22 June 2021: Original Paper
Shift in Living Kidney Donor Demographics Over the Past 50 Years in a German Transplant Center
Claudia Sommerer1ACDEF*, Zoi Bougioukou1BCDEF, Vasileios L. Georgiou2CD, Arianeb Mehrabi3E, Martin Zeier1EDOI: 10.12659/AOT.929693
Ann Transplant 2021; 26:e929693
Abstract
BACKGROUND: Living kidney donors may face health risks after donation. Age, sex, body mass index, comorbidities, and relationship to the recipient have an impact on lifetime living kidney donor risk. In view of a changing landscape in renal transplantation with increasing organ shortages, the selection criteria for potential donors may have changed over time.
MATERIAL AND METHODS: We investigated donor demographics and outcomes in a cohort of 760 living kidney donors who donated from 1967 to 2016 at the transplant center in Heidelberg, Germany.
RESULTS: The living kidney donor age increased from 34.9±11.5 to 53.2±10.2 years, with 11.4% donors aged 65 years in the period from 2011 to 2016. The number of donors with comorbidities at the time of donation increased. The percentage of donors with a history of obesity, hypertension, smoking, and a family history of kidney disease enlarged to 18.6%, 36.1%, 37.0%, and 9.1%, respectively. De novo hypertension was a common problem in more than half of the donors at long-term follow-up, and donor renal function decreased about 30 mL/mi/1.73 m².
CONCLUSIONS: This detailed analysis of living kidney donor demographics over the last 50 years detected an increased proportion of donors with higher age and comorbidities today. Careful donor selection, regular follow-up visits, and systematic donor registries are required to further improve donor outcomes.
Keywords: Demography, Kidney Transplantation, Living Donors, Outcome Assessment (Health Care), Germany, Kidney Failure, Chronic, Nephrectomy, State Medicine, young adult
Background
Living donor kidney transplantation remains the optimal treatment option for patients with end-stage renal disease (ESRD), with excellent results in recipient and graft outcomes [1,2]. Due to organ shortage, the time on the transplant waiting list has increased in most countries, especially in Germany in the past years [3]. Living kidney donation is an opportunity to get early kidney transplantation, with the possibility of excellent renal function.
In Germany, the first renal allograft donations were performed in the late 1960s and early 1970s. Since then, the landscape of living kidney donors has changed. Especially in times of organ shortage, the number of people willing to donate to a family member or close friend increases, and the number of potential donors with comorbidities can also increase [4].
Large cohort studies have suggested that the risks of living kidney donation are minor [5–8]. Age, sex, body mass index (BMI), relationship to the recipient, and pre-existing comorbidities significantly alter the risk of donation [8]. Therefore, careful donor selection is necessary, and special medical care must be provided for living kidney donors in the short and long term after donation.
In the present study, we investigated whether living donor demographics at the time of donation has changed significantly over the past 50 years in a German transplant center to determine whether the proportion of donations from higher risk donors has changed.
Material and Methods
PATIENTS:
All living kidney donors from the Renal Transplant Center at the University Hospital of Heidelberg, Germany, from 1967 to 2016 were enrolled in this cohort study. Through reviewing published studies, we compared our patient cohort with that of other European transplant centers to determine if the characteristics of our cohort were representative of the those of donors from the other centers.
STUDY OBJECTIVES:
The main objective of the study was to analyze the change in age of living kidney donors within the last 50 years. We hypothesized that the mean age of recent donors is significantly higher than that of donors before 1990, since several elderly persons are willing to donate an organ to a family member or close friend. Secondary objectives were the shift in donor characteristics in regards to donor sex, BMI, blood pressure, hyperglycemia or prediabetes, albuminuria, hematuria, relationship to the recipient, and positive family history of renal disease or impaired renal function.
STUDY DESIGN:
Clinical data were obtained from the medical reports of yearly follow-up visits at the Department of Nephrology, Renal Center Heidelberg, Medical University Hospital of Heidelberg, Germany, as well as from outpatient clinic reports. Renal function was measured using serum creatinine levels and estimated glomerular filtration rates (eGFR), which were calculated by the Modification of Diet in Renal Disease (MDRD) or Chronic Kidney Disease Epidemiology Collaboration (CKDepi) formula [9,10].
The study was approved by the local institutional ethics review board (reference number S-104/2011, Ethics Committee University Hospital Heidelberg, Germany). All patient-related information was captured in medical confidentiality and according to the German Federal Data Protection Act. The study was conducted following the principles of the Declaration of Helsinki and in compliance with Good Clinical Practice guidelines.
STATISTICS:
The statistical analysis was conducted using SPSS version 24 (IBM Corp., Armonk, NY, USA), and R language version 3.5.3 (R Foundation for Statistical Computing, Vienna, Austria). Descriptive measures of the variables of interest are reported as frequencies and percentages for categorical variables and mean±standard deviation for continuous variables. The normality assumption of the data was evaluated 2 ways: visually using histograms and quantile-quantile plots and with the Shapiro-Wilk normality test. For the comparisons of mean and median values of variables across the different periods, when the normality assumption was met, the ANOVA test was used in cases of equal variances and the Welch test was used in cases of unequal variances. When the normality assumption was not met, the non-parametric Kruskal-Wallis test was used. For the analysis of categorical data, the chi-square test of independence was used. Additional post hoc analysis with Bonferroni correction was used for the pairwise comparison of proportions between the periods. To avoid losing important data, the maximum available number of observations per each analysis was utilized. In all hypothesis tests, a 5% significance level was used.
Results
STUDY POPULATION:
A total of 765 living kidney donations were performed from 1967 to 2016. Altogether, 760 donors with available baseline characteristics were analyzed. Data were analyzed for the whole study cohort and for 4 groups divided by the years (roughly, decades) of donation: 1967 through 1990, 1991 through 2000, 2001 through 2010, and 2011 through 2016, including 51, 103, 342, and 264 donors per period, respectively.
COMPARISON TO OTHER DONOR COHORTS:
The main donor demographic characteristics in 2001 and 2016 in our cohort were comparable to those of other cohorts in regards to sex distribution (60% female), median age of 50 years, and BMI of about 25 kg/m2 (Table 1). Only a very low proportion of donors had an eGFR <70 mL/min/1.73 m2, across all study cohorts. Among all cohorts, a mean of 62% of donors were biologically related to recipients (ranging from 49.1% in the United States to 79.9% in Norway). The percentage of smokers in our study was similar to that of the other cohorts, but the prevalence of hypertension was significantly higher in the present study than in the other cohorts.
PATIENT CHARACTERISTICS OVER TIME:
As hypothesized, donor age increased significantly from the periods 1967–1990 to 2011–2016, from 34.9±11.5 years to 53.2±10.2 years (Table 2, Figure 1A). The percentage of donors aged ≥65 years increased from 0% in the 1967–1990 period to 11.4% in the 2011–2016 period (P=0.01).
Altogether, 59.5% of donors were women, and the percentage of female donors decreased from 70.8% in the 1967–1990 period to 56.3% in the 2011–2016 period (
BMI increased from 22.9±3.4 kg/m2 in 1967–1990 to 26.6±7.6 kg/m2 in 2011–2016 (
One donor was an active smoker in the years 1967–1990, whereas 31% and 37% of donors were smokers during the periods of 2001–2010 and 2011–2016, respectively.
Regarding the donor-recipient relationship, most donations (about 70–80%) were to first-degree family members from 1967 to 2000; thereafter, the number of spouses donating a kidney increased.
Donors with a family background of kidney disease increased from 0% in 1967–1990 to 9.1% in 2011–2016 (
RENAL FUNCTION:
Renal function was assessed by serum creatinine levels and eGFR by MDRD or CKDepi. EGFR values increased significantly from the first to last periods (1967–1990 to 2011–2016) (Table 3A, Figure 1B). About 27% of the donors had a CKDepi eGFR <80 mL/min/1.73 m2 in 1991–2000, compared with 12.2% in 2011–2016 (P=0.005). The number of donors with microalbuminuria increased from 1.7% in the 1967–1990 period to 8.7% in the 2011–2016 period.
There was no significant difference in the prevalence of microhematuria, and only a few donors had a kidney biopsy performed before donation (1.7% in the 1967–1990 period to 4.8% in the 2001–2010 period).
HYPERTENSION AND METABOLIC PARAMETERS:
The prevalence of living kidney donors with hypertension before donation significantly within the last 50 years, from 0% in the 1967–1990 period to 36.1% in the 2011–2016 period (Table 3B). Correspondingly, blood pressure levels increased (Figure 1C). The number of antihypertensives taken also increased significantly, with up to 10.7% of the donors taking at least 2 antihypertensives in the 2011–2016 period. Blood glucose and cholesterol levels did not change significantly, with about 5.5% of the living donors having an impaired glucose tolerance test across all periods.
OUTCOME DATA:
The median length of follow-up was 106±77 months, depending on the year of donation. All patients had at least 24 months of follow-up. An increasing number of living kidney donors had regular follow-up visits at the transplant center, with 66.7% of donors having had external follow-up in the 1970s and 1980s, compared with 13.5% of donors in the last period of 2011–2016 (P<0.001, Table 4).
Serum creatinine levels and eGFR at discharge were similar throughout all time periods. However, serum creatinine at the last follow-up increased and eGFR decreased from 2001–2010 to 2011–2016 (Table 4, Figure 2). The percentage of living kidney donors with stage 3 or 4 CKD was low and comparable among all time periods. No ESRD was observed.
In the long-term donor group who donated in the year 2000 or earlier, up to 55.3% developed de novo hypertension. The mean HbA1c level was 5.59±0.42%, and the incidence of HbA1c ≥6.5% ranged from 0% in the first period to 2.7% in the last time period.
The mean incidence of cardiovascular events varied significantly, decreasing from 40% in the 1967–1990 period to 4.4% in the 2011–2016 period. The incidence of malignancy ranged from 4.8% in the 1967–1990 period to 11.9% in the 2011–2016 period.
Altogether, 22 (2.9%) donor deaths were recorded in the total observational period from 1967 to 2016. Malignancy (n=10) and cardiovascular events (n=7) were the main causes of donor deaths. All donor deaths are listed in Table 5.
Discussion
The present analysis provided comprehensive information on 760 living kidney donors prior to donation and data on long-term donor risk and death from a German transplant center. A significant increase in donor age has been observed over the past 50 years, with more than 11% of all donors today aged over 65 years at the time of donation. Our results showed that the percentage of patients with hypertension increased significantly to 36.1%, and 10.7% of all the most recent donors were taking 2 or more antihypertensive drugs. An increase in BMI was found in donors, with 18.6% having obesity, with a BMI ≥30 kg/m2 in the last period. Also, the prevalence of active smoking and family history for renal disease or impaired renal function increased. The distribution of sex was consistent among time periods, with women accounting for 60% of donors in all time periods. The number of donors with foreign residency was consistent, between 5.1% and 10.2%. In the last period, 8.7% of donors presented with microalbuminuria (about 10% had microhematuria, and 12.2% presented with a CKDepi eGFR <80 mL/min/1.73 m2).
Renal function at the long-term follow-up visit was slightly lower in the last period than in the earlier periods, but none of the patients had ESRD and 0.4% had stage 4 CKD. Cardiovascular events were identified in up to 40% of the early donors between 1967 and 1990, but in only 21.7% of the donors between 1991 and 2000, a period that had more detailed follow-up visits. At least 28 years after donation, 10% of donors had a malignancy, and >50% had de novo hypertension. Altogether, 2.9% of donors died across all time periods, with 7.8% of donors to date having died from the 1967–1990 period. The main reasons for death were cardiovascular diseases and malignancy. Fortunately, the quality of medical follow-up improved, with 86.5% donors between 2011 and 2016 accepting at least yearly follow-up visits at the transplant center.
Even in the United States, with large donor programs and about 5500 individuals becoming living donors each year, the mean donor age increased over the last decades, with 3% of donors aged ≥65 years and 32.2% of White donors aged >50 years [11,12]. In addition to the challenges associated with increased donor age, living kidney donation is challenged by comorbidities, as an increasing number of donors have hypertension, obesity, pre-existing cardiovascular diseases, and smoking habits. Similar to the results of our analysis, the number of donors at risk has also increased in the United States [11,12].
A similar trend in increased donor age was detected in a French study of their national database for living kidney donors, with 3483 donors [13]. The mean age as well as the proportion of donors aged ≥56 years increased significantly, from 24.8% in 2007 to 33.1% in 2017. The proportion of donors with obesity was consistent, and the quality of follow-up improved significantly, from 19.6% in 2007 to 42.5% of the donors in 2017 having adequate follow-up visits. In contrast to the studies in the United States [11,12], the authors of this French study concluded that the recent increase of 16% in the living kidney donation rate in France does not seem to be associated with the selection of high-risk donors.
In a recent analysis from the United Kingdom, living kidney donors’ baseline demographics were similar to those of our cohort [14]. An increase in donor age was limited to the White population. In addition, the proportion of donations to unrelated and non-partner recipients increased from 2006 to 2017. A small increase of donations to unrelated recipients was also observed in our cohort.
A somewhat different donor population profile was reported in a study from Norway [6], in which 2269 donors were registered from 1963 to 2007 at the living kidney transplantation center in Oslo, with 89 donors aged older than 70 years and 6 donors aged younger than 20 years, resulting in a younger mean donor age of 46±11.5 years after exclusion of “marginal” donors. A BMI ≥30 kg/m2 was observed in only 5.5% of the donors, and hypertension, identified as blood pressure > 140/90 mmHg or use of blood pressure medication, in only 4.7% of the donors. In total, 41 donors presented with an eGFR below 70 mL/min at the time of donation. The percentage of donors who were actively smoking was significantly higher at 41.5% than in our cohort at 32.9%. In a median follow-up time of 24.9 (0.1–26) years, 224 donors died, with 30.4% of deaths due to cardiovascular events.
Recently, living kidney data from the Australian and New Zealand Living Kidney Donor Registry, including all 3253 donors in Australia from 2004 to 2013 and in New Zealand from 2004 to 2012, were published [15]. Donor characteristics were nearly the same as those of the living kidney donors from 2000 to 2016 at our center, with 57% female donors and a median age at the time of donation of 50 (IQR 42–58) years, with 7.7% of donors ≥65 years old. The number of patients with obesity was 17%, similar to the 17.2% of patients with obesity in the present study. However, 2% of the donors had impaired glucose tolerance at the time of transplantation and 3 donors had diabetes mellitus (<1%). Self-reported hypertension (10.3%) was significantly lower than the rate of hypertension in the present study; however, hypertension was diagnosed by 24-h blood pressure measurement and/or antihypertensive treatment in our study, whereas hypertension in the referenced study was self-reported. The percentage of active smokers was 39%, which was similar to the 37% of active smokers in our study in the last time period, from 2011 to 2016. These data show that donor characteristics are consistent across different regions and contribute to donor risk and outcome.
The donor characteristics in this analysis at the Heidelberg transplant center were similar to those of the many German transplant centers that participated in the SOLKID study [16] and are therefore representative of most German transplant centers. Increased donor age, BMI, and number of comorbidities indicated that donor risk needs to be evaluated carefully. Previous studies have shown the overall safety of living kidney donors, even at an older age, but recovery of kidney function seems to occur more slowly for elderly donors [17,18].
The advantage of the present cohort study is that the comprehensive documentation of donor data and follow-up events was conducted in a mostly prospective method. Large donor registries have been established, such as the Organ Procurement and Transplantation Network database of the United Network of Organ Sharing (UNOS/OPTN National Registry). However, data sampling in registries is limited, and important or sufficient details of donor characteristics are often missing. In addition, follow-up is mostly limited to a short time: 2 years after transplantation in the United States’ transplant programs and 3 years in the current quality management program of Germany.
One of the main gaps in our knowledge of living donation is the ability to understand the long-term post-donation outcomes for living donors as they relate to the decade of donation. Striking variation exists concerning the follow-up of living donors. A study of donors with obesity in the United States showed that high-volume centers had the lowest probability of follow-up [19], with the acceptance of regular donor follow-up visits being a matter of debate for the transplant centers and the donors themselves. A survey of the transplant centers in the United States reported that only 68% of centers believed that follow-up with living kidney donors has a high priority. Although 92% reported that their center informed donors about the requirement for follow-up, only 67% had specific plans for achieving follow-up with donors [20]. Consensus conferences and recommendations have highlighted the importance of living donor follow-up to ensure donor safety [21,22]. In accordance with these recommendations, a follow-up rate of 86.5% was achieved at the Heidelberg transplant center in the last decade.
A national, or even international, registry for long-term monitoring of living donors is desired for each country. Currently, such a registry is lacking in many countries, including Germany. Even the observations of the present long-term single-center study strongly support the need for systematic renal and health monitoring and routine regular check-ups for living kidney donors at least on a national, but preferably on an international basis.
Conclusions
In conclusion, the present analysis provides an in-depth look at the shift of living kidney donor demographics over the last 50 years in a single German transplant center. Donor characteristics seem to be similar among most of the German transplant centers [23]. Age, BMI, and the number of comorbidities of living kidney donors have been increasing. Long-term kidney function in donors decreased slightly, whereas the proportion of donors with hypertension increased by more than 50%. Cardiovascular events and malignancies showed comparable incidences over time, between 4% and 12%, and represent the main causes of death in donors. The living kidney donors in this study were not an entirely healthy population and the proportion of donors at risk increased over time; however, with careful evaluation and follow-up, donor outcomes may be improved.
Figures
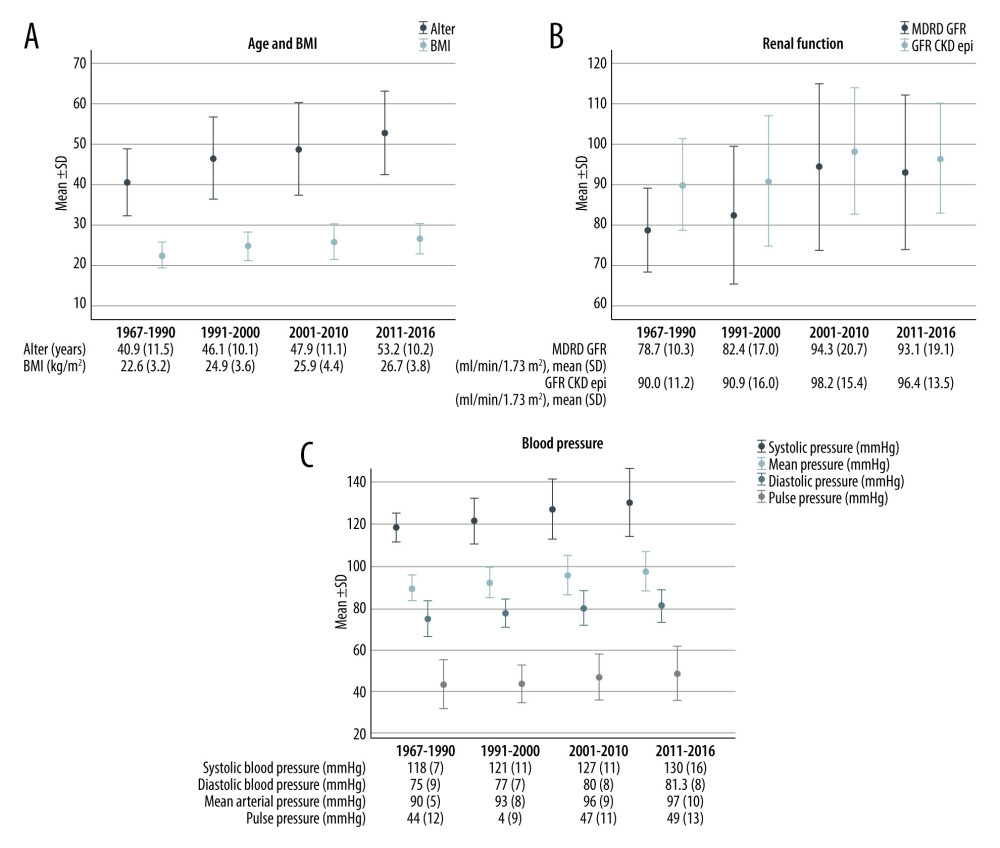 Figure 1. A shift in 3 main patient characteristics in living kidney donors from 1967 to 2016. (A) Age at donation; (B) renal function at donation (CKDepi eGFR); (C) patients with hypertension. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease.
Figure 1. A shift in 3 main patient characteristics in living kidney donors from 1967 to 2016. (A) Age at donation; (B) renal function at donation (CKDepi eGFR); (C) patients with hypertension. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease. 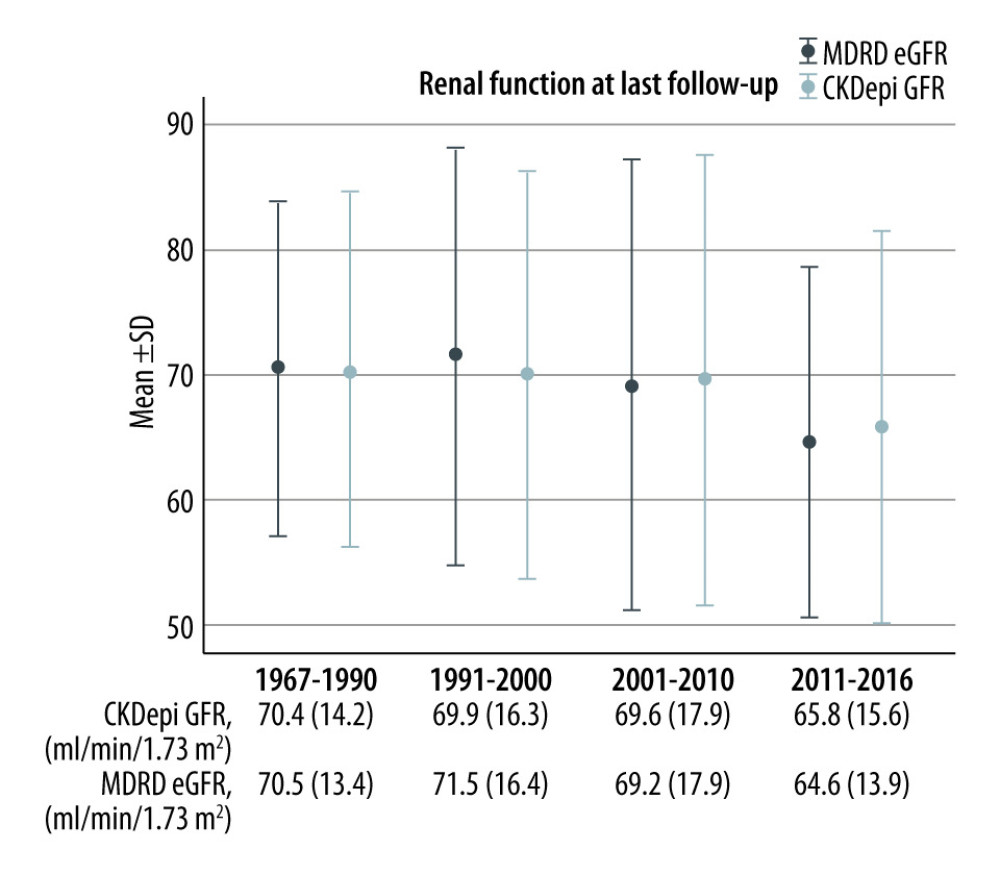 Figure 2. Renal function (CKDepi, MDRD eGFR) at last follow-up. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease.
Figure 2. Renal function (CKDepi, MDRD eGFR) at last follow-up. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease. References
1. Ibrahim HN, Foley R, Tan L, Long-term consequences of kidney donation: N Engl J Med, 2009; 360; 459-69
2. Mehrabi A, Wiesel M, Zeier M, Results of renal transplantation using kidneys harvested from living donors at the University of Heidelberg: Nephrol Dial Transplant, 2004; Suppl 4; iv48-54
3. Deutsche Stiftung Organtransplantation: Jahresbericht Organspende und Transplantation in Deutschland, 2019 [Cited 2020 June 1]. [in Germam]https://www.dso.de/SiteCollectionDocuments/DSO-Jahresbericht%202019.pdf
4. Textor SC, Medically complex living kidney donors: Where are we now?: Kidney Int Rep, 2019; 5; 4-6
5. Maggiore U, Budde K, Heemann U, Long-term risks of kidney living donation: Review and position paper by the ERA-EDTA Descartes Working group: Nephrol Dial Transplant, 2017; 32; 216-23
6. Mjøen G, Hallan S, Hartmann A, Long-term risks for kidney donors: Kidney Int, 2014; 86; 162-67
7. Muzaale AD, Massie AB, Wang M-C, Risk of end-stage renal disease following live kidney donation: JAMA, 2014; 311; 579-86
8. Grams ME, Sang Y, Levey ASChronic Kidney Disease Prognosis Consortium, Kidney-failure risk projection for the living kidney-donor candidate: N Engl J Med, 2016; 374; 411-21
9. Levey AS, Bosch JP, Lewis JB, A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of diet in renal disease study group: Ann Intern Med, 1999; 130; 461-70
10. Levey AS, Stevens LA, Schmid CHCKD-EPI (Chronic Kidney Disease Epidemiology Collaboration), A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12 Erratum in: Ann Intern Med. 2011; 155:408
11. Henderson ML, Thomas AG, Shaffer A, The national landscape of living kidney donor follow-up in the United States: Am J Transplant, 2017; 17; 3131-40
12. Al Ammary F, Bowring MG, Massie AB, The changing landscape of live kidney donation in the United States from 2005 to 2017: Am J Transplant, 2019; 19; 2614-21
13. Gaillard F, Jacquemont L, Roberts V, Temporal trends in living kidney donation in France between 2007 and 2017: Nephrol Dial Transplant, 2019; 36; 730-38
14. Bailey PK, Wong K, Robb M, Has the UK living kidney donor population changed over time? A cross-sectional descriptive analysis of the UK living donor registry between 2006 and 2017: BMJ Open, 2020; 10; e033906
15. De La Mata NL, Clayton PA, Kelly PJ, Survival in living kidney donors: An Australian and New Zealand cohort study using data linkage: Transplant Direct, 2020; 6(3); e533
16. Ebeling N, Suwelack B, Burgmer M, Gender disparity: Do female donors suffer from living kidney donation? [abstract]: Am J Transplant, 2019; 19(Suppl 3); B244
17. Segev DL, Muzaale AD, Caffo BS, Perioperative mortality and long-term survival following live kidney donation: JAMA, 2010; 303; 959-66
18. Minnee RC, Bemelman WA, Maartense S, Left or right kidney in hand-assisted donor nephrectomy? A randomized controlled trial: Transplantation, 2008; 85; 203-8
19. Reed RD, MacLennan PA, Shelton BA, Center variation and risk factors for failure to complete 6 month postdonation follow-up among obese living kidney donors: Transplantation, 2019; 103; 1450-56
20. Waterman AD, Dew MA, Davis CL, Living-donor follow-up attitudes and practices in U.S. kidney and liver donor programs: Transplantation, 2013; 95; 883-88
21. Lentine KL, Kasiske BL, Levey AS, Summary of kidney disease: Improving Global Outcomes (KDIGO) Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors: Transplantation, 2017; 101; 1783-92
22. Moore DR, Serur D, Rudow DL, Living donor kidney transplantation: improving efficiencies in live kidney donor evaluation – recommendations from a consensus conference: Clin J Am Soc Nephrol, 2015; 10; 1678-86
23. Eurotransplant International Foundation: Eurotransplant Statistics Report Library [Cited 2020 September 5]https://statistics.eurotransplant.org/index.php?search_type=transplants+%28living+donor%29&search_organ=&search_region=All+ET&search_period=by+year&search_characteristic=&search_text=
Figures
 Figure 1. A shift in 3 main patient characteristics in living kidney donors from 1967 to 2016. (A) Age at donation; (B) renal function at donation (CKDepi eGFR); (C) patients with hypertension. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease.
Figure 1. A shift in 3 main patient characteristics in living kidney donors from 1967 to 2016. (A) Age at donation; (B) renal function at donation (CKDepi eGFR); (C) patients with hypertension. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease. Figure 2. Renal function (CKDepi, MDRD eGFR) at last follow-up. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease.
Figure 2. Renal function (CKDepi, MDRD eGFR) at last follow-up. CKDepi – Chronic Kidney Disease Epidemiology Collaboration; eGFR – estimated glomerular filtration rate; MDRD – Modification of Diet in Renal Disease. Tables
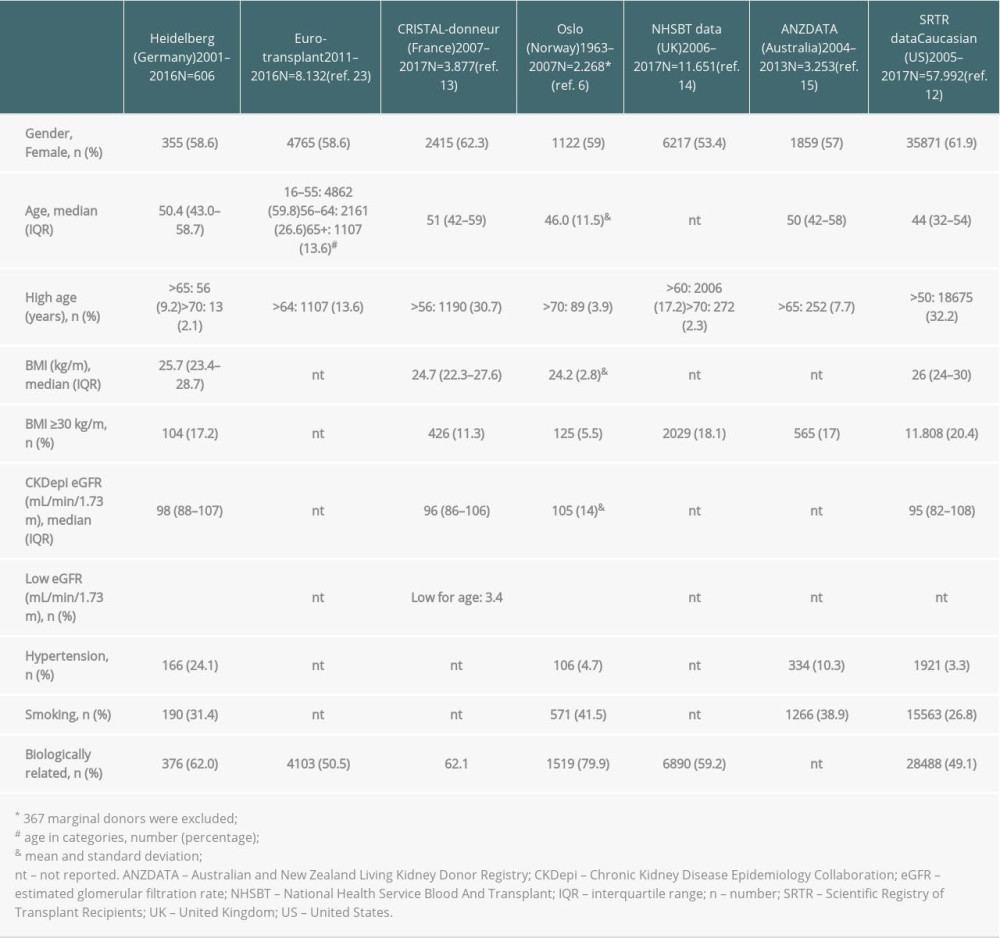 Table 1. Demographics of living kidney donors from different countries.
Table 1. Demographics of living kidney donors from different countries.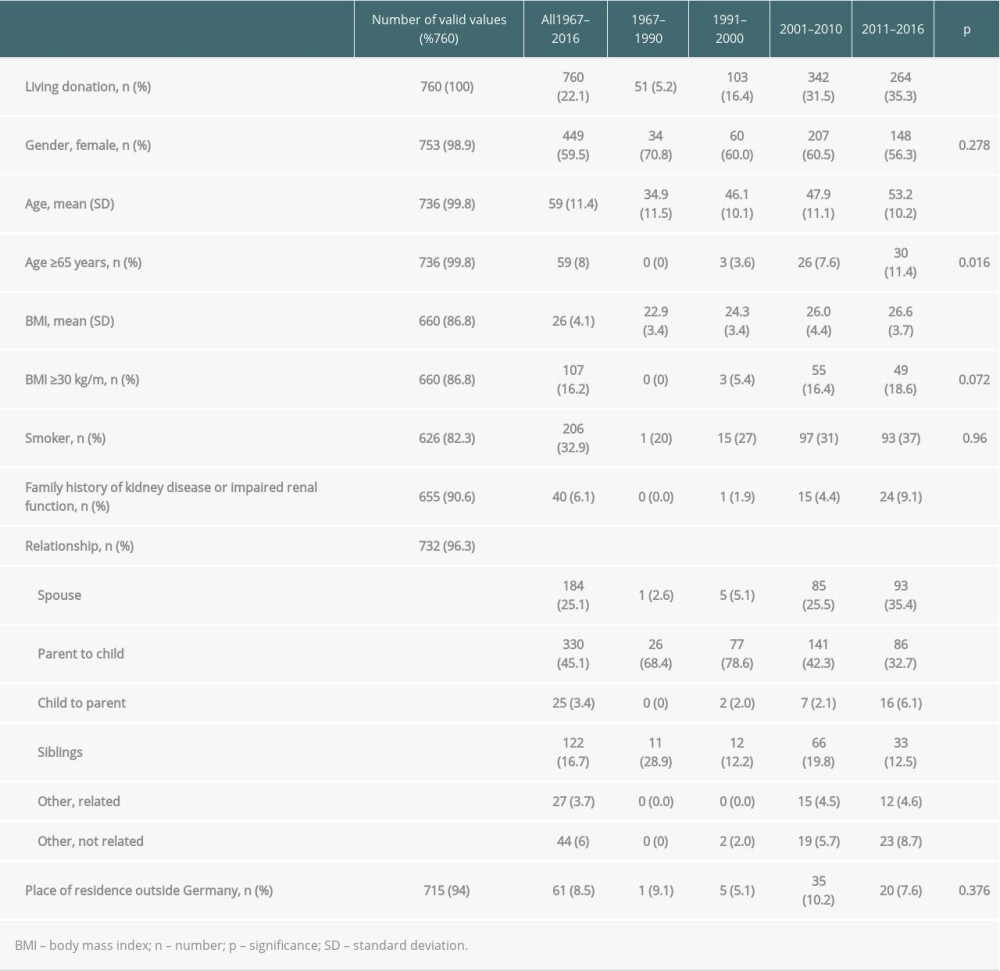 Table 2. Demographics of living kidney donors from 1967 to 2016.
Table 2. Demographics of living kidney donors from 1967 to 2016.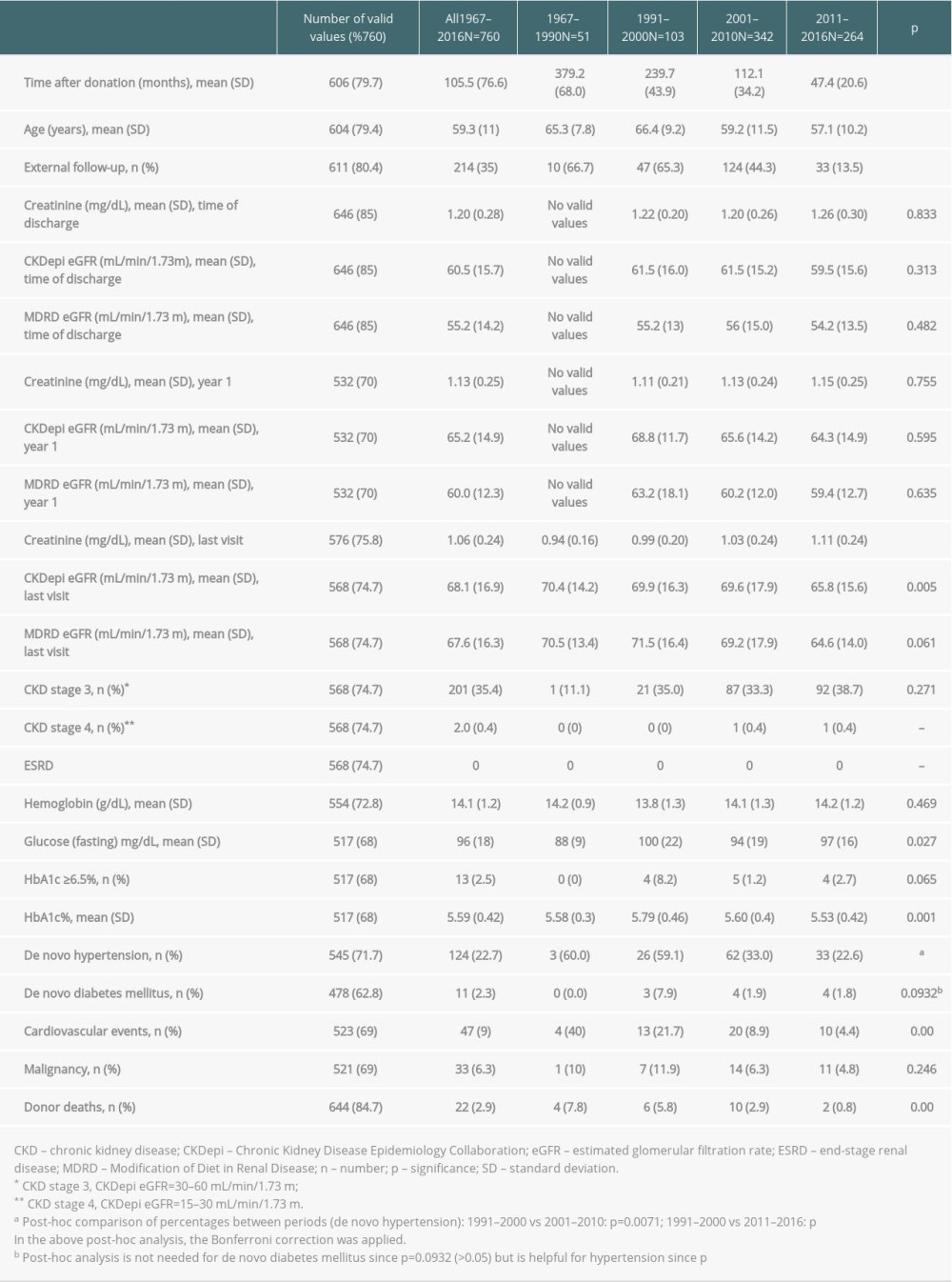 Table 4. Renal function and outcome data of living kidney donors from 1967 to 2016.
Table 4. Renal function and outcome data of living kidney donors from 1967 to 2016.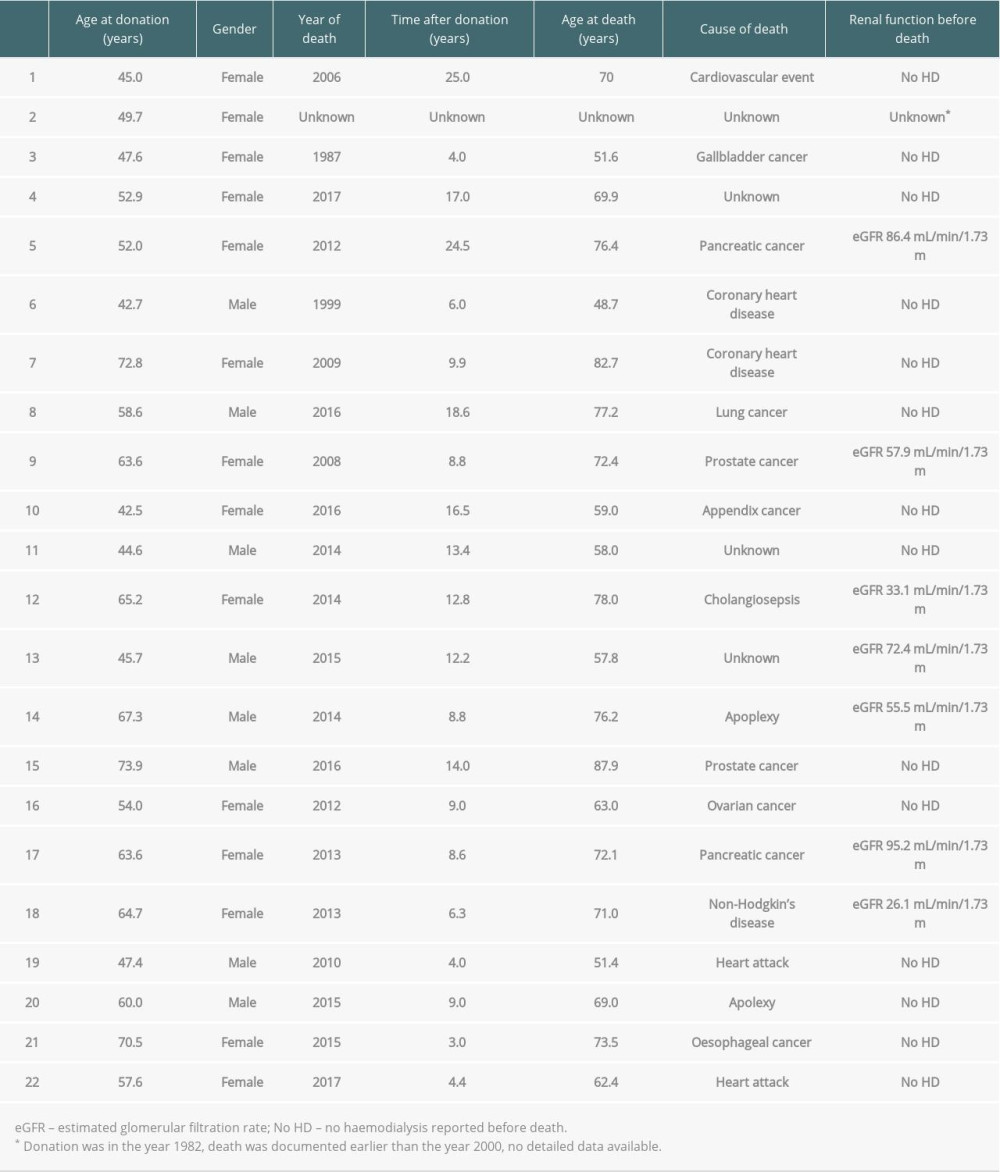 Table 5. Description of donor deaths.
Table 5. Description of donor deaths. Table 1. Demographics of living kidney donors from different countries.
Table 1. Demographics of living kidney donors from different countries. Table 2. Demographics of living kidney donors from 1967 to 2016.
Table 2. Demographics of living kidney donors from 1967 to 2016. Table 4. Renal function and outcome data of living kidney donors from 1967 to 2016.
Table 4. Renal function and outcome data of living kidney donors from 1967 to 2016. Table 5. Description of donor deaths.
Table 5. Description of donor deaths. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








