29 October 2021: Original Paper
Impact of the Share 35 Policy on Perioperative Management and Mortality in Liver Transplantation Recipients
Warangkana Lapisatepun12ABCDEF, Vatche G. Agopian3BDE, Victor W. Xia1ADEF, Worakitti Lapisatepun34ABCDEF*DOI: 10.12659/AOT.932895
Ann Transplant 2021; 26:e932895
Abstract
BACKGROUND: The Share 35 policy was introduced in 2013 by the Organ Procurement and Transplantation Network (OPTN) to increase opportunities of sicker patients to access liver transplantation. However, it has the disadvantage of higher MELD score associated with adverse postoperative transplant outcomes. Early data after implementation of the Share 35 policy showed significantly poorer post-transplantation survival in some UNOS regions. We aimed to analyze the impact of Share 35 on demographics of patients, perioperative management, and perioperative mortality.
MATERIAL AND METHODS: A retrospective analysis of data was performed from an institutional liver transplantation cohort from 1 January 2008 to 31 December 2017. Adult patients who underwent liver transplantation before 2013 were defined as the pre-Share 35 group and the other group was defined as the post-Share 35 group. The MELD score of each patient was calculated at the time of transplantation. Perioperative mortality was defined as death within 30 days after the operation.
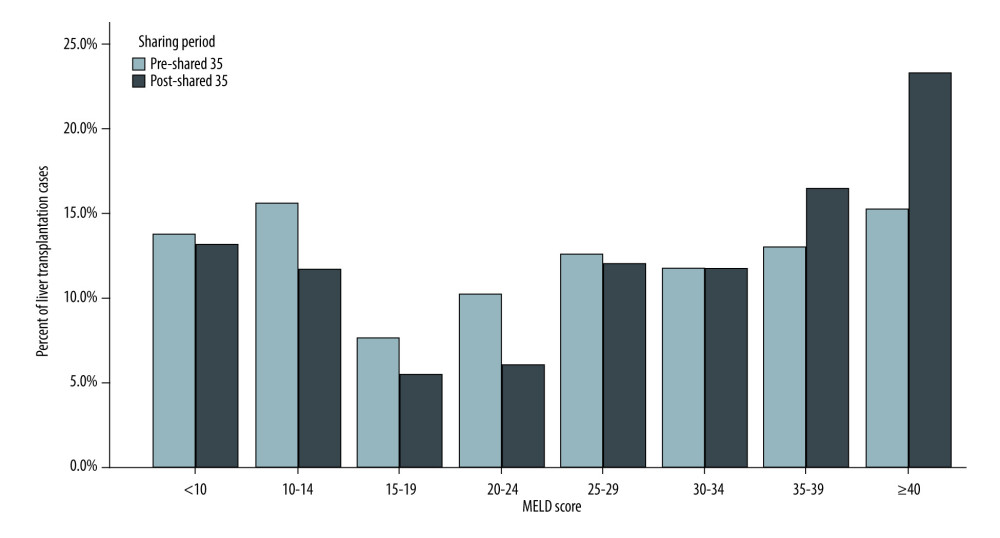
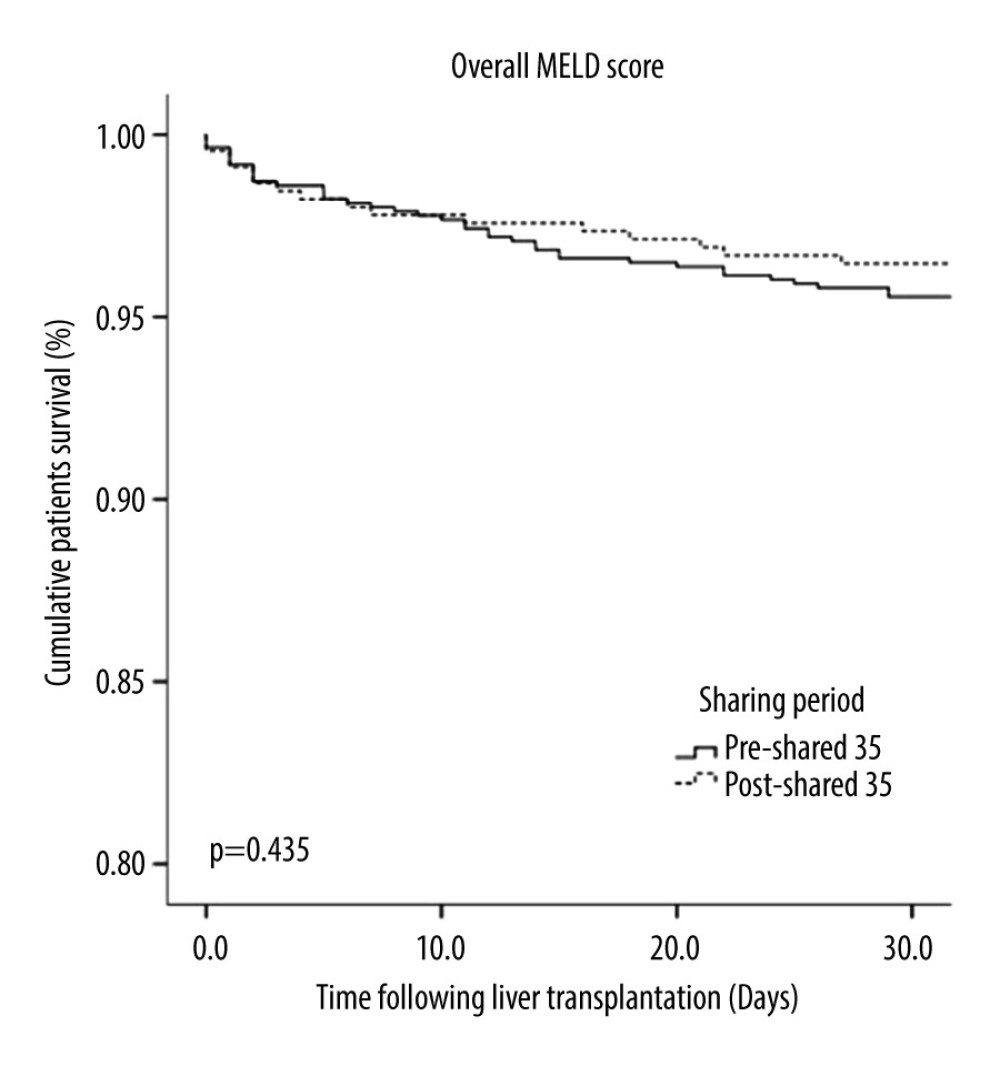
RESULTS: A total of 1596 patients underwent liver transplantation. Of those, 895 recipients underwent OLT in the pre-Share 35 era and 737 in the post-Share 35 era. The median MELD score was significantly higher in the post-Share 35 group (30 vs 26, P<0.001) and 45.7% of the post-Share 35 group had MELD scores ≥35. In intraoperative management, patients required significantly more blood component transfusion, intraoperative vasopressor, and fluid replacement. Veno-venous bypass (VVB) usage was significantly higher in the post-Share 35 era (47.2% vs 38.1%, P<0.001). In the subgroup of patients with MELD scores ≥35, the median waiting time was significantly shorter (18.5 vs 14.5 days, P=0.045). Overall perioperative mortality was not significantly difference between groups (P=0.435).
CONCLUSIONS: After implementation of the Share 35 policy, we performed liver transplantation in significantly higher medical acuity patients, which required more medical resources to obtain a result comparable to that of the pre-Share 35 era.
Keywords: Liver Transplantation, Perioperative Care, Tissue and Organ Procurement, Cohort Studies, Humans, policy, Severity of Illness Index, Waiting Lists
Background
The Share 35 policy was introduced in 2013 by the United Network for Organ Sharing and Organ Procurement and Transplantation Network (UNOS/OPTN) to increase chances to access liver allografts and to decrease wait-list mortality. The Share 35 policy promoted broader regional sharing for patients with MELD score more than 35 (in the 11 UNOS allocation regions in the USA) by prioritizing these patients over local sharing to patients with MELD score less than 35. After implementation of the Share 35 policy, several studies showed that recipient MELD score, need for organ support, and ICU admission prior to transplantation increased with decreased waiting time and wait-list mortality [1,2]. However, it has the disadvantage that higher MELD score recipients are more likely to have with adverse postoperative transplant outcomes, especially worsening post-transplant survival, which had been shown in some organ-sharing regions [2–4]. However, later studies after implementation of the Shared 35 policy showed no significance difference in overall mortality in MELD ≥35 recipients at the national level [5,6]. A recent study showed improved 1-year overall survival with donor-recipient matching and high MELD score recipient (MELD ≥35) matched with low-risk liver allograft (lower donor risk index) [7]. Most previous studies of the Share 35 policy mainly focused on preoperative baseline characteristics, waiting time, cost of treatment, and donor quality, but there has been no published study on changes in perioperative management in higher medical acuity liver transplantation recipients. The present study focused on the effect of the Share 35 policy on perioperative management and 30-day mortality.
Material and Methods
STUDY POPULATION:
After IRB approval, we retrospectively reviewed the prospectively collected data of 1596 adult patients who underwent liver transplantation at a single center (the University of California, Los Angeles, USA) from 1 January 2008 to 31 December 2017. We included all adult patients (≥18 years old) who underwent liver transplantation in our institute. Patients who underwent liver transplantation before 2013 were assigned to the pre-Share 35 group and patients who underwent liver transplantation later were assigned to the post-Share 35 group.
DATA COLLECTION AND OUTCOMES MEASUREMENTS:
Patients’ demographic data including age, comorbidities, etiology of liver disease, and baseline laboratory values were prospectively collected. MELD scores in this study were the MELD score calculated at the time of liver transplantation without any exception point. Intraoperative variables, including duration of surgery, transfusion of red blood cell and frozen fresh plasma, requirement for vasopressors, post-reperfusion syndrome, and veno-venous bypass usage, were collected by chart review. Postoperative variables included duration of ventilator use, postoperative complications, and 30-day patient survival. The primary outcome of this study was perioperative mortality, which was defined as death on the day of surgery or death within 30 days after surgery [8]. The secondary outcomes were perioperative management, blood transfusion, preoperative organ support, intraoperative continuous renal replacement therapy, cold ischemic time, veno-venous bypass usage, use of vasopressors, and postoperative complications.
STATISTICAL ANALYSIS:
Baseline characteristics of patients undergoing liver transplantation were compared. Categorical variables are reported as number and percentage and were compared by chi-square test. For continuous variables, the differences between 2 groups are reported as means (standard deviation) and compared by
Results
Demographic data and comorbidities are displayed in Table 1. A total of 1596 patients underwent liver transplantation during 2008–2017; 895 (56.07%) of these recipients underwent OLT in the pre-Share 35 era and 737 (46.18%) underwent OLT in the post-Share 35 era. The median age and BMI of the 2 groups were not significantly different. Interestingly, although most liver transplant recipients were male, the proportion of female recipients increased significantly in the post-Share 35 era (36.1% vs 42.6%,
Intraoperative parameters are shown in Table 2. Recipients required more red blood cell transfusions in the post-Share 35 group (15 units vs 19 units,
Postoperative data and complication are reported in Table 3. Postoperative ventilator stay was not significantly different between groups (3 days vs 2 days,
Discussion
This is one of the largest single-center studies to compare outcomes after liver transplantation between the pre-Share 35 and post-Share 35 eras. Our study demonstrated that recipients in the post-Share 35 era had significantly higher MELD scores at transplantation and shorter waiting times. Moreover, recipients in the post-Share 35 era required significantly more blood transfusion, vasopressor requirement, and intraoperative organ support. However, perioperative mortality was not significantly different between the 2 groups.
After implementation of the Share 35 policy in 2013, several studies showed a decrease in wait time and wait-list mortality among MELD ≥35 recipients [1,2]. A nation-wide study using UNOS data showed a significant increase in number and proportion of liver transplantation recipients with MELD score ≥35 after the Share 35 policy was implemented [1]. Moreover, the Share 35 policy showed decreased 1-year mortality and improved donor-recipient matching [7]. Interestingly, the Share 35 policy also improved graft and patient survival in recipients with either MELD score ≥40 or re-transplantation [9,10]. Berumen et al showed that the cost of treatment for liver transplantation increased after Share 35 policy implementation [11]. Even though many studies about the Share 35 policy have been published, there is scanty research on intraoperative management for higher medical acuity liver transplantation recipients after implementation of the policy.
Baseline characteristics of our liver transplantation recipients were similar to those in national data. After implementation of the Share 35 policy, we have encountered more patients with NASH/alcoholic cirrhosis, higher MELD score, a higher proportion of MELD ≥35 recipients, and more patients from the ICU. Furthermore, there were more patients requiring preoperative CRRT and vasopressor infusion [12]. In the post-Share 35 era, we performed more veno-venous bypass (VVB) in patients with MELD score ≥35 (56.4% vs 64.1%, P = 0.026). Our recent publication demonstrated VVB can lower the incidence of postoperative AKI in patients with compromised preoperative renal function [13]. Although VVB was used, there are significantly more patients who require vasopressor infusion and intermittent bolus to maintain adequate blood pressure and organ perfusion during the operation, which may be due to the significantly increased incidence of post-reperfusion syndrome (PRS). PRS appears to be associated with exposure to extracorporeal circuit such as VVB and longer cold ischemia time [14–16]. Longer warm-ischemia time and surgical time might result from greater complexity of the operation and increased incidence of VVB usage, as described in several reports [17,18]. Intraoperative CRRT was more frequently used due to the significantly higher number of patients with preoperative renal impairment and dialysis. A recent study showed intraoperative CRRT improved postoperative recovery of native renal function [19].
Our center is located in UNOS region 5, which has one of the highest median MELD allocation scores and rate of regional sharing after Share 35 [1,11]. The longer distance organs must travel due to regional sharing may have prolonged the cold ischemia time in our center.
There were several studies demonstrating the adverse effect of red blood cell transfusion on overall survival [20,21]. Although liver transplantation recipients in the post-Share 35 era required more red blood cell transfusions, there was no significant difference in perioperative survival in our cohort, and there was also no significant difference in postoperative mechanical ventilator duration. However, the post-Share 35 group had significantly longer postoperative hospital stay, possibly due to more postoperative complications. Several studies, including the present study, showed higher pre-transplant acuity, MELD score, pre-transplant mechanical ventilator use, preoperative renal failure, and increase risks of perioperative complications, especially from bleeding, which might prolong the hospital stay [22,23].
There are several limitations of this study. First, its retrospective design may have led to selection bias. Second, this was a single-center study from 1 UNOS region, so the results may not be generalizable to other centers or UNOS regions. Finally, there was potential bias from the evolution of surgical treatment and perioperative care between the 2 eras.
Conclusions
We performed liver transplantation in patients with higher medical acuity in the post-Share 35 era. Perioperative management in higher medical acuity patients required more medical resources, which might be a significant factor that decreases perioperative mortality.
References
1. Massie AB, Chow EK, Wickliffe CE, Early changes in liver distribution following implementation of Share 35: Am J Transplant, 2015; 15(3); 659-67
2. Halazun KJ, Mathur AK, Rana AA, One Size does not fit all – regional variation in the impact of the Share 35 liver allocation policy: Am J Transplant, 2016; 16(1); 137-42
3. Lai Q, Molinaro A, Mennini G, Preoperative donor scores and postoperative early measures of graft function: Relevance to the outcome of liver transplantation: Transplant Proc, 2010; 42(4); 1209-11
4. Klein KB, Stafinski TD, Menon D, Predicting survival after liver transplantation based on pre-transplant MELD score: A systematic review of the literature: PLoS One, 2013; 8(12); e80661
5. Edwards EB, Harper AM, Hirose R, Mulligan DC, The impact of broader regional sharing of livers: 2-year results of “Share 35”: Liver Transpl, 2016; 22(4); 399-409
6. Nicolas CT, Nyberg SL, Heimbach JK, Liver transplantation after Share 35: Impact on pretransplant and posttransplant costs and mortality: Liver Transpl, 2017; 23(1); 11-18
7. Kwong AJ, Goel A, Mannalithara A, Kim WR, Improved posttransplant mortality after Share 35 for liver transplantation: Hepatology, 2018; 67(1); 273-81
8. Watters DA, Hollands MJ, Gruen RL, Perioperative mortality rate (POMR): A global indicator of access to safe surgery and anaesthesia: World J Surg, 2015; 39(4); 856-64
9. Nekrasov V, Matsuoka L, Rauf M, National outcomes of liver transplantation for model for end-stage liver disease score >/=40: The impact of Share 35: Am J Transplant, 2016; 16(10); 2912-24
10. Brooks JT, Koizumi N, Neglia E, Improved retransplant outcomes: Early evidence of the Share35 impact: HPB (Oxford), 2018; 20(7); 649-57
11. Berumen J, Misel M, Vodkin I, The effects of Share 35 on the cost of liver transplantation: Clin Transplant, 2017; 31(5); ctr.12937
12. Starzl TE, Iwatsuki S, Van Thiel DH, Evolution of liver transplantation: Hepatology, 1982; 2(5); 614-36
13. Sun K, Hong F, Wang Y, Venovenous bypass is associated with a lower incidence of acute kidney injury after liver transplantation in patients with compromised pretransplant renal function: Anesth Analg, 2017; 125(5); 1463-70
14. Paugam-Burtz C, Kavafyan J, Merckx P, Postreperfusion syndrome during liver transplantation for cirrhosis: Outcome and predictors: Liver Transpl, 2009; 15(5); 522-29
15. Segal H, Sheikh S, Kallis P, Complement activation during major surgery: The effect of extracorporeal circuits and high-dose aprotinin: J Cardiothorac Vasc Anesth, 1998; 12(5); 542-47
16. Solberg R, Scholz T, Videm V, Heparin coating reduces cell activation and mediator release in an in vitro venovenous bypass model for liver transplantation: Transpl Int, 1998; 11(4); 252-58
17. Kuo PC, Alfrey EJ, Garcia G, Orthotopic liver transplantation with selective use of venovenous bypass: Am J Surg, 1995; 170(6); 671-75
18. Johnson MW, Powelson JA, Auchincloss H, Selective use of veno-venous bypass in orthotopic liver transplantation: Clin Transplant, 1996; 10(2); 181-85
19. Zimmerman MA, Selim M, Kim J, Outcome analysis of continuous intraoperative renal replacement therapy in the highest acuity liver transplant recipients: A single-center experience: Surgery, 2017; 161(5); 1279-86
20. de Boer MT, Christensen MC, Asmussen M, The impact of intraoperative transfusion of platelets and red blood cells on survival after liver transplantation: Anesth Analg, 2008; 106(1); 32-44
21. Ramos E, Dalmau A, Sabate A, Intraoperative red blood cell transfusion in liver transplantation: influence on patient outcome, prediction of requirements, and measures to reduce them: Liver Transpl, 2003; 9(12); 1320-27
22. DiNorcia J, Lee MK, Harlander-Locke M, Reoperative complications after primary orthotopic liver transplantation: A contemporary single-center experience in the post-model for end-stage liver disease era: J Am Coll Surg, 2014; 219(5); 993-1000
23. Moghadamyeghaneh Z, Alameddine M, Jue JS, A nationwide analysis of re-exploration after liver transplant: HPB (Oxford), 2018; 20(3); 216-21
Figures
Tables
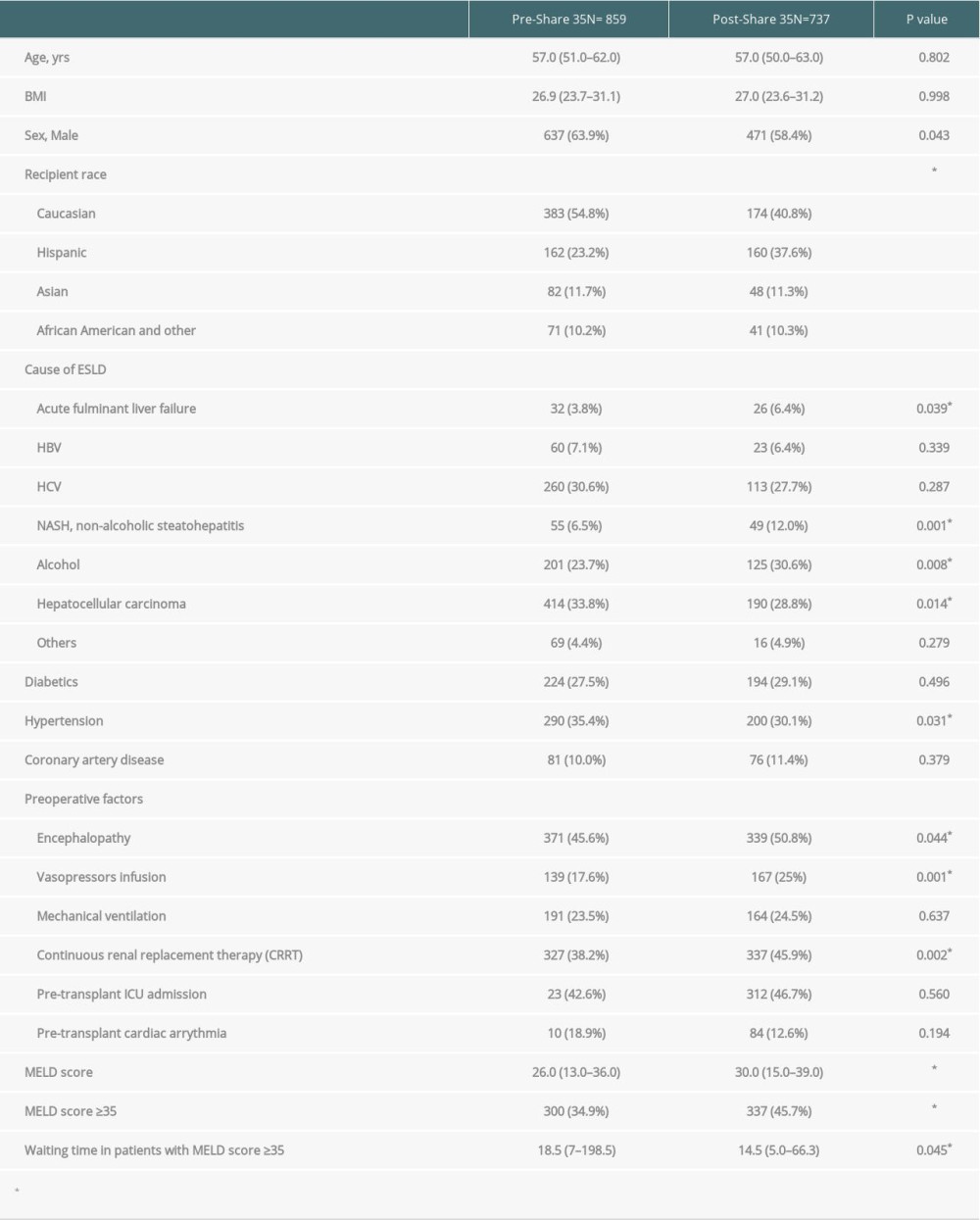 Table 1. Recipient demographics, comorbidities and medical acuity of pre- and post-Share 35 transplantation.
Table 1. Recipient demographics, comorbidities and medical acuity of pre- and post-Share 35 transplantation.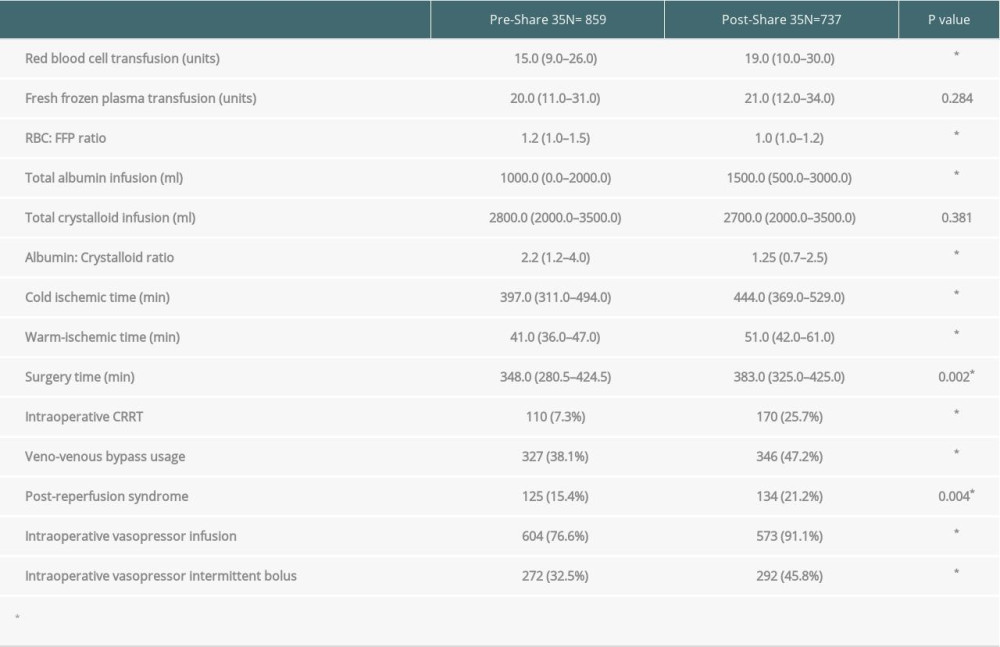 Table 2. Intraoperative data of pre- and post-Share 35 transplantation.
Table 2. Intraoperative data of pre- and post-Share 35 transplantation.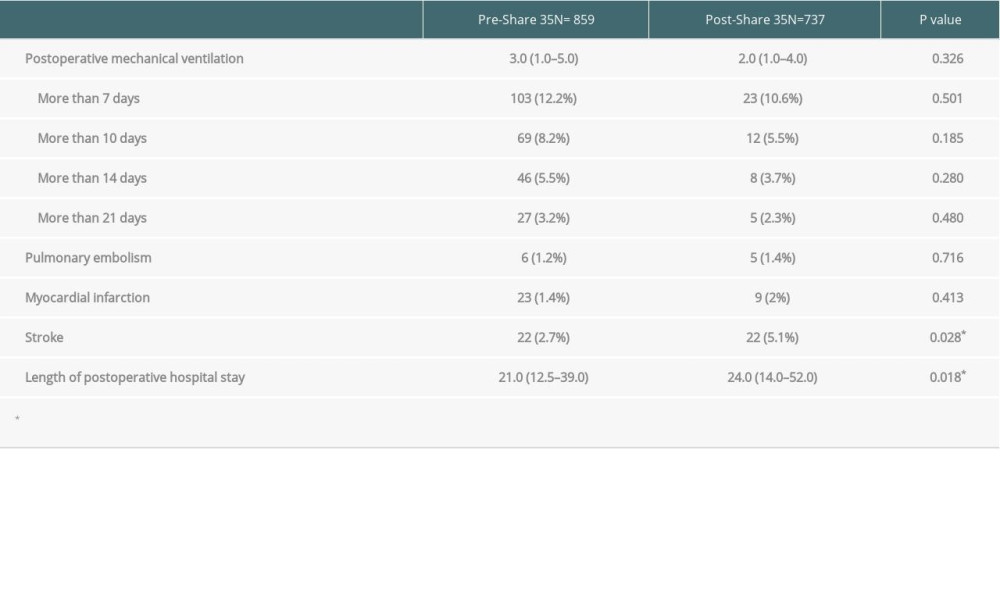 Table 3. Postoperative data of pre- and post-Share 35 transplantation.
Table 3. Postoperative data of pre- and post-Share 35 transplantation. Table 1. Recipient demographics, comorbidities and medical acuity of pre- and post-Share 35 transplantation.
Table 1. Recipient demographics, comorbidities and medical acuity of pre- and post-Share 35 transplantation. Table 2. Intraoperative data of pre- and post-Share 35 transplantation.
Table 2. Intraoperative data of pre- and post-Share 35 transplantation. Table 3. Postoperative data of pre- and post-Share 35 transplantation.
Table 3. Postoperative data of pre- and post-Share 35 transplantation. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860