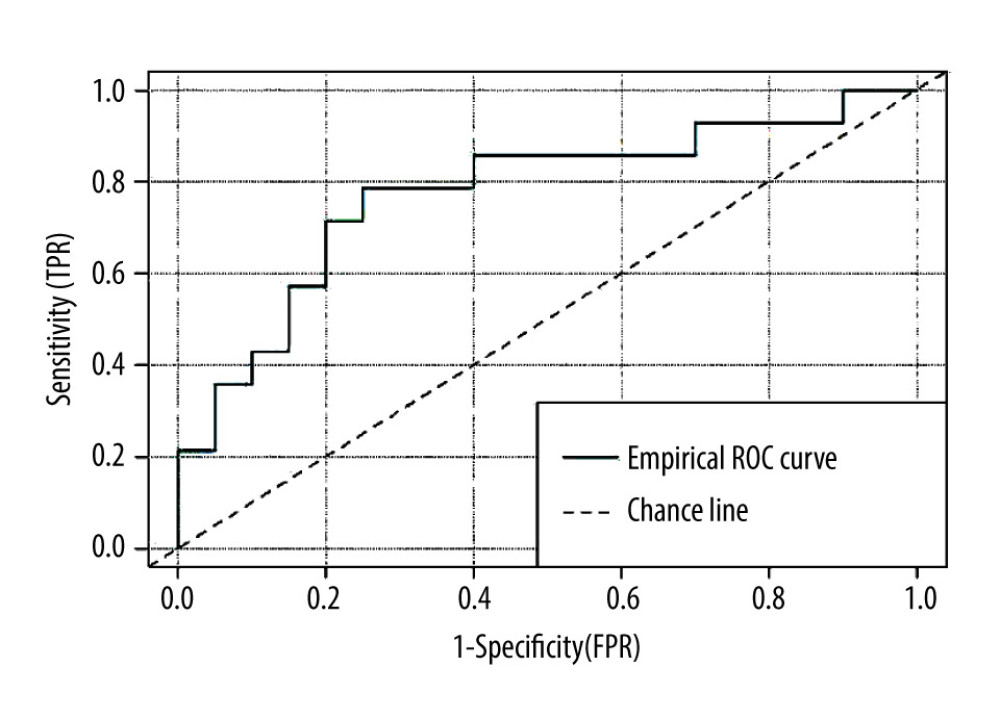 Figure 2. Receiver operating characteristic curve of urine LRG-1 for proteinuria detection. The diagnostic accuracy urine LRG− 1 for the diagnosis of proteinuria >0.5 mg/g was moderate (AUC, 0.77; 95% CI: 0.60–0.94). Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria).
Figure 2. Receiver operating characteristic curve of urine LRG-1 for proteinuria detection. The diagnostic accuracy urine LRG− 1 for the diagnosis of proteinuria >0.5 mg/g was moderate (AUC, 0.77; 95% CI: 0.60–0.94). Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria). References
1. Au E, Wong G, Chapman JR, Cancer in kidney transplant recipients: Nat Rev Nephrol, 2018; 14(8); 508-20
2. Neuberger JM, Bechstein WO, Kuypers DR, Practical recommendations for long-term management of modifiable risks in kidney and liver transplant recipients: A guidance report and clinical checklist by the consensus on managing modifiable risk in transplantation (COMMIT) group: Transplantation, 2017; 101(4S Suppl 2); S1-56
3. Shirai R, Hirano F, Ohkura N, Ikeda K, Inoue S, Up-regulation of the expression of leucine-rich alpha(2)-glycoprotein in hepatocytes by the mediators of acute-phase response: Biochem Biophys Res Commun, 2009; 382(4); 776-79
4. O’Donnell LC, Druhan LJ, Avalos BR, Molecular characterization and expression analysis of leucine-rich alpha2-glycoprotein, a novel marker of granulocytic differentiation: J Leukoc Biol, 2002; 72(3); 478-85
5. Lee H, Fujimoto M, Ohkawara T, Honda H, Leucine rich α-2 glycoprotein is a potential urinary biomarker for renal tubular injury: Biochem Biophys Res Commun, 2018; 498(4); 1045-51
6. Terada Y, Hirofumi N, Tatsuki M, Leucine rich α-2 glycoprotein is a novel urinary and serum biomarker for acute kidney injury: Nephrol Dial Transplant, 2019; 34(Suppl 1); FP285
7. Honda H, Fujimoto M, Miyamoto S, Sputum Leucine-rich alpha-2 glycoprotein as a marker of airway inflammation in asthma: PLoS One, 2016; 11(9); e0162672
8. Bini L, Magi B, Marzocchi B, Two-dimensional electrophoretic patterns of acute-phase human serum proteins in the course of bacterial and viral diseases: Electrophoresis, 1996; 17(3); 612-16
9. Chen JH, Chang YW, Yao CW, Plasma proteome of severe acute respiratory syndrome analyzed by two-dimensional gel electrophoresis and mass spectrometry: Proc Natl Acad Sci USA, 2004; 101(49); 17039-44
10. Fujimoto M, Serada S, Suzuki K, Brief report: leucine-rich α-2-glycoprotein as a potential biomarker for joint inflammation during anti-interleukin-6 biologic therapy in rheumatoid arthritis: Arthritis Rheumatol, 2015; 67(8); 2056-60
11. Zhong D, He G, Zhao S, LRG1 modulates invasion and migration of glioma cell lines through TGF-β signaling pathway: Acta Histochem, 2015; 117(6); 551-58
12. Zhang J, Zhu L, Fang J, LRG1 modulates epithelial-mesenchymal transition and angiogenesis in colorectal cancer via HIF-1α activation: J Exp Clin Cancer Res, 2016; 35(1); 29
13. Xie ZB, Zhang YF, Jin C, LRG-1 promotes pancreatic cancer growth and metastasis via modulation of the EGFR/P38 signaling: J Exp Clin Cancer Res, 2019; 38(1); 75
14. Wen SY, Zhang LN, Yang XM, LRG1 is an independent prognostic factor for endometrial carcinoma: Tumor Biol, 2014; 35(7); 7125-33
15. Fukamachi K, Hagiwara Y, Futakuchi M, Evaluation of a biomarker for the diagnosis of pancreas cancer using an animal model: J Toxicol Pathol, 2019; 32(3); 135-41
16. Serada S, Fujimoto M, Terabe F, Serum leucine-rich alpha-2 glycoprotein is a disease activity biomarker in ulcerative colitis: Inflamm Bowel Dis, 2012; 18(11); 2169-79
17. Ahn SS, Park Y, Jung SM, Serum leucine-rich α2-glycoprotein is elevated in patients with systemic lupus erythematosus and correlates with disease activity: Clin Chim Acta, 2018; 486; 253-58
18. Glorieux G, Mullen W, Duranton F, New insights in molecular mechanisms involved in chronic kidney disease using high-resolution plasma proteome analysis: Nephrol Dial Transplant, 2015; 30(11); 1842-52
19. Banon-Maneus E, Diekmann F, Carrascal M, Two-dimensional difference gel electrophoresis urinary proteomic profile in the search of nonimmune chronic allograft dysfunction biomarkers: Transplantation, 2010; 89(5); 548-58
20. Weivoda S, Andersen JD, Skogen A, ELISA for human serum leucine-rich alpha-2-glycoprotein-1 employing cytochrome c as the capturing ligand: J Immunol: Methods, 2008; 336(1); 22-29
21. Yang FJ, Hsieh CY, Shu KH, Plasma leucine-rich α-2-glycoprotein 1 predicts cardiovascular disease risk in end-stage renal disease: Sci Rep, 2020; 10(1); 5988
22. Liu JJ, Pek SLT, Ang K, Plasma leucine-rich α-2-glycoprotein 1 predicts rapid egfr decline and albuminuria progression in type 2 diabetes mellitus: J Clin Endocrinol Metab, 2017; 102(10); 3683-91
23. Neild GH: Chronic renal failure: The scientific basis of urology, 2004; 257-64, CRC Press
24. Nankivell BJ, Chapman JR, Chronic allograft nephropathy: Current concepts and future directions: Transplantation, 2006; 81(5); 643-54
25. Bonventre JV, Vaidya VS, Schmouder R, Next-generation biomarkers for detecting kidney toxicity: Nat Biotechnol, 2010; 28(5); 436-40
Figures
 Figure 1. Linear regression analysis between serum and urine LRG-1. Every 1 ug/ml increase in urine LRG-1 was associated with a 53-units increase in serum LRG-1. Linear regression accounted for R2=25% of the variance of the serum LRG-1. Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria).
Figure 1. Linear regression analysis between serum and urine LRG-1. Every 1 ug/ml increase in urine LRG-1 was associated with a 53-units increase in serum LRG-1. Linear regression accounted for R2=25% of the variance of the serum LRG-1. Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria). Figure 2. Receiver operating characteristic curve of urine LRG-1 for proteinuria detection. The diagnostic accuracy urine LRG− 1 for the diagnosis of proteinuria >0.5 mg/g was moderate (AUC, 0.77; 95% CI: 0.60–0.94). Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria).
Figure 2. Receiver operating characteristic curve of urine LRG-1 for proteinuria detection. The diagnostic accuracy urine LRG− 1 for the diagnosis of proteinuria >0.5 mg/g was moderate (AUC, 0.77; 95% CI: 0.60–0.94). Created using R-Studio (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria). Tables
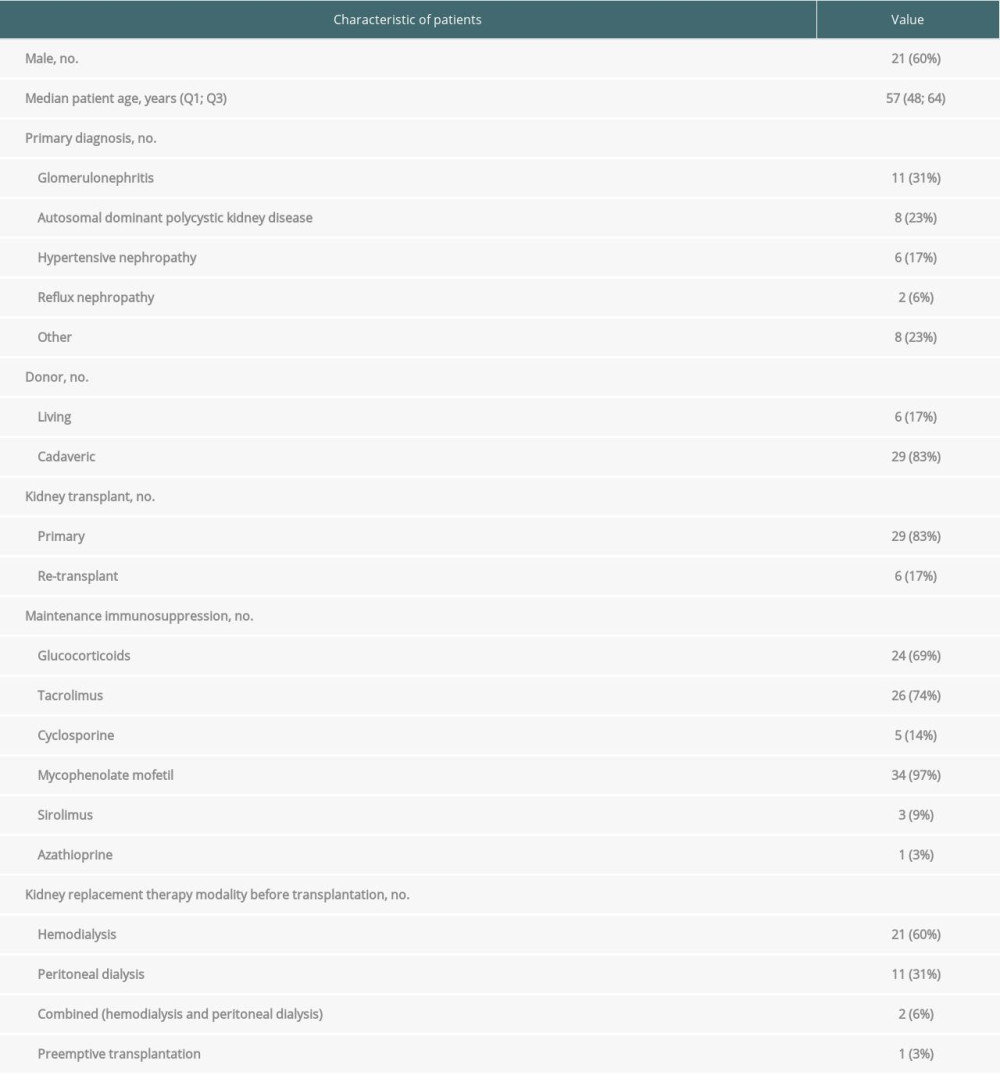 Table 1. Baseline patient characteristics.
Table 1. Baseline patient characteristics.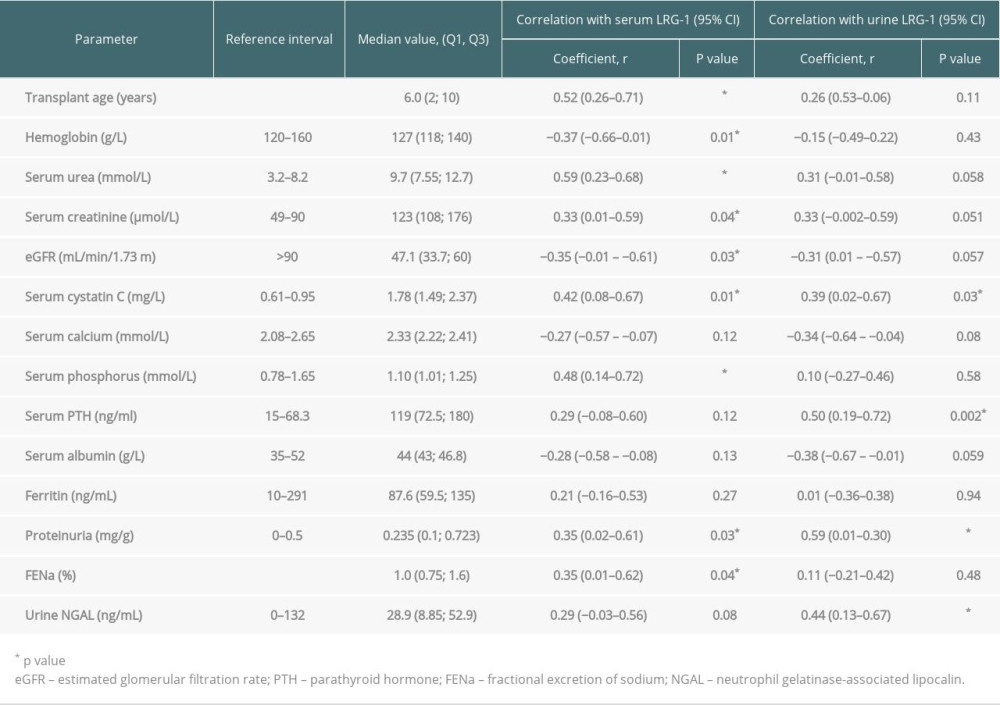 Table 2. Correlation between patient serum LRG-1, urine LRG-1, transplant age, and laboratory analysis results after adjustment for patient age.
Table 2. Correlation between patient serum LRG-1, urine LRG-1, transplant age, and laboratory analysis results after adjustment for patient age. Table 3. Diagnostic performance of urine LRG-1.
Table 3. Diagnostic performance of urine LRG-1. Table 1. Baseline patient characteristics.
Table 1. Baseline patient characteristics. Table 2. Correlation between patient serum LRG-1, urine LRG-1, transplant age, and laboratory analysis results after adjustment for patient age.
Table 2. Correlation between patient serum LRG-1, urine LRG-1, transplant age, and laboratory analysis results after adjustment for patient age. Table 3. Diagnostic performance of urine LRG-1.
Table 3. Diagnostic performance of urine LRG-1. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








