02 August 2022: Original Paper
The Association Between Tacrolimus Levels and Hyperkalemia in Allograft Recipients Who Underwent Heart or Lung Transplantation in New Orleans, USA Between 2013 and 2019: A Single-Center Retrospective Study
Olivia L. GriffisDOI: 10.12659/AOT.935607
Ann Transplant 2022; 27:e935607
Abstract
BACKGROUND: Tacrolimus, a calcineurin inhibitor, is the cornerstone of immunosuppressive strategies in transplantation. Other studies have concluded that hyperkalemia correlates with tacrolimus therapy, though the impact is poorly understood. In 2 separate analyses, the aim of this study was to investigate if the presence and/or magnitude of exposure to tacrolimus increased potassium levels in heart or lung transplant recipients.
MATERIAL AND METHODS: This retrospective study identified allograft recipients who underwent heart or lung transplantation from January 2013 to December 2019 at Ochsner Health in New Orleans, USA. The first analysis (A-1) comprised 103 lung transplant patients’ assessed potassium levels and prevalence of hyperkalemia in the absence of tacrolimus before transplant versus 30 days after transplant on tacrolimus. The second analysis (A-2) included 187 heart or lung transplant recipients and compared potassium levels and prevalence of hyperkalemia at day 30 after transplant during higher tacrolimus exposure (High-TAC) vs day 300 during lower tacrolimus exposure (Low-TAC).
RESULTS: In A-1, patients on tacrolimus had higher median potassium (4.7 mmol/L vs 4.1 mmol/L, P<0.0001) and prevalence of hyperkalemia (32.04% vs 5.83%, P<0.0001). In A-2, patients in the High-TAC group had higher median potassium (4.6 mmol/L vs 4.4 mmol/L, P=0.0005) and prevalence of hyperkalemia (22.46% vs 12.30%, P=0.0056).
CONCLUSIONS: Our findings support those from previous studies. Presence of and higher levels of tacrolimus were associated with hyperkalemia following heart and lung transplantation. These findings highlight the importance of long-term monitoring of potassium levels in patients treated with tacrolimus.
Keywords: Heart Transplantation, Hyperkalemia, Lung Transplantation, Potassium Compounds, Tacrolimus, Allografts, Graft Rejection, Humans, Immunosuppressive Agents, New Orleans, Potassium
Background
Initially, allograft dysfunction secondary to acute rejection was the major cause of mortality in heart and lung organ transplantation [1]. The arrival of calcineurin inhibitors (CNIs) to the immunosuppressive cocktail revolutionized cardiothoracic transplantation by limiting immune-related injury and reducing episodic acute rejection. As a result, allograft and patient survival improved significantly [1,2]. To improve acute cellular rejection (ACR) rates, most heart and lung transplantation centers implement tacrolimus as the CNI of choice for de novo, triple-drug immunosuppressive regimens in conjunction with an antiproliferative agent and corticosteroids [3,4].
Tacrolimus is lipophilic drug with an average bioavailability of 25% and is predominantly metabolized via CYP3A4/CYP3A5 enzymes in the gut and liver [5]. Genetic polymorphisms of CYP metabolism within patient populations impact blood concentrations alongside numerous drug-drug/drug-food interactions [5,6]. Fluctuations in tacrolimus levels are widely influenced by intra- and inter-patient variability that shape clinical outcomes and augment risk for adverse drug events [1,5,7]. Thus, CNIs are dosed frequently as a narrow therapeutic index (NTI) medication to limit toxicities while providing therapeutic success [5]. Area under the curve (AUC) monitoring has been considered for identifying clinical effects. Trough concentrations are currently used for monitoring and are poorly correlated to AUC [5]. To date, cost and labor associated with population-based Bayesian forecasting limit this practice [5]. Adequate tacrolimus exposure measured by increased time-in-therapeutic range (TTR) predicted ACR, chronic lung allograft dysfunction (CLAD), infectious risk, and mortality at 1-year post-lung transplantation [8]. Therapeutic drug monitoring (TDM) is currently recommended for tacrolimus therapy with blood concentrations drawn at either 12-h or 24-h, contingent to dosing frequency of the immediate release formulation [5,9]. This trough level is based upon inter-patient variability with prespecified goal range to maintain drug concentration, adjusted per center-specific protocol [5,7].
Despite the efficacy of TDM, all CNIs are limited by an extensive adverse effects profile that includes electrolyte abnormalities [1]. Elevated potassium levels frequently occur after transplantation, and prompt pharmacologic management is paramount to prevent cardiac arrhythmias associated with severe hyperkalemia [10]. Hyperkalemia presents a life-threatening complication comprising different mechanisms, including drug-induced hyperkalemia [11]. Yamada et al found that 1 in every 6 transplant patients experienced hyperkalemia in the acute care setting, which was associated with a 3-fold increase in hospital mortality [10]. In heart transplant recipients, Uriel et al found that the 1-year incidence of potassium greater than 5.5 mmol/L was 63%, with more than 50% of patients requiring potassium chelating therapy [12]. In clinical practice, hyperkalemia is often attributed to the use of calcineurin inhibitors after transplant [13]. Sahu et al published the first case report of tacrolimus-induced hyperkalemia in a heart transplant recipient [14]. The causal mechanisms of calcineurin inhibitor-induced hyperkalemia may involve activation of the sodium-chloride cotransporter within the renal distal convoluted tubule, leading to impaired potassium secretion and impairment of mineralocorticoid receptor expression [14–16].
Although it is the most frequently prescribed CNI, minimal literature exists beyond case reports that detail a relationship between tacrolimus and hyperkalemia in heart and lung transplantation [3,4,14]. It is unclear whether higher tacrolimus exposure increases hyperkalemia. Therefore, this retrospective study was conducted at a single cardiothoracic transplant center in New Orleans, USA to investigate the association between tacrolimus levels and hyperkalemia in patients who underwent heart or lung transplantation between 2013 and 2019.
Material and Methods
We conducted a single-center retrospective study after approval from the Ochsner Medical Center Institutional Review Board (IRB). We received a waiver of consent for our study from the IRB, which was approved given that our study was retrospective and all patient information was protected. The first analysis of the study (A-1) assessed whether the presence of tacrolimus affected serum potassium within a cohort of lung transplant recipients. We compared potassium levels and the prevalence of hyperkalemia 1 day prior to transplant while off tacrolimus vs 30 days after transplant while on tacrolimus. When collecting data, we reported lab values within 7 days before or after the 30-day post-transplant mark. Patients without labs at 30 days after transplant were excluded from our analysis. Lab values for potassium levels and tacrolimus levels were taken on the same day for each time period (for example, at the determined “day 30”, all lab values were taken on the same day). Hyperkalemia was defined as a serum potassium level greater than 5 mmol/L. Inclusion criteria were consecutive patients who received their first lung transplant between January 2013 and January 2019. Patients were excluded if they were less than 18 years old, received a dual organ transplant, died within 30 days after transplant, received a loop or thiazide diuretic within 3 days before transplant, did not receive tacrolimus-based immunosuppression at 30 days after transplant, or underwent renal replacement therapy at 30 days after transplant. In A-1, we did not include heart transplant patients to avoid confounding from the widespread use of loop diuretics and electrolyte replacements as a result of heart failure prior to transplantation.
The second analysis of the study (A-2) assessed whether tacrolimus trough levels affected serum potassium within a cohort of lung or heart transplant recipients. We compared potassium levels and the prevalence of hyperkalemia at day 30 after transplant during a period of higher tacrolimus trough goal levels (High-TAC) vs day 300 during lower tacrolimus trough goal levels (Low-TAC). Due to the relative infrequency of labs at 300 days compared to 30 days, we reported lab values within the 10th month after transplant for the 300-day value. Patients who did not have a value at 300 days or within the 10th month were excluded from the analysis. Lab values for potassium levels and tacrolimus levels were taken on the same day for each time period (for example, at the determined “day 300”, all lab values were taken on the same day). A-2 included patients who received their first heart or lung transplant between January 2013 and January 2019. Patients were excluded if they were less than 18 years old, received a dual organ transplant, died within 300 days after transplant, did not receive tacrolimus-based immunosuppression at 30 or 300 days after transplant, or did not receive sulfamethoxazole/trimethoprim (SMX/TMP) for infection prophylaxis at 30 or 300 days after transplant.
The following data were collected for both analyses: Demographics, serum potassium levels, blood glucose, estimated glomerular filtration rate (eGFR) using the 4-variable modified diet in renal disease calculation, and medications influencing potassium homeostasis.
Institutional guidelines in heart or lung transplant recipients during the study period required tacrolimus as the preferred calcineurin inhibitor. Analysis of tacrolimus exposure was conducted using 12-h trough levels with frequency of sampling per organ-specific protocol. Tacrolimus trough levels were obtained using whole-blood samples that were analyzed with liquid chromatography-tandem mass spectrometry. Tacrolimus trough goals for lung transplant patients were 10–12 ng/mL at day 30 and 8–10 ng/mL at day 300. Tacrolimus trough goals for heart transplant patients were 10–15 ng/mL at day 30 and 5–10 ng/mL at day 300. SMX/TMP was given as a single-strength tablet daily for 1 year in heart transplant recipients. By contrast, lung transplant recipients received a double-strength tablet 3 times a week for life. In addition, lung recipients were prescribed universal azole antifungal prophylaxis for a minimum of 3 months determined by protocol bronchoscopies and cultures. Heart and lung transplant recipients both received intravenous methylprednisolone 1000 mg in the operating room followed by a taper to 20–40 mg/day of prednisone by discharge. Subsequently, prednisone was weaned per physician discretion with a maintenance dose goal of ≤5 mg/day by 6–12 months.
Comparisons of categorical variables were analyzed using the McNemar test. Continuous variables were analyzed using paired
Results
The off tacrolimus vs on tacrolimus analysis (A-1) included 161 patients who underwent lung transplantation at our center from January 2013 through January 2019. After exclusions, a total of 103 patients were analyzed in A-1 (Figure 1). Baseline and clinical characteristics are listed in Table 1. Females comprised 40.78% of the study population and the median age was 58 years. Estimated glomerular filtration rate (eGFR) and the use of an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker (ACE/
The low tacrolimus goal group (Low-TAC) vs the high tacrolimus goal group (High-TAC) analysis (A-2) included 331 patients who underwent heart or lung transplantation from January 2013 through January 2019. After exclusions, a total of 187 patients were analyzed in A-2 (Figure 3). Baseline and clinical characteristics are listed in Table 3. Females comprised 38.5% of the study population and the median age was 55 years. Over half the patients (55.08%) were lung transplant recipients. As expected, tacrolimus trough levels were higher during the High-TAC vs Low-TAC period (11.3 ng/mL vs 8.9 ng/mL;
Discussion
Our results from the first analysis (A-1) confirm that potassium levels and the prevalence of hyperkalemia are significantly higher on tacrolimus vs off tacrolimus. The second analysis (A-2) illustrates that potassium levels and the prevalence of hyperkalemia are also significantly higher during periods of high tacrolimus goals (High-TAC) compared to low tacrolimus goals (Low-TAC). We acknowledge other possible reasons for elevated potassium levels on tacrolimus in A-1. Sulfamethoxazole/trimethoprim (SMX/TMP) was utilized almost exclusively after transplant on tacrolimus and is known to cause hyperkalemia through inhibition of epithelial sodium channels, leading to decreased renal tubular secretion of potassium [13].
Furthermore, renal function, measured by estimated glomerular filtration rate (eGFR), was worse on tacrolimus, which is also a risk factor for hyperkalemia. Nonetheless, A-2 strengthens the evidence of an association between tacrolimus and potassium despite these confounders. Specifically, we found higher potassium levels during the period of High-TAC vs Low-TAC despite higher eGFR during High-TAC and 100% SMX/TMP use during both periods of exposure.
Although recognized as a life-threatening complication, the literature supporting the link between tacrolimus and hyperkalemia is limited to mechanistic reviews and case reports in cardiothoracic transplantation [11,14]. A retrospective study in kidney transplant patients found increased hyperkalemia in patients taking tacrolimus vs cyclosporine [17]. However, there are few comparative studies exploring differences in potassium levels between immunosuppression that is tacrolimus-based vs non-calcineurin inhibitor-based. As such, we attempted to answer the research question by comparing pre- vs post-transplant potassium levels, and found increased post-transplant potassium was associated with the use of tacrolimus. We suggest that future prospective comparative studies investigating novel immunosuppression strategies in heart and lung transplantation report potassium levels between patients on tacrolimus vs non-calcineurin inhibitor-based immunosuppression.
Higher tacrolimus exposure is believed to increase potassium levels, but few studies have adequately explored this relationship. Similar to our results, a study in liver transplantation found fewer episodes of hyperkalemia with lower dosages of tacrolimus over time during the post-transplant period [18]. At month 1, the prevalence of hyperkalemia defined as a serum potassium value >5.5 mmol/L was 18.4% vs 6.1% at month 12 in patients with liver transplantation, whereas the prevalence of hyperkalemia defined as a serum potassium value >5.0 mmol/L in our study was 22.5% at day 30 during High-TAC vs 12.3% at day 300 during Low-TAC in the patients with heart or lung transplantation. Notably, higher tacrolimus doses were present at month 1 compared to month 12, which suggests that higher drug exposure contributed to hyperkalemia. However, due to a narrow therapeutic range and high inter-patient pharmacokinetic variability, tacrolimus trough levels obtained in our study are preferred over dose as a measure of drug exposure, because tacrolimus trough level measurements are better correlated to clinical outcomes than dose [9]. Therefore, it is unclear whether the degree of tacrolimus drug exposure was linked to hyperkalemia in the study evaluating hyperkalemia after liver transplant. Another comparison between that study and ours was the hyperkalemia cutoff value. The aforementioned study used a potassium level greater than 5.5 mmol/L as the breakpoint, though the mean and median values were 5.0 mmol/L or less [18]. Clinical intervention for hyperkalemia typically occurs before potassium levels reach 5.5 mmol/L in heart and lung transplantation due to risk of life-threatening arrhythmias. After cardiac surgery, the optimum range is often defined as 3.8–4.3 mmol/L [11]. Therefore, we chose 5.0 mmol/L as our defined breakpoint for hyperkalemia.
To our knowledge, only 1 study in cardiothoracic transplantation has compared potassium levels between patients on high vs low trough concentrations of calcineurin inhibitors [19]. The Nordic Certican Trial in Heart and Lung Transplantation was conducted late after transplantation (at 56–72 months) in contrast with our study, which took place in the first year. Two hundred and eighty-two heart or lung transplant recipients were randomized to continue high exposure calcineurin inhibitors vs everolimus plus reduced exposure calcineurin inhibitors (≥50% reduction in cyclosporine or tacrolimus trough levels). At 1-year follow up, the potassium reduction from baseline was slightly higher in the reduced exposure group (−0.2 mmol/L vs −0.1 mmol/L,
This investigation is relevant because it identifies the time early after transplant corresponding to high tacrolimus trough levels as a time when elevated potassium is especially prevalent. During this time, clinicians should closely monitor potassium levels and consider avoiding the use of antihypertensives or diuretics that can cause hyperkalemia. This strategy may reduce the need for potassium reversal agents and facilitate the uninterrupted use of effective infection prophylaxis with SMX/TMP. Uriel et al found that 39% of heart transplant patients with hyperkalemia had SMX/TMP discontinued, of which, 62% developed subsequent bacterial infection [12].
Our study has several limitations. Ideally, using an independent comparator group off calcineurin inhibitors would have strengthened the study design, but we were limited to comparisons within the same group of patients. Given the increased rejection risk of calcineurin inhibitor-free de novo immunosuppression regimens in heart and lung transplantation, an independent comparator group would not have been feasible. Second, cross-sectional data with tacrolimus trough levels obtained on post-transplant days 30 and 300 may be a crude marker for exposure and thus a poor correlate to clinical outcomes, such as hyperkalemia. Indeed, tacrolimus trough levels are poorly correlated to the area under the curve, which is best associated with clinical effects [5,6]. Furthermore, within-patient variability of tacrolimus trough levels may be a better marker for clinical outcomes rather than a single value obtained on days 30 and 300. Despite this, our study reflects real-world practice, since tacrolimus trough levels are simple to obtain and widely used to guide dose adjustments for optimization of safety and efficacy. Finally, we did not collect information on the quantities of potassium consumed in patients’ diets.
Conclusions
The findings from our cardiothoracic transplant center support those from previous studies that have shown that the presence of tacrolimus and higher tacrolimus trough levels are associated with hyperkalemia following both heart and lung transplantation. These findings highlight the importance of long-term monitoring of potassium levels and monitoring of concomitant medications that increase potassium levels in patients treated with tacrolimus during the early post-transplant period.
Figures
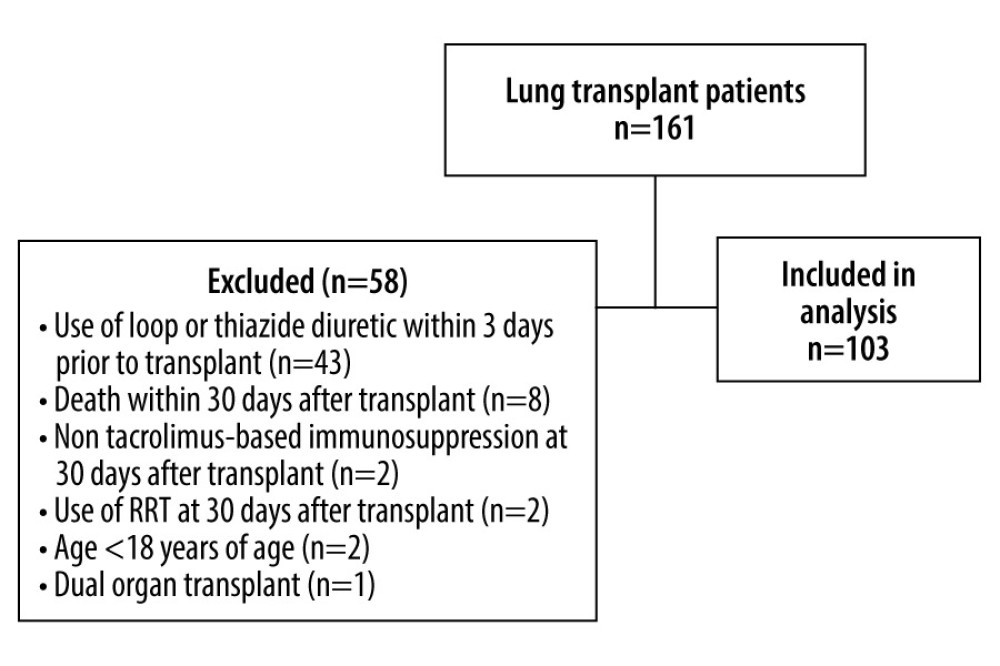 Figure 1. Study population of part IFigure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 1. Study population of part IFigure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. 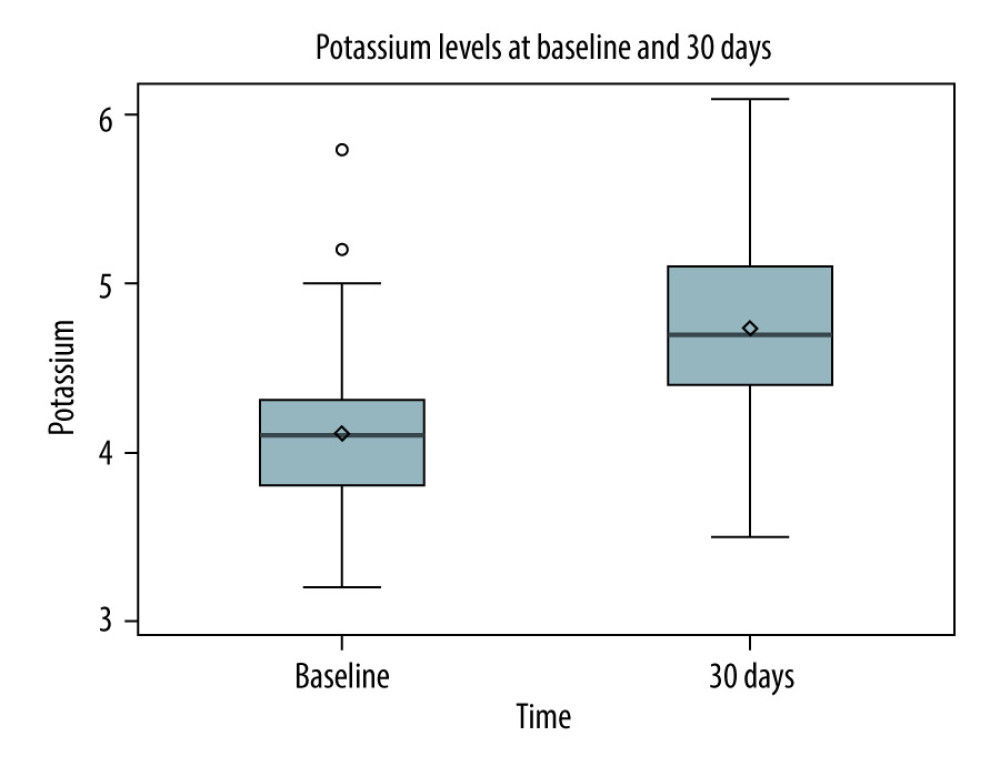 Figure 2. Potassium levels pre-transplant (no tacrolimus) versus 30 days post-transplant (tacrolimus)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 2. Potassium levels pre-transplant (no tacrolimus) versus 30 days post-transplant (tacrolimus)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. 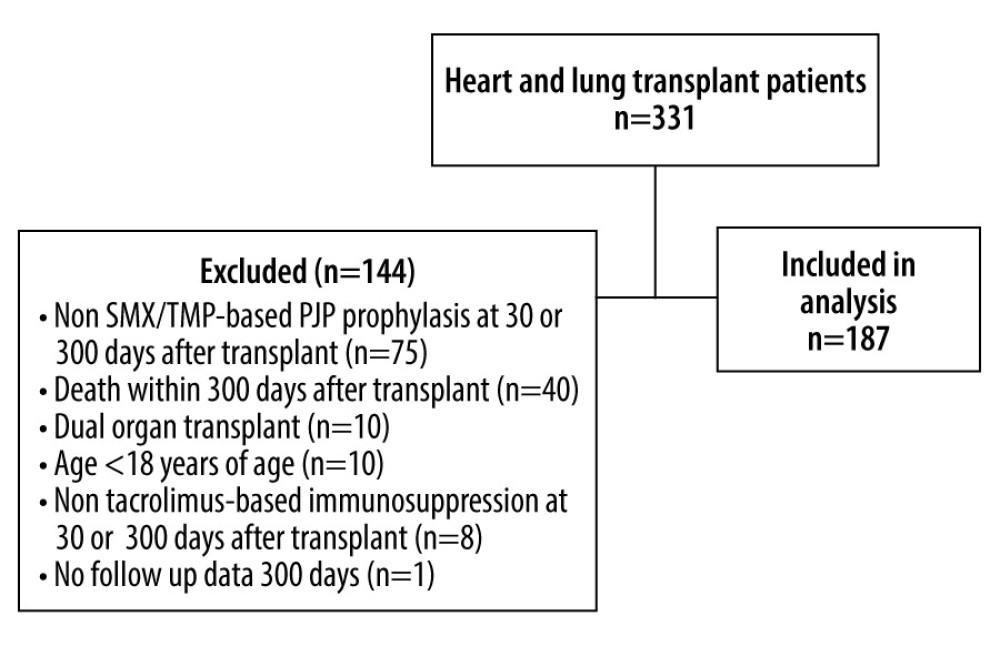 Figure 3. Study population of part IISMX/TMP – sulfamethoxazole/trimethoprim. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 3. Study population of part IISMX/TMP – sulfamethoxazole/trimethoprim. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. 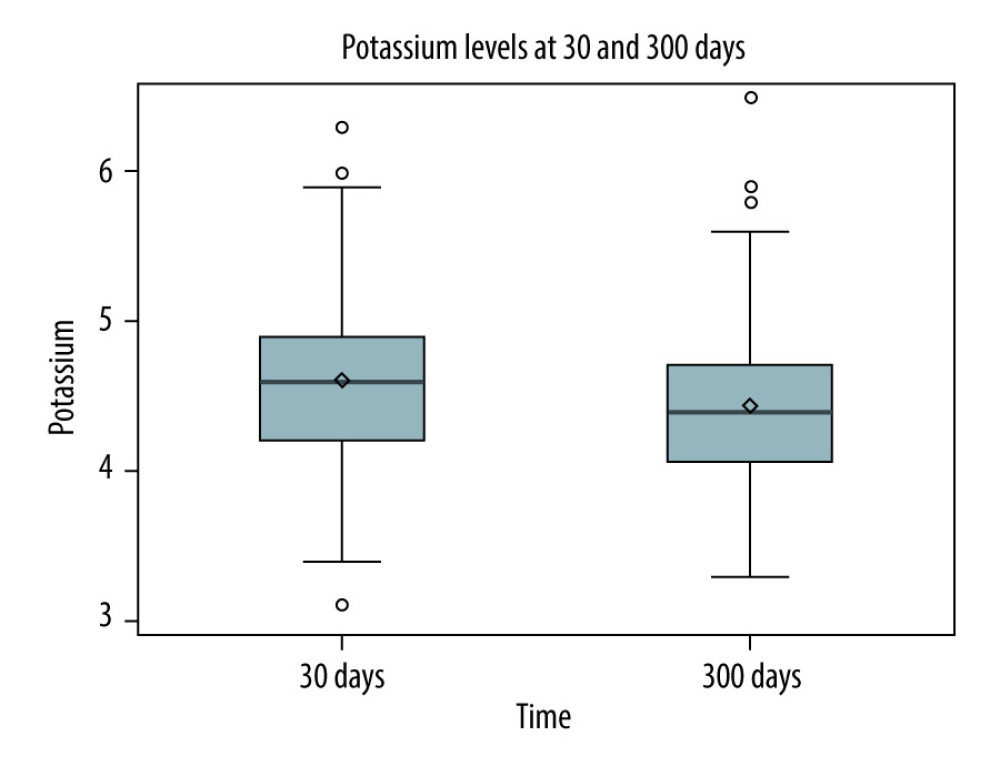 Figure 4. Potassium levels 30 days post-transplant (high tacrolimus exposure) versus 300 days post-transplant (low tacrolimus exposure)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 4. Potassium levels 30 days post-transplant (high tacrolimus exposure) versus 300 days post-transplant (low tacrolimus exposure)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. Tables
Table 1. Baseline characteristics off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).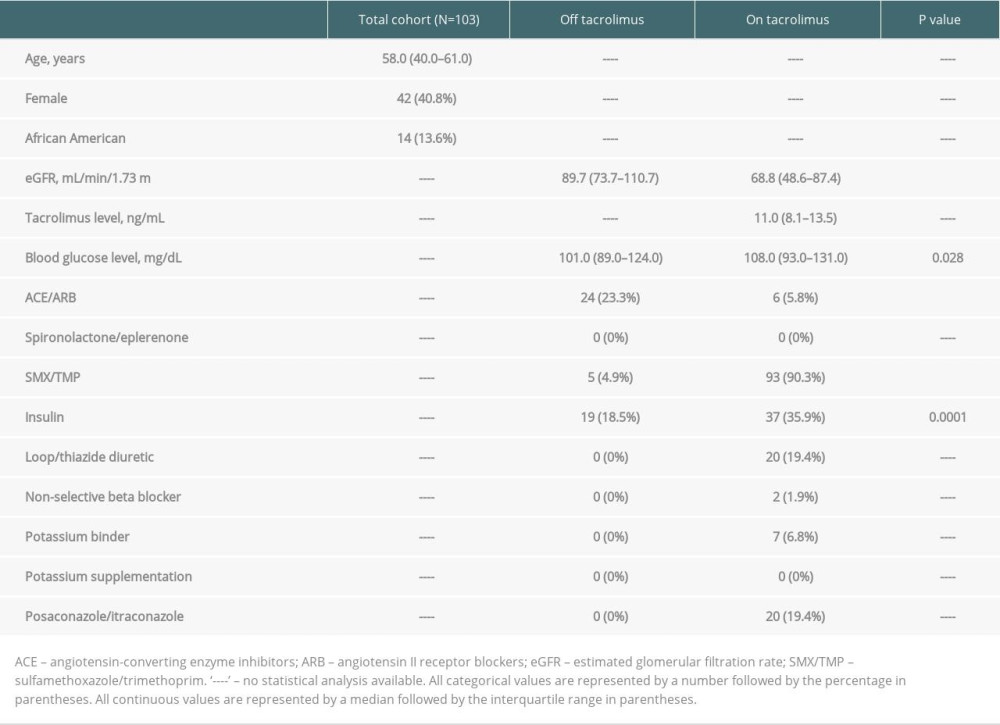 Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).
Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).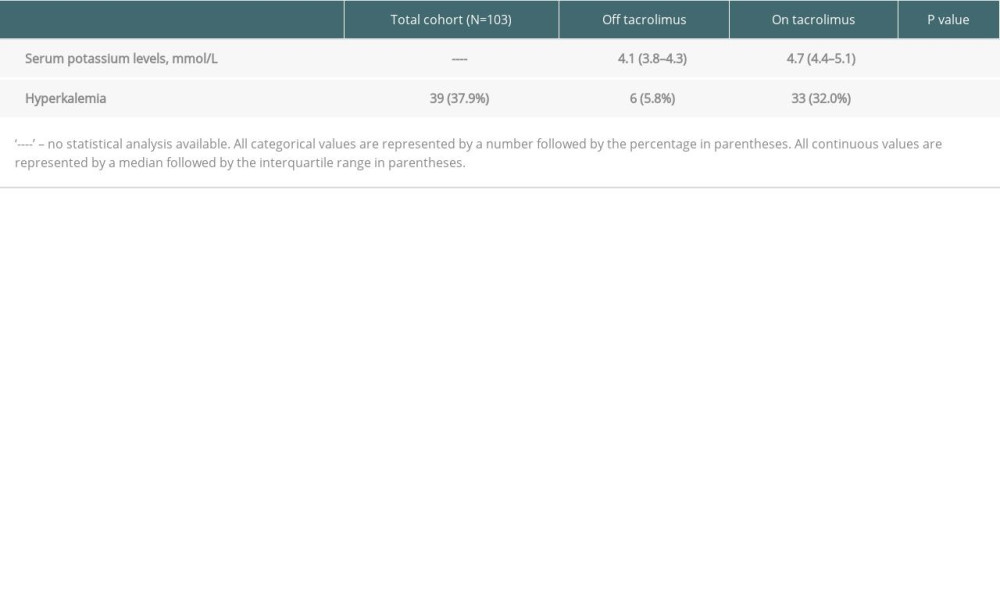 Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant).
Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant).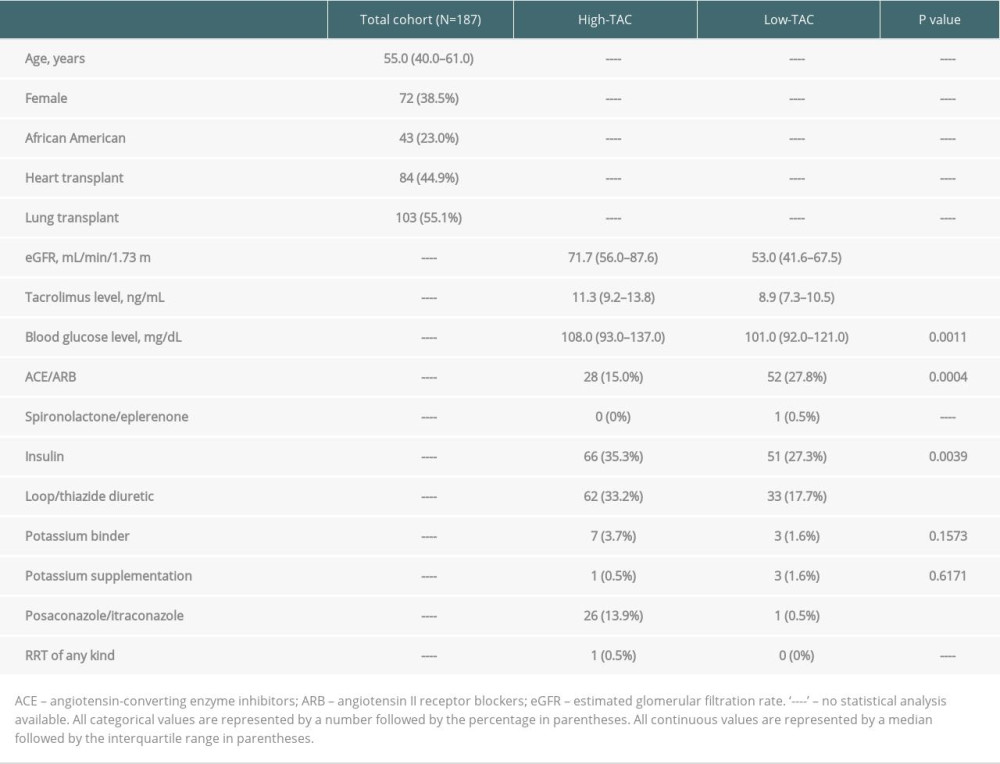 Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant).
Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant).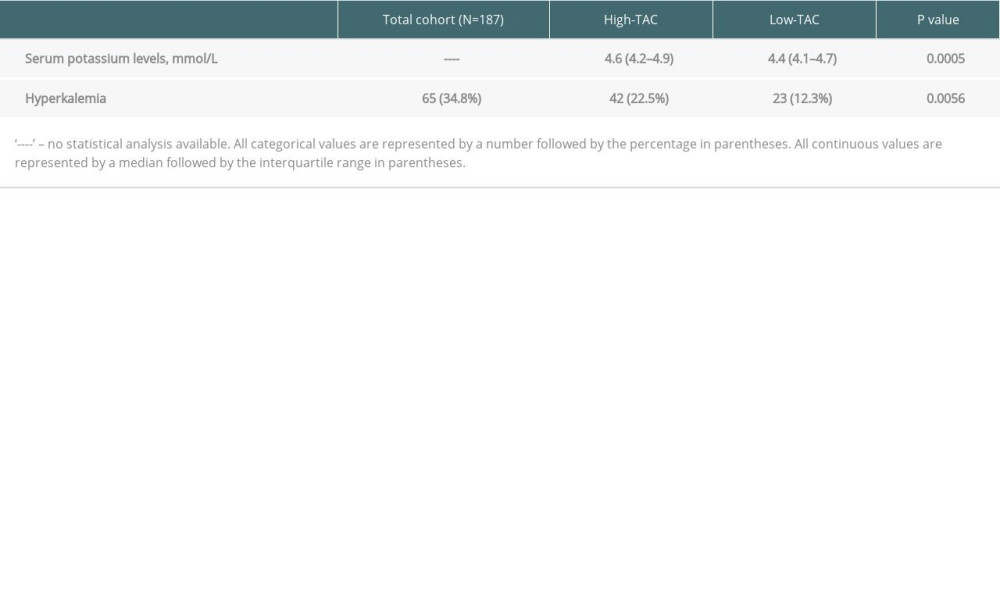
References
1. Kobashigawa J, Clinical trials in heart transplantation: The evolution of evidence in immunosuppression: J Heart Lung Transplant, 2017; 36(12); 1286-90
2. McDermott JK, Girgis RE, Individualizing immunosuppression in lung transplantation: Glob Cardiol Sci Pract, 2018; 5; 1-22
3. Kush KK, Cherikh WS, Chambers DC, The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty sixth adult heart transplantation report – 2019;focus theme: Donor and recipient size match: J Heart Lung Transplant, 2019; 38(10); 1056-66
4. Chambers DC, Cherikh WS, Harhay MO, The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty sixth adult lung and heart–lung transplantation Report – 2019;Focus theme: Donor and recipient size match: J Heart Lung Transplant, 2019; 38(10); 1042-55
5. Brunet M, van Gelder T, Åsberg A, Therapeutic drug monitoring of tacrolimus-personalized therapy: Second consensus report: Ther Drug Monit, 2019; 41(3); 261-307
6. Trofe-Clark J, Brennan DC, West-Thielke P, Results of ASERTAA, a randomized prospective crossover pharmacogenetic study of immediate-release versus extended-release tacrolimus in African American kidney transplant recipients: Am J Kidney Dis, 2018; 71(3); 315-26
7. Whalen HR, Glen JA, Harkins V, High intrapatient tacrolimus variability is associated with worse outcomes in renal transplantation using a low-dose tacrolimus immunosuppressive regime: Transplantation, 2017; 101(2); 430-36
8. Ensor CR, Iasella CJ, Harrigan KM, Increasing tacrolimus time-in-therapeutic range is associated with superior one-year outcomes in lung transplant recipients: Am J Transplant, 2018; 18(6); 1527-33
9. Costanzo MR, Dipchand A, Starling R, The International Society of Heart and Lung Transplant Guidelines for the care of heart transplant recipients: J Heart Lung Transplant, 2010; 29(8); 914-56
10. Yamada M, Meganathan K, Kaur T, Incidence and outcomes of hyperkalemia in solid organ transplant recipients. Abstract SA-OR034
11. Singh J, Kichloo A, Vipparla N, Hyperkalemia: Major but still understudied complication among heart transplant recipients: World J Transplant, 2021; 11(6); 203-11
12. Uriel M, Holzhauser L, Nguyen A, Incidence and clinical significance of hyperkalemia following heart transplantation: Transplant Proc, 2021; 53(2); 673-80
13. Rizk A, Quan D, Gabardi S, Novel approaches to management of hyperkalaemia in kidney transplantation: Curr Opin Nephrol Hypertens, 2021; 30(1); 27-37
14. Sahu MK, Singh SP, Das A, High blood tacrolimus and hyperkalemia in a heart transplant patient: Ann Card Anaesth, 2017; 20(2); 270-71
15. Hoorn EJ, Walsh SB, McCormick JA, The calcineurin inhibitor tacrolimus activates the renal sodium chloride cotransporter to cause hypertension: Nat Med, 2011; 17(10); 1304-9
16. Heering PJ, Kurschat C, Vo DT, Aldosterone resistance in kidney transplantation is in part induced by a down-regulation of mineralocorticoid receptor expression: Clin Transplant, 2004; 18(2); 186-92
17. Higgins R, Ramaiyan K, Dasgupta T, Hyponatremia and hyperkalemia are more frequent in renal transplant recipients treated with tacrolimus than with cyclosporin. Further evidence for differences between cyclosporin and tacrolimus nephrotoxicities: Nephrol Dial Transplant, 2004; 19(2); 444-50
18. Ribeiro HS, Oliveira MC, Anastácio LR, Prevalence and risk factors of hyperkalemia after liver transplantation: ABCD Arq Bras Cir Dig, 2018; 31(1); e1357
19. Gullestad L, Iversen M, Mortensen SA, Everolimus with reduced calcineurin inhibitor in thoracic transplant recipients with renal dysfunction: A multicenter, randomized trial: Transplantation Apr 15, 2010; 89(7); 864-72
Figures
 Figure 1. Study population of part IFigure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 1. Study population of part IFigure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. Figure 2. Potassium levels pre-transplant (no tacrolimus) versus 30 days post-transplant (tacrolimus)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 2. Potassium levels pre-transplant (no tacrolimus) versus 30 days post-transplant (tacrolimus)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. Figure 3. Study population of part IISMX/TMP – sulfamethoxazole/trimethoprim. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 3. Study population of part IISMX/TMP – sulfamethoxazole/trimethoprim. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. Figure 4. Potassium levels 30 days post-transplant (high tacrolimus exposure) versus 300 days post-transplant (low tacrolimus exposure)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation.
Figure 4. Potassium levels 30 days post-transplant (high tacrolimus exposure) versus 300 days post-transplant (low tacrolimus exposure)Figure represents the data drawn from a Spearman correlation analysis. The box contains the standard deviation for each data set. The horizontal line in the box represents the median, and the diamond in the box represents the mean. The lines that extend from the box represent the interquartile ranges for each dataset. Finally, any circles outside of the box and lines represent any outliers in the dataset. Figure created with Microsoft Word 2016, manufactured by the Microsoft Corporation. Tables
 Table 1. Baseline characteristics off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).
Table 1. Baseline characteristics off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant). Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).
Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant). Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant).
Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant). Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant).
Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant). Table 1. Baseline characteristics off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).
Table 1. Baseline characteristics off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant). Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant).
Table 2. Outcomes off tacrolimus (pre-transplant) vs on tacrolimus (30-days post-transplant). Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant).
Table 3. Baseline characteristics of high-TAC (30-days post-transplant) vs low-TAC (300-days post-transplant). Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant).
Table 4. Outcomes of high-TAC (30 days post-transplant) vs low-TAC (300 days post-transplant). In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








