09 November 2021: Original Paper
Type 1 Diabetic Patients After Simultaneous Pancreas and Kidney Transplantation Have Less Intense Periodontal Inflammation Compared to Kidney Recipients Treated with Insulin
Mikołaj Musiał1ABCDEF*, Rafał Wiench2ADEF, Aureliusz Kolonko3BD, Piotr Choręza4CD, Elżbieta Świętochowska5BC, Damian Niedzielski6BF, Agnieszka Machorowska-Pieniążęk1DG, Dariusz Skaba2DE, Andrzej Więcek3DE, Aleksander J. Owczarek7CD, Iwona Niedzielska6AD, Robert Król8DE, Jacek Ziaja8ADEFDOI: 10.12659/AOT.932426
Ann Transplant 2021; 26:e932426
Abstract
BACKGROUND: Long-term diabetes predisposes to pathological changes in periodontal tissues. Improvement in this respect can be expected in patients after pancreas transplantation. The aim of this study was to assess and compare the intensity of periodontium pathological lesions and inflammation markers concentration in gingival crevicular fluid (GCF) in patients with type 1 diabetes (T1D) after kidney (KTx) or simultaneous pancreas and kidney transplantation (SPK).
MATERIAL AND METHODS: The study included 20 T1D patients after SPK and 16 after KTx, and 15 non-diabetic kidney recipients (control). Periodontal clinical parameters and concentration of selected biochemical markers of inflammation in GCF were assessed. The following tests were used in statistical data analysis: Shapiro-Wilk test, the t test, the Mann-Whitney U tests, one-way ANOVA with Tukey’s post hoc test, and χ² test (also with Yate’s correction). Moreover, linear regression and Pearson or Spearman correlation coefficient was used.
RESULTS: There were no differences in modified Sulcus Bleeding Index (mSBI) and GCF volume between the SPK group and control group, whereas values of these parameters in the KTx group were higher than in the SPK and control groups. Maximal clinical attachment loss and pocket depth and Periotest values were higher in diabetic recipients compared to controls, and did not differ between SPK and KTx. The concentration of IL-1β, MMP-8, resistin, TNFα, and YKL40 in the GCF in the KTx group was higher than in the SPK and control groups. In the combined group of T1D patients, there was a correlation between blood HbA1c and mSBI, GCF volume, and resistin, TNF-α and YKL40 concentrations, and between resistin concentration and mSBI.
CONCLUSIONS: T1D patients after SPK show lower levels of inflammatory markers in GCF and present reduced intensity of periodontitis compared to kidney recipients treated with insulin. The severity of morphological changes in periodontium in T1D patients after KTx or SPK is higher than in non-diabetic kidney recipients.
Keywords: Diabetes Mellitus, Type 1, Gingival Crevicular Fluid, Pancreas Transplantation, periodontal diseases, Humans, Inflammation, Insulin, Kidney, Kidney Transplantation, Pancreas
Background
Long-term type 1 diabetes mellitus (T1D) causes a number of pathologies in the cardiovascular system [1], nervous system, kidneys [2] and the retina of the eye [3]. In the oral cavity, long-term consequences of T1D include impaired salivation, infections, caries [4], and periodontal disease [5].
Pathology in the periodontium consists of inflammatory state related to a response of monocytes, macrophages, and neutrophils to bacterial biofilm accumulated in the gingival crevice [6]. These cells secrete a number of proinflammatory cytokines, mainly IL-1β and TNF-α, as well as metalloproteinases (MMP), mainly MMP-8, resistin, and YKL-40 [7–10]. The release of these cytokines at the site of periodontal infection causes breakdown of collagen fibers of the periodontal ligament, formation and deepening of the periodontal socket, resorption of alveolar bone and, consequently, progressive loss of connective tissue attachment [5]. Clinically, periodontitis results in a higher percentage of gum swelling and bleeding, increased tooth mobility, and, ultimately, loss of teeth [11]. Loss of dentition affects the ability to chew food and, consequently, eating habits and nutrient intake [12].
In diabetic patients, periodontal disease occurs more frequently than in the general population, and the described changes develop faster and with greater intensity compared to patients without diabetes [5,13]. This is due, in part, to the impaired bactericidal functions of polymorphonuclear leukocytes, and in particular their reduced chemotaxic and phagocytic capacity [14]. The inflammatory response to bacterial plaque build-up occurs earlier and is usually more severe [15]; in patients with T1D, monocytes produce more proinflammatory cytokines than in patients without diabetes [16], and their elevated level can be measured in gingival crevicular fluid (GCF) [17]. The processes of tooth suspension apparatus destruction in diabetic patients are also associated with faster collagen degradation caused by a higher concentration of matrix metalloproteinases [18].
In many patients, long-term T1D leads to impaired renal function and, consequently, need for dialysis treatment [2,19]. Regardless of the cause of renal failure, periodontal disease occurs more frequently and with greater intensity in dialysis patients [20,21].
As in diabetes, the development of periodontitis in patients with end-stage renal disease (ESRD) is favored by an impaired immune system response against pathogens, and by systemic inflammation [21]. Patients on dialysis are more likely to have dry mouth, which leads to increased accumulation of dental plaque [22]. Also, renal osteodystrophy, which occurs in dialysis patients, leads to atrophy of the alveolar bone and thus contributes to damage of the tooth suspension apparatus [23].
Kidney transplantation is the treatment of choice in dialysis patients, and in patients with ESRD in the course of T1D it can be supplemented with pancreatic transplantation [24–26].
Patients after pancreas and kidney transplantation tend to have slowing of the process of coronary and intracerebral arteries damage, improvement in lipid profile [27], partial improvement in polyneuropathy [28], stabilization of retinopathy [29], and a marked improvement in quality of life [30] compared to T1D kidney recipients. In patients undergoing simultaneous pancreas-kidney transplantation (SPK), an improvement in some parameters of the oral cavity, such as salivation and the incidence of fungal infections, was observed compared to patients with T1D after kidney transplantation [31].
Bearing in mind the above-mentioned facts, one can expect that the normoglycemia achieved by pancreas transplantation will also lead to a reduction in severity of the inflammatory process in periodontal tissues and, consequently, improvement in the periodontium morphology.
The aim of the present study was to analyze the intensity of pathological lesions within the periodontium and the concentration of inflammation markers in gingival crevicular fluid in type 1 diabetic patients after kidney or pancreas and kidney transplantation.
Material and Methods
STUDY GROUPS:
Two groups of patients with ESRD caused by long-lasting T1D (diagnosed in childhood or adolescence) were enrolled into the study [31]:
The control group consisted of 15 patients with ESRD without diabetes mellitus as a cause of kidney disease or new-onset diabetes mellitus after transplantation. The minimal post-transplant follow-up period was 12 months. The matching procedure included recipient sex, age at the time of transplantation and at the time of study, duration of dialysis vintage, and body mass index (BMI). All organs for transplantation were procured from deceased, brain-dead donors.
Transplanted kidneys were placed extraperitoneally in the iliac fossa with standard anastomosis of graft vessels and ureter. Simultaneously, the transplanted pancreas was procured from the same kidney donor and it was placed intraperitoneally with blood supply from the donor common iliac artery, venous blood drainage into the vena cava inferior, and pancreatic juice drainage into the loop of the jejunum.
We excluded patients with a pre-emptive, living-donor, or second/third KTx, with significantly impaired kidney graft function (estimated glomerular filtration rate [eGFR] ≤30 ml/min/1.73 m2), bacterial infection within 1 month before the study, diagnosis of cancer or liver cirrhosis, and noncompliance. Also, patients after SPK who lost pancreatic graft function less than 1 year prior to the study were not included.
The exclusion criteria concerning the oral cavity included number of teeth less than 8, prosthetic restoration, oral mucosa lesions outside the marginal gingiva, precancerous and cancerous lesions, and pathological lesions in the maxillary/mandibular bone other than periodontal disease confirmed by radiological examination.
Clinical data and results of blood tests came from a Kidney Transplantation Database maintained by the transplant center in Katowice, Poland.
The comparative analysis of clinical parameters included sex, age at the time of transplantation and at the time of study, BMI, T1D duration (at the time of study and total), dialysis vintage and follow-up period, and medication with immunosuppressive agents such as calcineurin inhibitors (Tacrolimus or Cyclosporine A), antimetabolites (Mofetil/Sodium Mycophenolate or Azathioprine), and steroids, blood glycated hemoglobin concentration (HbA1c), serum creatinine concentrations, and eGFR values calculated using the MDRD (Modification of Diet in Renal Disease) short formula.
PERIODONTAL LESIONS ASSESSMENT:
Periodontal clinical parameters measured in all teeth, measured with the UNC 15 periodontal probe (American Eagle Instruments, Inc., Missoula, MT, USA), were determined in all enrolled patients.
The mobility of the teeth was assessed in all patients using the Periotest device (Periotest Medizintechnik Gulden e. K, Modautal, Germany). The Periotest device is placed horizontally at a distance of 0.6–2.0 mm from the tooth surface. With each measurement, the device performs 16 blows per tooth within 4 s.
The contact time of the accelerated head, moving along the impact path, with the tooth is converted into the Periotest value (PTV). The obtained results are displayed on the screen of the device. The Periotest value range is −8 to +50. The lower the value, the higher the stabilization of the tested object. Periotest values correlate with the four-level classification of tooth mobility according to Miller. The scale correlates with Miller’s index, with Periotest value (PTV) −8 to +9 being no movement distinguishable, PTV +10 to +19 first distinguishable sign of mobility, PTV +20 to +29 crown deviates within 1 mm of its normal position, and PTV +30 to +50 mobility is easily noticeable [34].
Subsequently, the volume of gingival cervicular fluid (GCF) was measured using the Periotron 8000® device (Pro-Flow Inc, Amityville, NY, USA). The volume in each patient was measured twice in the deepest gingival pocket. The GCF was collected by inserting Periopaper paper strips (OraFlow, Inc, Hewlett, NY, USA) into the periodontal pocket to 1 mm depth without saliva and blood contamination. The strips were left in situ for 30 s and then transferred for volume determination with an electronic device (Periotron 8000). The readings were converted to actual volume (μl) by reference to the standard curve.
ANALYSIS OF THE CONCENTRATION OF INFLAMMATORY MARKERS IN GINGIVAL CREVICULAR FLUID:
To assess the concentration of inflammatory markers in the gingival crevicular fluid, immediately after reading the GFC volume, the absorption strips were placed in an Eppendorf tube with 100 μl phosphate-buffered saline (PBS, pH 7.2). The liquid from the strips was eluted using a refrigerated centrifuge (10 min, 10 000 g at 4°C), and the strips from the obtained samples were removed. The samples prepared in this way were encoded and stored at −70°C for further analysis [35,36].
The concentration of analyzed biochemical parameters in gingival crevicular fluid was determined by the enzyme immunoassay method (interleukin-1β [IL-1β], matrix metalloproteinase-8 [MMP-8], and resistin using BioVendor – Laboratorní medicína a.s., Czechia test, cat. No. RD 194559200R, RD 191016100, and RD 191016100); tumor necrosis factor-α (TNF-α) using Thermo Fisher Scientific, USA, test (cat. No. BMS223HS); and YKL40 using YKL40 ELISA kit, QUIDEL, Quidel Corporation, OH, USA (test cat. No. 088337) according to the manufacturer’s instructions. To determine the concentrations of the samples tested, a calibration curve was prepared using the standards contained in the kit.
Absorbance readings were performed using the Universal Microplate Spectrophotometry μQUANT BIO-TEK INC device (Bio-Tek World Headquarters, CA, USA): for IL-1β, MMP-8, and resistin at 450 nm and 630 nm reference wavelength; for TNF-α at 450 and 610 nm reference wavelength; and for YKL40 at 570 nm wavelength. The results were processed using the KCJunior computer program (KC Junior v1.41.8, Bio-Tek Instruments, Winooski, VT, USA).
The sensitivity of the set, intra-assay variation, and inter-assay variation were: for IL-1β – 0.4 pg/ml, 4.2% and 6.7%; for an MMP-8 – 0.025 ng/ml, 3.6%, and 8.1%; for resistin – 0.012 ng/ml, 5.2%, and 7.0%; for TNF-α – 0.13 pg/ml, 8.5%, and 9.8%; and for YKL40 – 0.4 pg/ml, 7.1%, and 8.4%, respectively.
This study is a part of the research project: “Evaluation of the oral status, oral hygiene, and clinical periodontal indicators, including saliva parameters and mycological examination, in long-term follow-up of patients with end-stage renal desease in the course of type 1 diabetes after simultaneous kidney and pancreas transplantation in comparison with patients after kidney transplantation” [31].
The study protocol was approved by Bioethics Committee of the Medical University of Silesia in Katowice (KNW/0022/KB1/105/18), and each participant gave informed consent.
STATISTICAL ANALYSIS:
As previously described, nominal and ordinal data are expressed as percentages, while interval data are expressed as mean±standard deviation in the case of normal distribution or as median (lower quartile-upper quartile) in the case of data with skewed or non-normal distribution [31]. Nominal and ordinal data are presented as counts and rates.
The Shapiro-Wilk test and a quantile-quantile plot were used to estimate distribution of variables. Interval data with normal distribution are presented as mean±standard deviation, and these with skewed or non-normal distribution as median (lower and upper quartiles).
The
One-way ANOVA with Tukey’s post hoc test was applied to evaluate the relationships among 3 groups with normal data distribution, and Kruskal-Wallis one-way ANOVA and the median test as post hoc test were used in other cases. Homogeneity of variances was checked with the Fisher-Snedecor
Stepwise backward linear regression was used to assess correlations between clinical and biochemical parameters. The associations between variables were assessed with the Pearson or Spearman correlation coefficient, respectively. All tests were two-tailed.
Results
CLINICAL AND BIOCHEMICAL CHARACTERISTICS, AND IMMUNOSUPPRESSIVE TREATMENT:
As assumed, the analyzed groups of patients did not differ in terms of sex (P=0.41), age at the time of transplantation (P=0.21), or at the time of study (P=0.46), BMI (P=0.19), rate of overweight patients (P=0.19), and duration of dialysis therapy prior to transplantation (P=0.58) (Table 1).
The duration of diabetes prior to transplantation was 4.5 years longer in the SPK group compared to the KTx group (
The follow-up duration after transplantation was similar in SPK and KTx groups, and was only longer in the control group compared to the SPK group (
Analyzed groups of patients did not differ as regards kidney graft function, and in terms of immunosuppressive treatment, only patients in the control group presented lower rates of tacrolimus administration (P<0.05) (Table 1).
SELECTED PARAMETERS OF PERIODONTAL LESIONS:
Maximal clinical attachment loss, maximal pocket depth, and tooth mobility presented as Periotest values were higher in both groups of diabetic recipients compared to the control group (P<0.001, P<0.001, P<0.05, P<0.01), and did not differ between SPK and KTx groups (P=1.00, P=1.00, P=0.99) (Table 2, Figure 1).
Interestingly, there was no significant difference in modified Sulcus Bleeding Index and gingival crevicular fluid volume between the SPK group and control group (
CONCENTRATION OF ANALYZED PARAMETERS IN GINGIVAL CREVICULAR FLUID:
The concentration of all analyzed inflammation markers was lower in the SPK group than in the group of T1D patients after KTx (P<0.001) (Table 2, Figure 2).
The level of all the markers in the KTx group was higher than in the control group, whereas in the SPK group concentrations of some analyzed markers were also higher, and the others were comparable to controls (Table 2).
SUB-ANALYSIS OF TYPE 1 DIABETIC PATIENTS:
In the combined group of T1D patients after SPK or KTx, univariate analysis revealed that gingival crevicular fluid volume was correlated with blood HbA1c (β=0.340, P<0.05) and serum creatinine concentration (β=0.346, P<0.05), the value of modified Sulcus Bleeding Index was correlated with blood HbA1c (β=0.439, P<0.05), and maximal clinical attachment loss and tooth mobility was correlated with patient age at the time of study (β=0.348, P<0.05) (Table 3).
The same analysis also revealed that blood HbA1c was correlated with concentration of a row of inflammatory markers (resistin, TNFα [β=0.340, p<0.05], and YKL40 [β=0.475, P<0.05]) measured in GCF, and some of them were also influenced by total diabetes duration ([TNFα [β=0.335, P<0.05] and YKL40 [β=0.471, p<0.05]) and serum creatinine concentration (YKL40) (β=0.298, P<0.05) (Table 3).
We also found that resistin concentration in GCF was correlated with modified Sulcus Bleeding Index (β=0.394, P<0.05, Figure 3), but there were no other correlations between periodontal parameters and concentration of analyzed markers in GCF in the combined group of patients with ESRD in the course of T1D after SPK or KTx.
The multivariable stepwise best model analysis included the following independent factors: SPK group, log10 of total diabetes duration, age at the time of study, log10 blood HbA1c level, and serum creatinine concentration, as well as TNF-α, IL-1β, MMP-8, resistin, and YKL40 concentrations in gingival crevicular fluid, showing that high resistin level was associated with a higher modified Sulcus Bleeding Index (β=1.9622; 95% CI: 0.4369–3.4876;
The analysis confirmed also association of age at the time of study with log10 of tooth mobility (β=0.0153; 95% CI: 0.0049–0.0257;
Discussion
The most important finding in our study is that, in kidney recipients with T1D who successfully underwent simultaneous pancreas transplantation, lower concentration of resistin in GCF was associated with the severity of inflammation in periodontal tissue, expressed by decreased modified Sulcus Bleeding Index.
Diabetic patients with uncontrolled glycemia, including these with T1D, present features of intense periodontal inflammation with severe morphological changes, which can be confirmed by clinical examination. Two tests that can be applied in diagnosis of periodontitis or to determine the severity of periodontal inflammation are the modified Sulcus Bleeding Index (mSBI), which gives the rate of gingival bleeding sites during probing [33], and measurement of GCF volume, which increases with the increase of blood vessel walls permeability as a consequence of inflammatory mediator activity [37].
Our study revealed that the values of these parameters were comparable in patients after SPK and in controls, and lower than in patients with T1D after kidney transplantation. In T1D kidney recipients, these parameters were 2–4 times higher than in the control group. Moreover, univariate regression analysis showed a positive relationship between blood HbA1c concentration and the mSBI and GCF volume, wherein these relationships were not confirmed in multivariate regression analysis.
It is difficult to compare our results to the literature data due to the lack of studies on periodontal pathology in patients after pancreas transplantation. However, there are sparse reports confirming the differences in the scope of the analyzed clinical parameters between diabetic patients with adequate and poor glycemic control. Meenwat et al, comparing diabetic patients with poor and acceptable (moderate or good) glycemic control, found significant differences in gingival index, which corresponded to the mSBI index, in favor of those with normal glycaemia [38]. Lim et al revealed that subjects with acceptable glycemic control (HbA1c <8%) were characterized by lower percentage of sites with bleeding on probing, a parameter similar to mSBI, compared to subjects with insufficient glycemic control [39]. Also, Marinho et al showed that GFC volume was higher in patients with T2D and periodontal disease compared to healthy patients [40]. Of note, contrary to previous reports, Engebretson et al did not show a significant relationship between GFC volume and diabetes, but in their study the GFC volume was lower in diabetic patients compared to non-diabetics [41].
Severity of the inflammatory process in the periodontium can by confirmed not only with instrumental measurements, but also with biochemical analysis of GCF samples [5,17]. Being a vascular filtrate, GCF reflects the intensity of the response of periodontal cells and tissues aiming to regain homeostasis by not only increasing its volume, but also in changes of its composition, particularly the concentration of inflammatory mediators [42,43].
In the studies of Yücel et al [44] and Goutoudi et al [45], patients with periodontitis had a significantly higher total amount of IL-1β in their GFC compared to the control group without periodontitis. McCrudden et al showed significantly higher MMP-8 activity in GFC from sites with periodontal disease compared to healthy sites that showed basal or no MMP-8 activity [46]. Mittal et al found that the level of resistin in GCF was high in patients with periodontal disease and low in healthy patients [47]. Heralgi et al observed that patients with periodontitis had higher mean TNF-α levels in GFC compared to a healthy group [48]. In a study by Kranti et al, the mean amount of YKL-40 in the GCF samples from the sites of chronic periodontitis was about 4.5 times higher than in the healthy sites [49].
In our study, the improvement in clinical parameters was to some degree confirmed by results of biochemical analysis of GCF. In the SPK group, lower values of all analyzed markers in GCF were observed compared to the KTx group, and in some of them they were comparable to non-diabetic kidney recipients. As in the case of the clinical parameters, the results of the biochemical analysis can only be related to studies on inflammatory markers concentrations in GCF in diabetic patients with well-controlled or uncontrolled glycemia. Engebretson et al showed that patients with HbA1c above 8% had significantly higher levels of IL-1β in GCF compared to patients with HbA1c below 8% [50]. In a study by Kranti et al, T2D patients with periodontitis showed almost 2 times higher levels of YKL-40 in GCF samples from sites of chronic periodontitis compared to patients with periodontitis only [49]. On the contrary, Longo et al observed no differences in TNF-α concentrations in GCF in patients with periodontitis and without T2D, with T2DM with adequate glycemic control, and with T2DM with inadequate glycemic control [51].
The univariate regression analysis conducted in our study revealed a relationship between blood HbA1c level and concentration of several markers of inflammation measured in GCF: resistin, TNF-α, and YKL-40. A similar correlation was observed in a multistage regression analysis by Gokhale et al, who demonstrated the relationship between resistin concentration in GCF and HbA1c combined group of healthy patients, patients with periodontal disease, patients with T2D, and patients with T2D and periodontal disease [52]. The results of this study are also confirmed by Chang-chun et al, who proved that IL-1β level in GCF was correlated with blood HbA1c [53].
The only significant relationship between clinical parameters and concentration of inflammatory markers measured in GCF, confirmed in both univariate and multivariate regression analysis, was the correlation between levels of resistin in GCF and mSBI. The finding is comparable to other authors’ observations, who analyzed the relationship between the clinical parameters of periodontitis and the concentration of inflammatory markers in GCF. Perozini et al observed a correlation between concentration of IL-1β and volume of GCF in patients without diabetes [54]. Zhang et al showed a correlation between volume of GCF and concentration of TNF-α in GCF in patients with periodontal disease but without diabetes [55]. Kido et al revealed a correlation between the level of YKL-40 in GCF and the gingival index, which describes inflammation and corresponds to the mSBI index in diabetic patients with periodontal disease [56]. Also, in the study of Hiroshima et al, the level of resistin in GFC was correlated with the value of the gingival index [57].
Since GCF is a blood filtrate [8] and blood HbA1c concentration correlates with the severity of systemic inflammation [58] and in patients undergoing pancreas transplantation [59,60], it may be speculated that the local improvement obtained is a consequence of improvement in the severity of the systemic inflammation after pancreas transplantation.
The observed beneficial profile of analyzed biochemical parameters in GCF, and reduced inflammatory status in our SPK group, were not directly related to the improvement of clinical parameters reflecting morphological destruction of the periodontium, as indicated by assessment of pocket depth, clinical attachment loss, and tooth mobility.
In studies by other authors, such differences have been observed in patients with diabetes with or without good metabolic control of the disease. Meenwat et al, comparing patients with T1D with poor metabolic control of the disease with those with moderate or good control, found differences in pocket depth and clinical attachment loss in favor of patients with normal glycemia [38]. Demmer et al observed that uncontrolled T1D and T2D was associated with the progression of clinical attachment loss [61]. Lim et al revealed that subjects with acceptable glycemic control (HbA1c <8%) had a lower rate of sites with pocket depths ≥5 mm compared to subjects with insufficient glycemic control [39]. The only analysis confirming our results is the study of Longo et al, who observed similar depths of periodontal pockets in patients with periodontal disease and in those with T2D and without T2D [51].
The differences between our observations in the field of periodontal morphology and the results of other studies can be explained by the irreversible nature of destructive changes in the periodontium, which result from long-term exposure of tissue to the chronic diabetes milieu, despite decreased inflammatory status achieved by normoglycemia in patients with T1D after SPK. This situation can be compared to calcification of the arterial walls in patients with long-term T1D, in whom, after pancreas transplantation, the intensity of changes reflected by measurements of intima-media thickness and pulse wave velocity was comparable to those treated with insulin or only slightly improved [62,63]. However, improvement in fields such as cardiovascular diseases is noticeable only after many years, and perhaps also in the field of periodontal diseases it will be apparent later [23,64].
The observed lack of beneficial effects in the aforementioned clinical parameters after pancreas transplantation and the differences between kidney or kidney and pancreas recipients and the group of kidney recipients without diabetes also have significant clinical implications. Bearing in mind our previous observations of the irreversibility of a number of oral pathologies resulting from long-term diabetes [31], it can be concluded that the group of patients with ESRD in the course of T1D after SPK or KTx requires special supervision in post-transplant care of the oral cavity, regardless of the effectiveness of pancreatic transplantation. In addition, because there is a two-way relationship between diabetes and periodontitis, in which uncontrolled or poorly controlled diabetes predisposes to periodontal disease, and this negatively affects glycemic control [5], in T1D patients who have only had a kidney transplant or have lost pancreatic transplant function after SPK, appropriate treatment of periodontal disease can help to control diabetes.
Finally, it should be considered that some factors unexplored by us, like the use of different brushing techniques [65], solutions [66], probiotics [67], or natural compounds [68], can have a significant influence on the oral environment and modify clinical parameters in periodontal patients. These factors could have also affected the present results and should be considered in future clinical trials.
The present study has several limitations, including its cross-sectional design, relatively small study groups (which precluded the possibility sample size calculation), and longer T1D duration before transplantation in the SPK group.
Actually, in our analysis, the value of any analyzed clinical parameters did not correlate with the overall duration of diabetes, but the lack of this correlation could be caused by the longer duration of diabetes before transplantation and, consequently, a greater severity of irreversible changes in the SPK group. An attempt to select compatible groups in terms of this parameter would have resulted in a further reduction of the number of analyzed patients, which would have affected the results of statistical analysis.
Additionally, we did not consider the concentrations of analyzed markers in plasma. Since elevated plasma resistin level was reported in patients with periodontitis by Furugen et al [69] and Saito et al [70], analysis of the correlation between plasma and GCF concentration in the analyzed inflammatory parameters elucidate the etiology of this abnormality.
Conclusions
Patients with end-stage renal disease caused by type 1 diabetes after simultaneous pancreas and kidney transplantation had lower levels of selected biochemical markers of inflammation in gingival crevicular fluid and had reduced intensity of periodontitis compared to kidney recipients treated with insulin.
The severity of morphological changes in the periodontium in patients with type 1 diabetes after kidney or pancreas and kidney transplantation is higher than in non-diabetic kidney recipients.
Figures
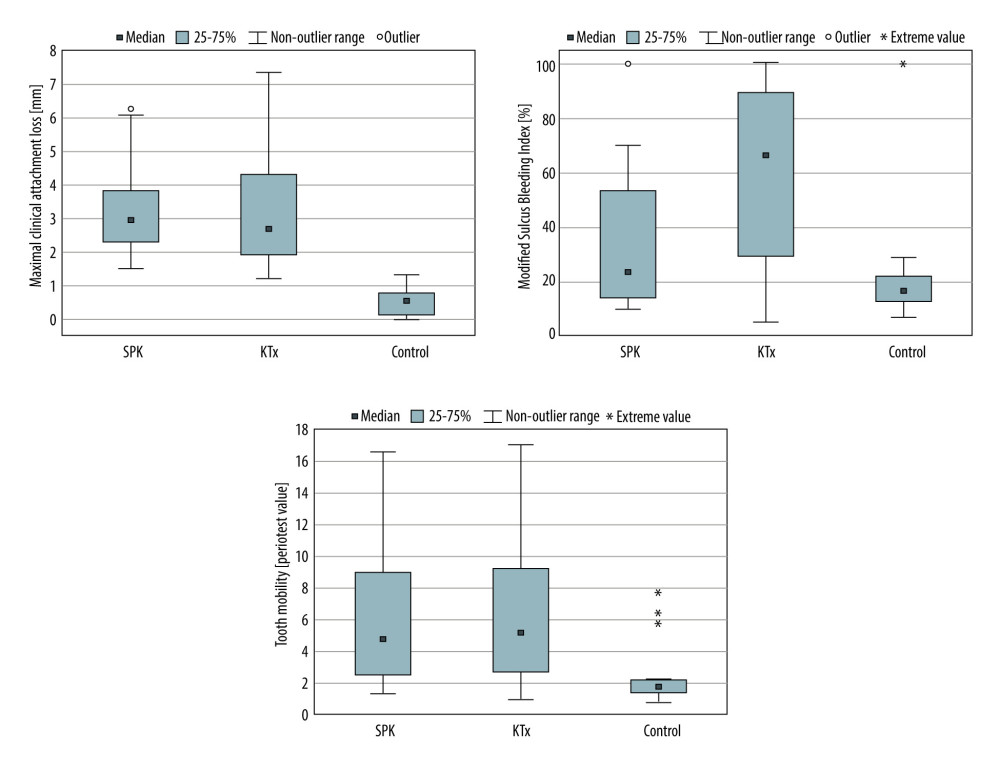 Figure 1. The box-plots of selected parameters of periodontal lesions in analyzed groups. Statistica 13.3 (TIBCO Software Inc., Palo Alto, CA, USA).
Figure 1. The box-plots of selected parameters of periodontal lesions in analyzed groups. Statistica 13.3 (TIBCO Software Inc., Palo Alto, CA, USA). 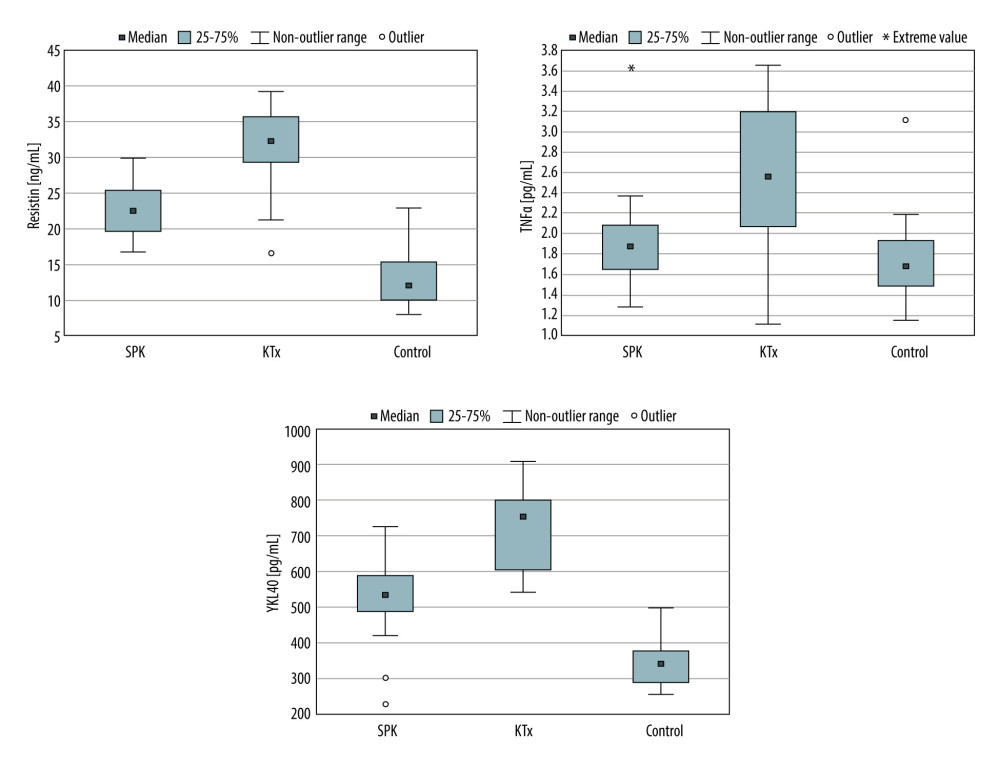 Figure 2. Box-plots of selected inflammation markers in analyzed groups. Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA).
Figure 2. Box-plots of selected inflammation markers in analyzed groups. Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA). 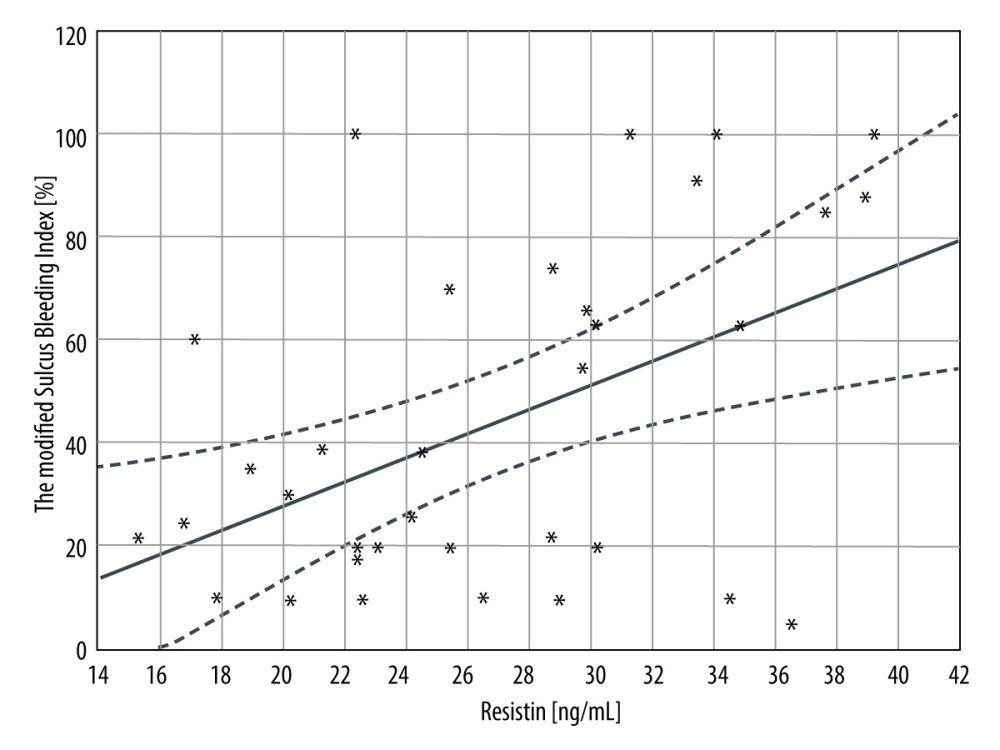 Figure 3. The linear regression between resistin concentration in gingival crevicular fluid and the modified Sulcus Bleeding Index in combined group of patients with end-stage renal disease in course of type 1 diabetes mellitus after simultaneous pancreas-kidney or kidney transplantation (r=0.394, p<0.05). Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA).
Figure 3. The linear regression between resistin concentration in gingival crevicular fluid and the modified Sulcus Bleeding Index in combined group of patients with end-stage renal disease in course of type 1 diabetes mellitus after simultaneous pancreas-kidney or kidney transplantation (r=0.394, p<0.05). Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA). Tables
Table 1. Clinical characteristics, biochemical parameters, and immunosuppressive treatment in type 1 diabetic patients after simultaneous pancreas and kidney transplantation (SPK) or kidney transplantation (KTx), and control group (numbers and rates or mean±SD or median and IQR).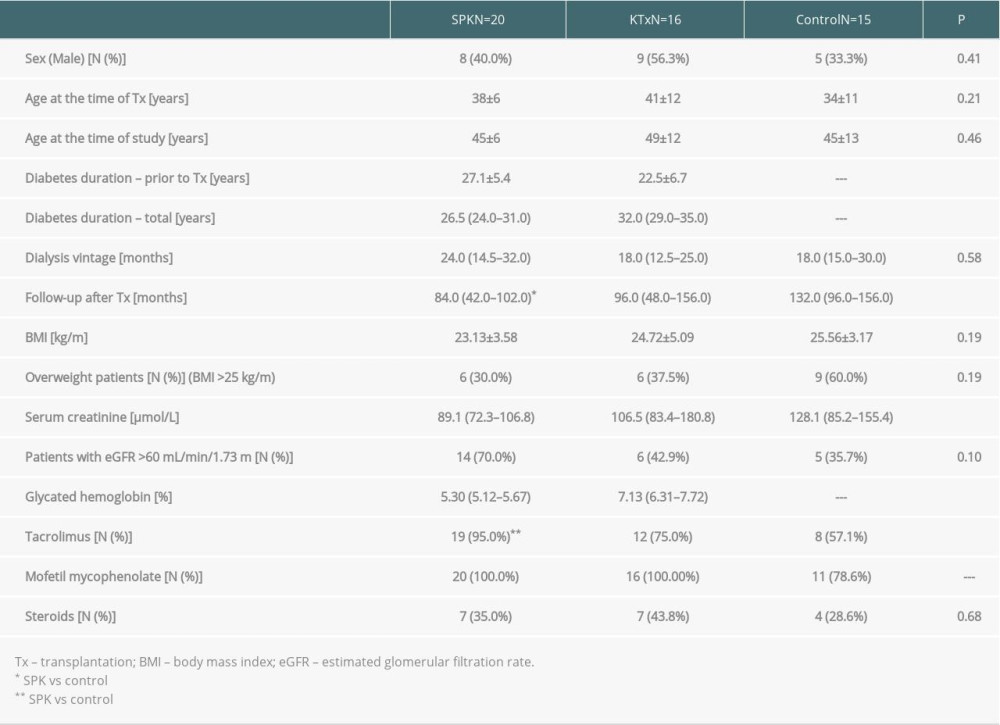 Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR).
Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR).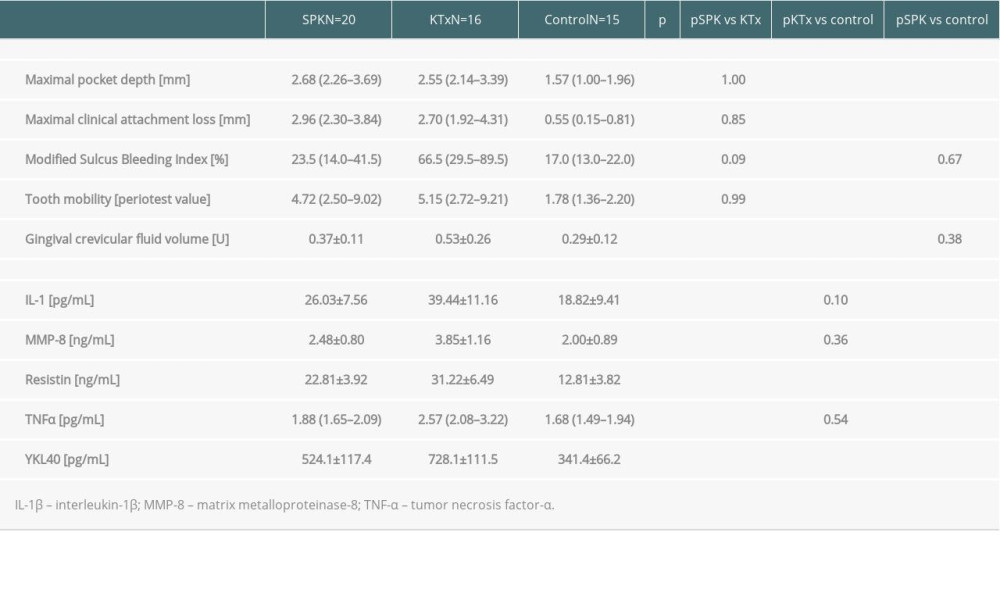 Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown).
Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown).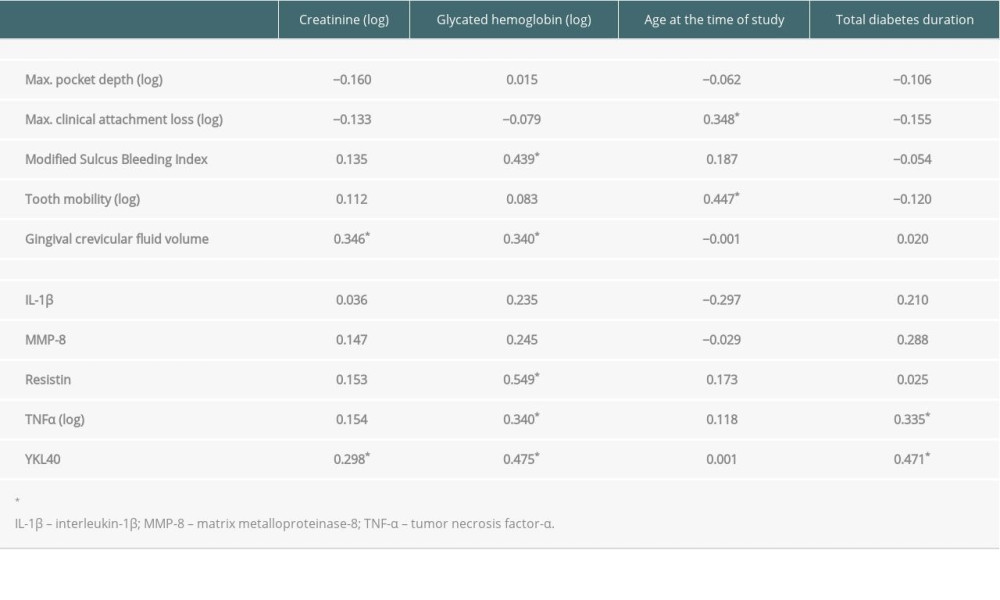
References
1. Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Research Group, Risk factors for cardiovascular disease in type 1 diabetes: Diabetes, 2016; 65; 1370-79
2. Finne P, Reunanen A, Stenman S, Incidence of end-stage renal disease in patients with type 1 diabetes: JAMA, 2005; 294; 1782-87
3. Hammes HP, Kerner W, Hofer SDPV-Wiss Study Group, Diabetic retinopathy in type 1 diabetes-a contemporary analysis of 8,784 patients: Diabetologia, 2011; 54; 1977-84
4. Carneiro VL, Fraiz FC, de Ferreira FM, The influence of glycemic control on the oral health of children and adolescents with diabetes mellitus type 1: Arch Endocrinol Metab, 2015; 59; 535-40
5. Popławska-Kita A, Siewko K, Szpak P, Association between type 1 diabetes and periodontal health: Adv Med Sci, 2014; 59; 126-31
6. Cekici A, Kantarci A, Hasturk H, Van Dyke TE, Inflammatory and immune pathways in the pathogenesis of periodontal disease: Periodontol 2000, 2014; 64; 57-80
7. Polepalle T, Moogala S, Boggarapu S, Acute phase proteins and their role in periodontitis: A review: J Clin Diagn Res, 2015; 9; 1-5
8. Barros SP, Williams R, Offenbacher S, Morelli T, Gingival crevicular fluid as a source of biomarkers for periodontitis: Periodontol 2000, 2016; 70; 53-64
9. Gamonal J, Acevedo A, Bascones A, Levels of interleukin-1 beta, -8, and -10 and RANTES in gingival crevicular fluid and cell populations in adult periodontitis patients and the effect of periodontal treatment: J Periodontol Oct, 2000; 71(10); 1535-45
10. Nogueira AVB, Nokhbehsaim M, Tekin S, Resistin is increased in periodontal cells and tissues: in vitro and in vivo studies: Mediators Inflamm, 2020; 2020; 9817095
11. Preferansow E, Sawczuk B, Gołębiewska M, Górska M, Pathologies of the oral cavity in patients with non-controlled diabetes type 1 and type 2 – analysis of periodontal status and periodontal treatment needs: Endokrynol Pol, 2015; 66; 428-33
12. Brennan DS, Spencer AJ, Roberts-Thomson KF, Tooth loss, chewing ability and quality of life: Qual Life Res, 2008; 17; 227-35
13. Silvestre FJ, Miralles L, Llambes F, Type 1 diabetes mellitus and periodontal disease: Relationship to different clinical variables: Med Oral Patol Oral Cir Bucal, 2009; 14; 175-79
14. Taylor JJ, Preshaw PM, Lalla E, A review of the evidence for pathogenic mech-anisms that may link periodontitis and diabetes: J Periodontol, 2013; 84(4 Suppl); S113-34
15. Salvi GE, Kandylaki M, Troendle A, Experimental gingivitis in type 1 diabetics: A controlled clinical and microbiological study: J Clin Periodontol, 2005; 32; 310-16
16. Nagata T, Relationship between diabetes and periodontal disease: Clinical Calcium, 2009; 19(9); 1291-98
17. Preshaw PM, Alba AL, Herrera D, Periodontitis and diabetes: A two-way relationship: Diabetologia, 2012; 55; 21-31
18. Hardy DC, Ross JH, Schuyler CA, Matrix metalloproteinase-8 expression in periodontal tissues surgically removed from diabetic and non-diabetic patients with periodontal disease: J Clin Periodontol, 2012; 39; 249-55
19. Nishimura R, Dorman JS, Bosnyak ZDiabetes Epidemiology Research International Mortality Study; Allegheny County Registry, Incidence of ESRD and survival after renal replacement therapy in patients with type 1 diabetes: A report from the Allegheny County Registry: Am J Kidney Dis, 2003; 42; 117-24
20. Kim YJ, Moura LM, Caldas CP, Evaluation of periodontal condition and risk in patients with chronic kidney disease on hemodialysis: Einstein (Sao Paulo), 2017; 15; 173-77
21. Hou Y, Wang X, Zhang CX, Risk factors of periodontal disease in maintenance hemodialysis patients: Medicine (Baltimore), 2017; 96; e7892
22. Schmalz G, Kauffels A, Kollmar O, Oral behavior, dental, periodontal and microbiological findings in patients undergoing hemodialysis and after kidney transplantation: BMC Oral Health, 2016; 16; 72
23. Costantinides F, Castronovo G, Vettori E, Dental care for patients with end-stage renal disease and undergoing hemodialysis: Int J Dent, 2018; 2018; 9610892
24. Young BY, Gill J, Huang E, Living donor kidney versus simultaneous pancreas-kidney transplant in type i diabetics: An analysis of the OPTN/UNOS database: Clin J Am Soc Nephrol, 2009; 4; 845-52
25. Lindahl JP, Hartmann A, Horneland R, Improved patient survival with simultaneous pancreas and kidney transplantation in recipients with diabetic end-stage renal disease: Diabetologia, 2013; 56; 1364-71
26. Sampaio MS, Poommipanit N, Cho YW, Transplantation with pancreas after living donor kidney vs. living donor kidney alone in type 1 diabetes mellitus recipients: Clin Transplant, 2010; 24; 812-20
27. Larsen JL, Colling CW, Ratanasuwan T, Pancreas transplantation improves vascular disease in patients with type 1 diabetes: Diabetes Care, 2004; 27; 1706
28. Argente-Pla M, Pérez-Lázaro A, Martinez-Millana A, Simultaneous pancreas kidney transplantation improves cardiovascular autonomic neuropathy with improved valsalva ratio as the most precocious test: J Diabetes Res, 2020; 2020; 7574628
29. Giannarelli C, Bianchini E, Bruno RM, Local carotid stiffness and intima-media thickness assessment by a novel ultrasound-based system in essential hypertension: Atherosclerosis, 2012; 223; 372-77
30. Meirelles RF, Salvalaggio P, Pacheco-Silva A, Pancreas transplantation: Review: Einstein (Sao Paulo), 2015; 13(2); 305-9
31. Musiał M, Wiench R, Kolonko A, Simultaneously transplanted pancreas improves salivation and reduces oral fungal infections incidence in type 1 diabetic renal transplant recipients: Transplant Proc, 2020; 52; 2067-73
32. Gupta N, Rath SK, Lohra P, Comparative evaluation of accuracy of periodontal probing depth and attachment levels using a Florida probe versus traditional probes: Med J Armed Forces India, 2015; 71(4); 352-58
33. Mühlemann HR, Son S, Gingival sulcus bleeding – a leading symptom in initial gingivitis: Helv Odontol Acta, 1971; 15; 107-13
34. Chakrapani S, Goutham M, Krishnamohan T, Periotest values: Its reproducibility, accuracy, and variability with hormonal influence: Contemp Clin Dent, 2015; 6(1); 12-15
35. Gilowski L, Kondzielnik P, Wiench R, Efficacy of short-term adjunctive subantimicrobial dose doxycycline in diabetic patients – randomized study: Oral Dis, 2012; 18(8); 763-70
36. Gilowski L, Wiench R, Płocica I, Krzemiński TF, Amount of interleukin-1β and interleukin-1 receptor antagonist in periodontitis and healthy patients: Arch Oral Biol, 2014; 59(7); 729-34
37. Rahnama M, Czupkałło Ł, Kozicka-Czupkałło M, Łobacz M, Gingival crevicular fluid – composition and clinical importance in gingivitis and periodontitis: Polish Journal of Public Health, 2014; 124; 96-98
38. Meenawat A, Punn K, Srivastava V, Periodontal disease and type I diabetes mellitus: Associations with glycemic control and complications: J Indian Soc Periodontol, 2013; 17; 597-600
39. Lim LP, Tay FBK, Sum CF, Thai AC, Relationship between markers of metabolic control and inflammation on severity of periodontal disease in patients with diabetes mellitus: J Clin Periodontal, 2007; 34; 118-23
40. Marinho MC, Pacheco ABF, Costa GCV, Quantitative gingival crevicular fluid proteome in type 2 diabetes mellitus and chronic periodontitis: Oral Dis, 2019; 25; 588-95
41. Engebretson SP, Vossughi F, Hey-Hadavi J, The influence of diabetes on gingival crevicular fluid beta-glucuronidase and interleukin-8: J Clin Periodontol, 2006; 33; 784-90
42. Khurshid Z, Mali M, Naseem M, Human gingival crevicular fluids (GCF) proteomics: An overview: Dent J (Basel), 2017; 22; 12
43. Subbarao KC, Nattuthurai GS, Sundararajan SK, Gingival crevicular fluid: An overview: J Pharm Bioallied Sci, 2019; 11(Suppl 2); S135-39
44. Yücel OO, Berker E, Gariboğlu S, Otlu H, Interleukin-11, interleukin-1beta, interleukin-12 and the pathogenesis of inflammatory periodontal diseases: J Clin Periodontol, 2008; 35; 365-70
45. Goutoudi P, Diza E, Arvanitidou M, Effect of periodontal therapy on crevicular fluid interleukin-1beta and interleukin-10 levels in chronic periodontitis: J Dent, 2004; 32(7); 511-20
46. Mc Crudden MTC, Irwin CR, El Karim I, Matrix metalloproteinase-8 activity in gingival crevicular fluid: development of a novel assay: J Periodontal Res, 2017; 52; 556-61
47. Mittal M, Hassan B, Desai K, GCF resistin as a novel marker in patients with chronic periodontitis and rheumatoid arthritis: J Clin Diagn Res, 2015; 9; 62-64
48. Heralgi R, Suchetha A, Apoorva S, Estimation of tumour necrosis factor-alpha levels in gingival crevicular fluid in periodontal health and disease in an Indian population: Int J Contem Dent, 2011; 2; 23-29
49. Kranti K, Pramod N, Ashwini S, Naik S, Levels of biomarker Ykl-40 and interleukin-6 in gingival crevicular fluid in patients with chronic periodontitis and type 2 diabetes: Int J Recent Sci Res, 2017; 8; 17115-19
50. Engebretson SP, Hey-Hadavi J, Ehrhardt FJ, Gingival crevicular fluid levels of interleukin-1beta and glycemic control in patients with chronic periodontitis and type 2 diabetes: J Periodontol, 2004; 75; 1203-8
51. Longo PL, Artese HP, Horliana AC, Inflammatory markers in gingival crevicular fluid of periodontitis patients with type 2 diabetes mellitus according to glycemic control: A pilot study: Dent Res J (Isfahan), 2015; 12; 449-55
52. Gokhale NH, Acharya AB, Patil VS, Resistin levels in gingival crevicular fluid of patients with chronic periodontitis and type 2 diabetes mellitus: J Periodontol, 2014; 85; 610-17
53. Bulut U, Develioglu H, Taner IL, Berker E, Interleukin-1 beta levels in gingival crevicular fluid in type 2 diabetes mellitus and adult periodontitis: J Oral Sci, 2001; 43; 171-77
54. Perozini C, Chibebe PC, Leao MV, Gingival crevicular fluid biochemical markers in periodontal disease: A cross-sectional study: Quintessence Int, 2010; 41(10); 877-83
55. Zhang Q, Chen B, Zhu D, Yan F, Biomarker levels in gingival crevicular fluid of subjects with different periodontal conditions: A cross-sectional study: Arch Oral Biol, 2016; 72; 92-98
56. Kido J, Bando Y, Bando M, YKL-40 level in gingival crevicular fluid from patients with periodontitis and type 2 diabetes: Oral Dis, 2015; 21; 667-73
57. Hiroshima Y, Bando M, Inagaki Y, Resistin in gingival crevicular fluid and induction of resistin release by Porphyromonas gingivalis lipopolysaccharide in human neutrophils: J Periodontal Res, 2012; 47; 554-62
58. Liu S, Hempe JM, McCarter RJ, Association between Inflammation and Biological Variation in Hemoglobin A1c in U.S. Nondiabetic Adults: J Clin Endocrinol Metab, 2015; 100; 2364-71
59. Becker BN, Brazy PC, Becker YT, Simultaneous pancreas-kidney transplantation reduces excess mortality in type 1 diabetic patients with end-stage renal disease: Kidney Int, 2000; 57; 2129-35
60. Lehmann R, Graziano J, Brockmann J, Glycemic control in simultaneous islet-kidney versus pancreas-kidney transplantation in type 1 diabetes: A prospective 13-year follow-up: Diabetes Care, 2015; 38; 752-59
61. Demmer RT, Holtfreter B, Desvarieux M, The influence of type 1 and type 2 diabetes on periodontal disease progression: Prospective results from the Study of Health in Pomerania (SHIP): Diabetes Care, 2012; 35; 2036-42
62. Fiorina P, La Rocca E, Venturini M, Effects of kidney-pancreas transplantation on atherosclerotic risk factors and endothelial function in patients with uremia and type 1 diabetes: Diabetes, 2001; 50; 496-501
63. Stadler M, Theuer E, Anderwald C, Persistent arterial stiffness and endothelial dysfunction following successful pancreas-kidney transplantation in Type 1 diabetes: Diabet Med, 2009; 26; 1010-18
64. Biesenbach G, Königsrainer A, Gross C, Margreiter R, Progression of macrovascular diseases is reduced in type 1 diabetic patients after more than 5 years successful combined pancreas-kidney transplantation in comparison to kidney transplantation alone: Transpl Int, 2005; 18; 1054-60
65. Preda C, Butera A, Pelle S, The efficacy of powered oscillating heads vs. powered sonic action heads toothbrushes to maintain periodontal and peri-implant health: A narrative review: Int J Environ Res Public Health, 2021; 18(4); 1468
66. Láng O, Nagy KS, Láng J, Comparative study of hyperpure chlorine dioxide with two other irrigants regarding the viability of periodontal ligament stem cells: Clin Oral Invest, 2021; 25; 2981-92
67. Butera A, Gallo S, Maiorani C, Probiotic alternative to chlorhexidine in periodontal therapy: evaluation of clinical and microbiological parameters: Microorganisms, 2020; 9(1); 69
68. Costa-Pinto AR, Lemos AL, Tavaria FK, Pintado M, Chitosan and hydroxyapatite based biomaterials to circumvent periprosthetic joint infections: Materials (Basel), 2021; 14(4); 804
69. Furugen R, Hayashida H, Yamaguchi N, The relationship between periodontal condition and serum levels of resistin and adiponectin in elderly Japanese: J Periodontal Res, 2008; 43; 556-62
70. Saito T, Yamaguchi N, Shimazaki Y, Serum levels of resistin and adiponectin in women with periodontitis: the Hisayama study: J Dent Res, 2008; 87; 319-22
Figures
 Figure 1. The box-plots of selected parameters of periodontal lesions in analyzed groups. Statistica 13.3 (TIBCO Software Inc., Palo Alto, CA, USA).
Figure 1. The box-plots of selected parameters of periodontal lesions in analyzed groups. Statistica 13.3 (TIBCO Software Inc., Palo Alto, CA, USA). Figure 2. Box-plots of selected inflammation markers in analyzed groups. Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA).
Figure 2. Box-plots of selected inflammation markers in analyzed groups. Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA). Figure 3. The linear regression between resistin concentration in gingival crevicular fluid and the modified Sulcus Bleeding Index in combined group of patients with end-stage renal disease in course of type 1 diabetes mellitus after simultaneous pancreas-kidney or kidney transplantation (r=0.394, p<0.05). Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA).
Figure 3. The linear regression between resistin concentration in gingival crevicular fluid and the modified Sulcus Bleeding Index in combined group of patients with end-stage renal disease in course of type 1 diabetes mellitus after simultaneous pancreas-kidney or kidney transplantation (r=0.394, p<0.05). Statistica 13.3 (TIBCO Software, Inc., Palo Alto, CA, USA). Tables
 Table 1. Clinical characteristics, biochemical parameters, and immunosuppressive treatment in type 1 diabetic patients after simultaneous pancreas and kidney transplantation (SPK) or kidney transplantation (KTx), and control group (numbers and rates or mean±SD or median and IQR).
Table 1. Clinical characteristics, biochemical parameters, and immunosuppressive treatment in type 1 diabetic patients after simultaneous pancreas and kidney transplantation (SPK) or kidney transplantation (KTx), and control group (numbers and rates or mean±SD or median and IQR). Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR).
Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR). Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown).
Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown). Table 1. Clinical characteristics, biochemical parameters, and immunosuppressive treatment in type 1 diabetic patients after simultaneous pancreas and kidney transplantation (SPK) or kidney transplantation (KTx), and control group (numbers and rates or mean±SD or median and IQR).
Table 1. Clinical characteristics, biochemical parameters, and immunosuppressive treatment in type 1 diabetic patients after simultaneous pancreas and kidney transplantation (SPK) or kidney transplantation (KTx), and control group (numbers and rates or mean±SD or median and IQR). Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR).
Table 2. Comparison of selected parameters concerning periodontal lesions and concentration of analyzed inflammatory markers in gingival crevicular fluid between the groups of type 1 diabetic patients after simultaneous pancreas-kidney transplantation (SPK) or kidney transplantation (KTx) and control group (numbers and rates or mean±SD or median and IQR). Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown).
Table 3. Univariate regression analysis between clinical and biochemical parameters, periodontal parameters, and concentration of inflammatory markers in gingival crevicular fluid in combined group of patients with ESRD in course of T1D after SPK or KTx (Pearson r or Spearman r correlation coefficients are shown). In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








