17 May 2022: Original Paper
Tacrolimus-Induced Neurotoxicity in Early Post-Liver Transplant Saudi Patients: Incidence and Risk Factors
Dema A. Alissa1ABCDEF*, Delal Alkortas1AE, Mohammed Alsebayel2AE, Rawan Abdullah Almasuood3EF, Wejdan AburasDOI: 10.12659/AOT.935938
Ann Transplant 2022; 27:e935938
Abstract
BACKGROUND: Tacrolimus is a calcineurin inhibitor (CNI) commonly used as an immunosuppressant to prevent the rejection of organ transplants. After liver transplantation, it can cause early neurological complications, known as early calcineurin inhibitor-induced neurotoxicity (ECIIN). Its management requires CNI withdrawal, a measure that can affect post-transplant outcomes, primarily allograft rejection. In addition, it can negatively impact the quality of life. The incidence and risk factor of ECIIN has not been reported in the Saudi population. We investigated the incidence and risk factors of ECIIN after liver transplant in Saudi patients. We also looked at the length of stay in the Intensive Care Unit, hospital, and 30-day mortality as secondary endpoints.
MATERIAL AND METHODS: This was a retrospective cohort study of adult patients on tacrolimus with mild, moderate, or severe neurological events within the first month after liver transplantation at a single center of patients who meet the inclusion criteria and were over age 14 years. A total of 338 patients were included in the analysis, and the sample size was calculated based on a pilot study.
RESULTS: Among 338 liver transplantation patients, 63 patients (19%) developed ECIIN. Forty-eight percent of patients had seizures, 23% had agitation, 21% had psychosis, 10% had severe tremors, 13% had confusion, and 6% developed coma. The median time of the incident to develop ECIIN was 9 (IQR: 5-13.5) days after transplant. Thirty-eight patients were managed by switching to cyclosporine, 12 required a reduction in the dose, and 3 were managed temporarily by discontinuing therapy. Autoimmune hepatitis as an underlying liver disease was one of the statistically significant risk factors (P=0.0311). The median length of hospital stay was 31 (IQR: 21-75.5) days, ICU length of stay was 10 (IQR: 5-20.5) days, and 8 patients died within 30 days after transplant.
CONCLUSIONS: The incidence of ECIIN in Saud Arabia was similar to that reported in other populations with similar risk factors. Electrolyte imbalance, mainly hyponatremia, was significantly associated with developing ECIIN. Therefore, ECIIN may potentially increase hospital and ICU length of stay.
Keywords: calcineurin inhibitors, Liver Transplantation, Saudi Arabia, Tacrolimus, Adolescent, Adult, Cyclosporine, Graft Rejection, Humans, Immunosuppressive Agents, Incidence, Pilot Projects, Quality of Life, Risk Factors
Background
Calcineurin inhibitors (CNI) are commonly used to prevent the rejection of organ transplants. The introduction of CNI transformed post-transplantation immunosuppressant therapy in the 1980s. Tacrolimus is the preferred CNI in orthotopic liver transplantation (OLT). It inhibits calcineurin phosphatase activity, which produces a potent inhibitory effect on T-lymphocyte activation. Tacrolimus has a narrow therapeutic index; subtherapeutic trough levels can lead to graft rejection [1,2].
Early post-liver transplantation (LT) complications remain a significant cause of morbidity and mortality. Early calcineurin inhibitor-induced neurotoxicity (ECIIN) occurs more frequently in LT recipients than in any other solid-organ transplant. It also significantly impacts OLT, worsening quality of life and lengthening hospital stay. ECIIN is suspected when neurological symptoms develop within the first 4 weeks after OLT [3–5].
Calcineurin inhibitors were linked for the first time with a neurotoxic effect in the mid-1990s. These included reversible headaches, vision loss, altered mental status, and seizures associated with computed tomography scans (CT) and magnetic resonance imaging (MRI) abnormalities [6]. Adverse neurological events in the literature range from 8% to 70%. The reason for the wide range is that multiple factors could be involved in the development of ECIIN, in addition to the great heterogeneity among different reports [7,8]. Clinical manifestations range from mild tremor to severe syndrome defined by confusion, agitation, altered level of consciousness, seizure, psychosis, focal symptoms, coma, or leukoencephalopathy, in the absence of other neurologic predisposing causes [9].
ECIIN is considered the most frequent adverse event leading to dose modification of the immunosuppressive regimen [10]. ECIIN can lead to allograft rejection and infections since managing such adverse effects requires adjusting doses or, in some cases, withdrawing CNI. In addition, it can negatively impact the quality of life, and many of its complications persist even after CNI conversion [4,5].
The ECIIN pathological pathway is still poorly understood. Tacrolimus-induced neurotoxicity is diagnosed by eliminating other possible causes. However, neurological symptoms can occur despite appropriate tacrolimus doses or blood levels [11]. For the first time, in 2019, ECIIN was linked to multiple risk factors such as previous hepatic encephalopathy, pre-LT sodium, and donor age. Older donor graft with impaired CNI metabolism was proposed to influence neurotoxicity development [12]. Surgical duration >7 h and post-LT hyponatremia have also been reported as risk factors for ECIIN [5].
Several medications can predispose to neurological symptoms in this patient population, including propofol, carbapenems, and linezolid. For such medications, balancing the need for the drug against the potential neurological risk is required when given concurrently with tacrolimus [13]. Additionally, sepsis was linked to neuroinflammation and was suggested to induce immunosuppression by various mechanisms; however, it has never been proven to be a risk factor for developing ECIIN [14].
Tacrolimus metabolism is affected by multiple factors, including patient genetic variation. Tacrolimus is metabolized predominantly by the CYP3A4 enzyme and to a lesser extent by CYP3A5. Patients who express CYP3A5 have at least 1 wild-type allele, and their metabolite production pattern differs from that of non-expressers. The distribution of the CYP3A5*1 wild-type allele varies among races. It is distributed in 45–73% of African Americans, 25% of Mexicans, 15–35% of Asians, and 5–15% of Whites. This may affect the dosing of tacrolimus required to ensure optimal efficacy and minimize adverse effects [15].
Current evidence suggests a similar frequency of CYP450 between the Kingdom of Saudi Arabia (KSA) and European populations [16].
Although neurological complications of calcineurin inhibitors are known, studies in the Saudi post-LT population are not yet available. Therefore, this study aimed to identify the incidence and risk factors of tacrolimus-induced neurotoxicity in Saudi patients early after LT.
Material and Methods
This study was performed in accordance with the Declaration of Helsinki and was approved by the Research Advisory Council (RAC) of King Faisal Specialist Hospital and Research Centre (KFSH&RC), approval reference number ORA/0276/36. The IRB did not require informed consent as this was not an interventional study, and an exemption was granted.
A retrospective cohort study was conducted of all adult patients who underwent LT at KFSH&RC over the last 14 years. The study included patients with mild, moderate, or severe neurological events (visual disturbance, altered level of consciousness, confusion, psychosis, seizure, leukoencephalopathy, and coma) that appeared in the absence of a neurologic lesion, central nervous system infection, stroke, or hemorrhage within the first month after LT and symptoms improved after holding tacrolimus or switching to cyclosporine. Exclusion criteria were younger than 18 years of age, on cyclosporin, previous history of seizures, currently on antiepileptics, neuro-Wilson’s disease, primary non-function, or patients undergoing transplantation with grade 5 hepatic encephalopathy. We defined neurological events as moderate or severe (visual disturbance, altered level of consciousness, confusion, psychosis, seizure, leukoencephalopathy, and coma). ECIIN was defined as a neurological event that led to discontinuation or holding tacrolimus, the addition of antiepileptic medications after any seizures, and presence of moderate or severe neurological changes evident by electroencephalogram (EEG), brain CT, or MRI in patients who had seizures or a rapid change in mental status. The primary endpoint was the incidence of ECIIN after LT, while ICU length of stay (LOS), hospital LOS, and 30-day mortality were secondary endpoints. The incidence of ECIIN was defined as the number of patients who experienced neurotoxic events while on tacrolimus within the first 30 days after LT. ICU LOS was counted as the number of days between admission and discharge from the ICU (in case of ICU re-admission, the total number of ICU stay was added regardless of the disruption of the stay). The hospital LOS was calculated as the number of days between admission and discharge for transplant procedure (re-admissions were not counted as part of the initial LOS). Finally, death as a result of any cause that occurred within 30 days after the LT was referred to as 30-day mortality.
The KFSH&RC LT protocol management guidelines consisted of tacrolimus-based immunosuppression throughout the study period. All patients received methylprednisolone (250 mg intravenously before the surgery and after graft reperfusion, 500 mg for the patients with autoimmune hepatitis) followed by a daily taper from 100 mg (250 mg for the patients with autoimmune hepatitis) to 20 mg of prednisolone over 5 days. This dose was maintained until 1 month after transplantation. If there were no signs of graft rejection, prednisone was then tapered progressively and stopped within 3–12 months after transplantation.
Tacrolimus was administered on the first day of transplantation at a dose of 0.05–0.1 mg/kg/day every 12 h and then adjusted to maintain blood levels at 7–10 ng/ml within the first 3 months. In patients with serum creatinine levels >130 umol/L before transplantation, tacrolimus administration was delayed until there was a reduction in serum creatinine. In the meantime, patients received induction therapy of basiliximab 6 h after portal vein reperfusion and day 4 after transplantation with prednisone and mycophenolate mofetil. Mycophenolate mofetil was given on the first day or when platelets were >40 at a dose of 500 mg every 12 h and then increased to 750–1000 mg every 12 h to allow lower tacrolimus levels. Cyclosporine was used only in patients who did not tolerate tacrolimus due to severe CNS or other adverse effects attributed to tacrolimus therapy, starting at a dose of 3–5 mg/kg/day every 12 h to achieve blood levels of 100–150 ng/ml during the first 3 months. In case of severe CNS adverse effects attributed to tacrolimus, therapy was held until resolution of adverse effects, then resumed cautiously at a lower dose or switched to cyclosporine. Antimicrobial Prophylaxis and Supplementary Medications is available in Supplementary Data online.
The sample size was calculated based on a pilot study that included 80 patients and an estimated incidence of 23% of ECIIN. Therefore, we estimated the sample size of 388 patients with a valid incidence rate to be within 5 percentage points to produce a study power of 80% and an alpha of 0.05.
Descriptive statistical analyses were carried out on all variables, including median and interquartile (First Quartile-Third Quartile) (IQR) for continuous variables and frequency tabulations for categorical variables. The significance of risk factors on the incidence of ECIIN was carried out using Fisher’s exact test with cross-tabulation since each of the risk factors was binary. The time to develop ECIIN analysis was calculated using the Kaplan-Meier survival curve model.
Results
A total of 338 LT patients were identified, with only 15 patients (5%) undergoing liver re-transplantation. Among the total cohort, 131 patients (39%) were females, with a median age of 53.6 (IQR: 44.57–59.81) years (Table 1).
Sixty-three (19%) of the 338 patients developed ECIIN, of which 14.2% developed neurotoxicity within the first 2 weeks of transplantation with a median time of 9 (IQR: 5–13.5) days post-transplant (Figure 1). The median time for tacrolimus initiation for all patients was 2 (IQR: 1–3) days and 2 (IQR: 2–4) days for patients who developed ECIIN. Furthermore, the steady-state level for all patients was 4 (IQR: 2.9–6.45) days and 5.05 (IQR: 2.83–7.45) days for patients who developed ECIIN, which was not statistically significant (P=0.1484). The most common type of neurotoxicity was seizures, which occurred in 30 patients (48%), followed by agitation in 15/63 (23%) (Table 2). Thirty-eight patients (60%) were managed by switching tacrolimus to cyclosporine, 12 (19%) required a reduction in the dose, 3 (4.7%) were managed temporarily by discontinuing therapy, and 10 patients (15.8%) did not require any change in their immunosuppressive therapy (Table 3).
A total of 8 patients died within 30 days after transplant. Four of them died within 14 days, and only 3 patients who developed ECIIN died within 30 days (P=0.376) (Table 4). The hospital median LOS was 19 (IQR: 13–31) days for all patients, while patients who developed ECIIN had a median LOS of 31 (IQR: 21–75.5) days (P≤0.0001) (Figure 2B). The median of ICU LOS was 4 (IQR: 2–9.75) days and 10 (IQR: 5–20.5) days (P≤0.0001) for all patients and those who developed ECIIN, respectively (Figure 2A).
Prior to transplant, we evaluated the association of various cofounders with the development of ECIIN, including primary disease, disease severity stage, MELD (model for end-stage liver disease) score, history of hepatic cellular carcinoma (HCC), history of hepatic encephalopathy, history of ascites, ICU admission prior to transplant, electrolytes, fasting lipids, and history of chronic diseases. Only history of hepatic encephalopathy and MELD score were associated with ECIIN among these cofounders (P=0.0135, P=0.0020, respectively). The only primary disease associated with ECIIN was autoimmune hepatitis (AIH) (P=0.0311). Univariate analysis was performed to assess the association with various confounding factors (Table 1).
Perioperative variables included warm ischemia time (WIT), cold ischemia time (CIT), and blood and blood products transfusion. The only statistically significant risk factor was the transfusion of fresh frozen plasma (FFP) (P=0.0344) (Table 1).
Several post-transplant risk factors for ECIIN were evaluated, including sepsis, CMV infection, use of dialysis, graft survival, and new onset of DM or HTN after transplant (Table 1). Electrolytes were reviewed on the seizure day, and only sodium was significantly associated with the developed ECIIN (P=0.0099) (Table 1). In addition, several neurotoxic medications, including antibiotics, were associated with the development of ECIIN (Table 5). Biopsy-proven rejection was not significant, and all patients received the proper immunosuppressant therapy as per the protocol.
Discussion
Neurotoxicity is a known complication after LT, and CNIs have been reported to be a risk factor. The etiology of ECIIN is multifactorial with several possible contributing risk factors, such as recipient age, MELD score, hepatic encephalopathy, tacrolimus dose and levels, and pre-transplant sodium levels [5,7,12,17,18]. Due to the variation in genetic polymorphism in CYP3A5 among races, tacrolimus metabolism and levels may vary widely. Data on the incidence and possible risk factors in non-White, non-Black populations are scarce. Very little is known about CYP3A5 polymorphism in the Saudi population undergoing LT. Our study explored the incidence and risk factors of post-LT ECIIN in KSA. We found a similar incidence rate to others reported globally [5,7,17]. A retrospective study in China found a significant association between tacrolimus concentration and the genetic polymorphism of CYP enzymes in post-liver transplant patients [19].
Hepatic encephalopathy was previously reported as a risk factor for ECIIN, similar to our findings [5,18]. However, autoimmune hepatitis as a primary cause of liver cirrhosis prior to transplant was found to be associated with an increased risk of post-LT ECIIN in our study. To the best of our knowledge this has not been described before. However, further studies are warranted to confirm this association.
In our cohort, most patients who experienced ECIIN were managed by either switching tacrolimus to cyclosporine or by reducing tacrolimus dose. Tacrolimus levels have been linked to post-transplant neurotoxicity [5,11,20]. Balderramo et al reported high tacrolimus levels (>15 ng/ml) in 21.5% of ECIIN-positive patients. In our study, there was no difference in tacrolimus average steady-state levels between the patients who developed ECIIN and those who did not. Tacrolimus dose modifications should be minimized if possible since it can lead to acute rejection in the immediate post-transplant phase. Hence, early neurological monitoring and intervention are advised.
Other neurotoxic agents were also evaluated in this study; carbapenems, linezolid, midazolam, propofol, haloperidol, lorazepam, risperidone, and olanzapine were associated with seizures. In addition, electrolyte imbalance was reported as a risk factor in other studies [5,7,12,17], namely hyponatremia.
To the best of our knowledge, this is the first report in the Arab world to study the incidence and risk factors of tacrolimus-induced neurotoxicity in the early post-LT period. Our study evaluated all previously reported risk factors in addition to several others that have not been reported before. In addition, the sample size included in our study was relatively larger compared to other studies.
The main limitation of this study is its retrospective design and that it was conducted in a single tertiary care hospital. We also did not test for the effect of CYP3A5 genotyping due to the study’s retrospective nature.
Conclusions
The incidence of ECIIN in KSA was similar to that reported in other populations with similar risk factors. Electrolyte imbalance, mainly hyponatremia, was significantly associated with developing ECIIN. ECIIN can increase hospital and ICU length of stay. Further studies are needed regarding the CYP polymorphism in the Saudi population and its clinical impact on the development of neurotoxicity. This is particularly important in the interaction of enhanced recovery after surgery (ERAS), ECIIN, and genetic polymorphism.
Figures
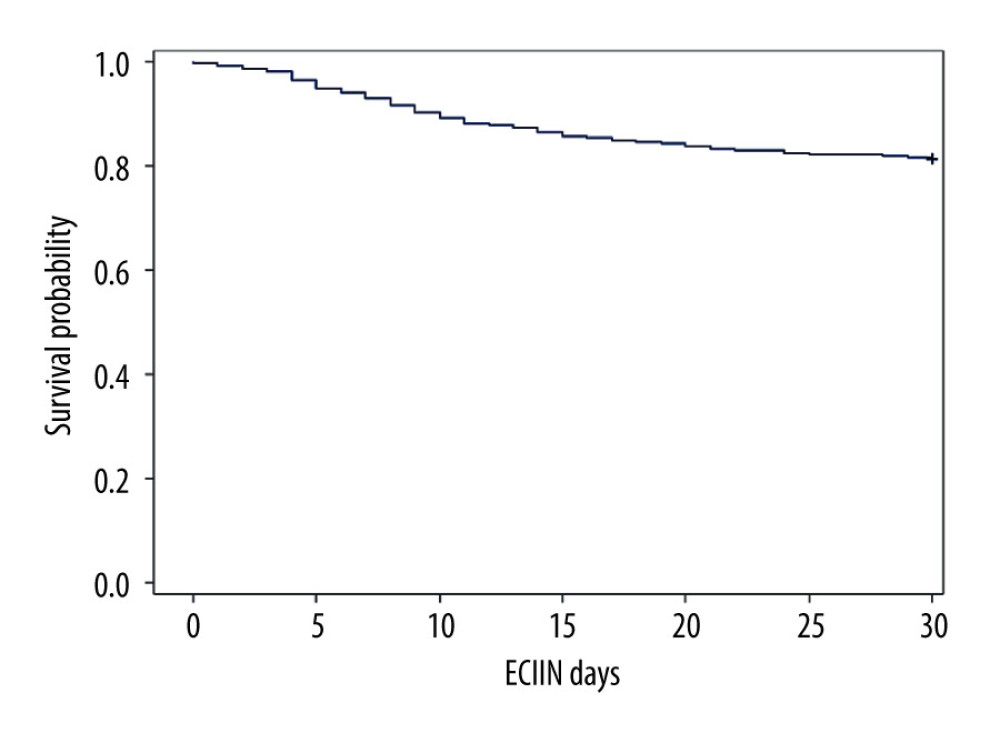 Figure 1. Time-to-event analysis for liver transplant recipients who developed ECIIN using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc.
Figure 1. Time-to-event analysis for liver transplant recipients who developed ECIIN using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. 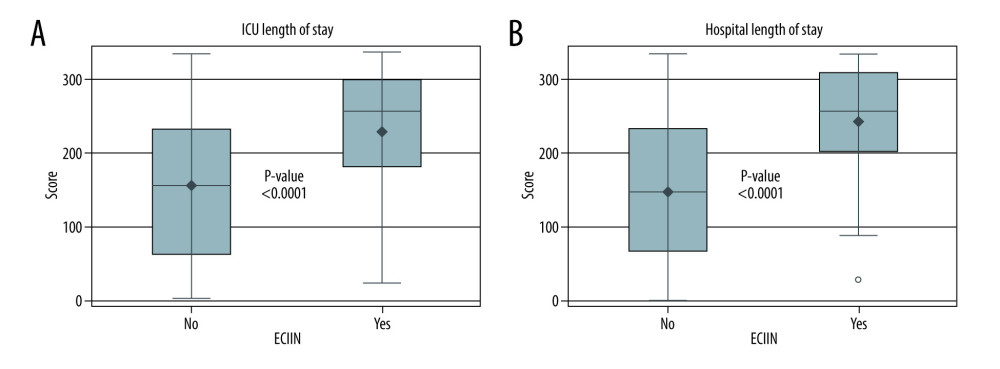 Figure 2. (A) Distribution of Wilcoxon score for ICU length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. (B) Distribution of Wilcoxon score for hospital length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc.
Figure 2. (A) Distribution of Wilcoxon score for ICU length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. (B) Distribution of Wilcoxon score for hospital length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. Tables
Table 1. Baseline characteristics and confounders associated with the development of early calcineurin inhibitor-induced neurotoxicity (ECIIN) using univariate analysis.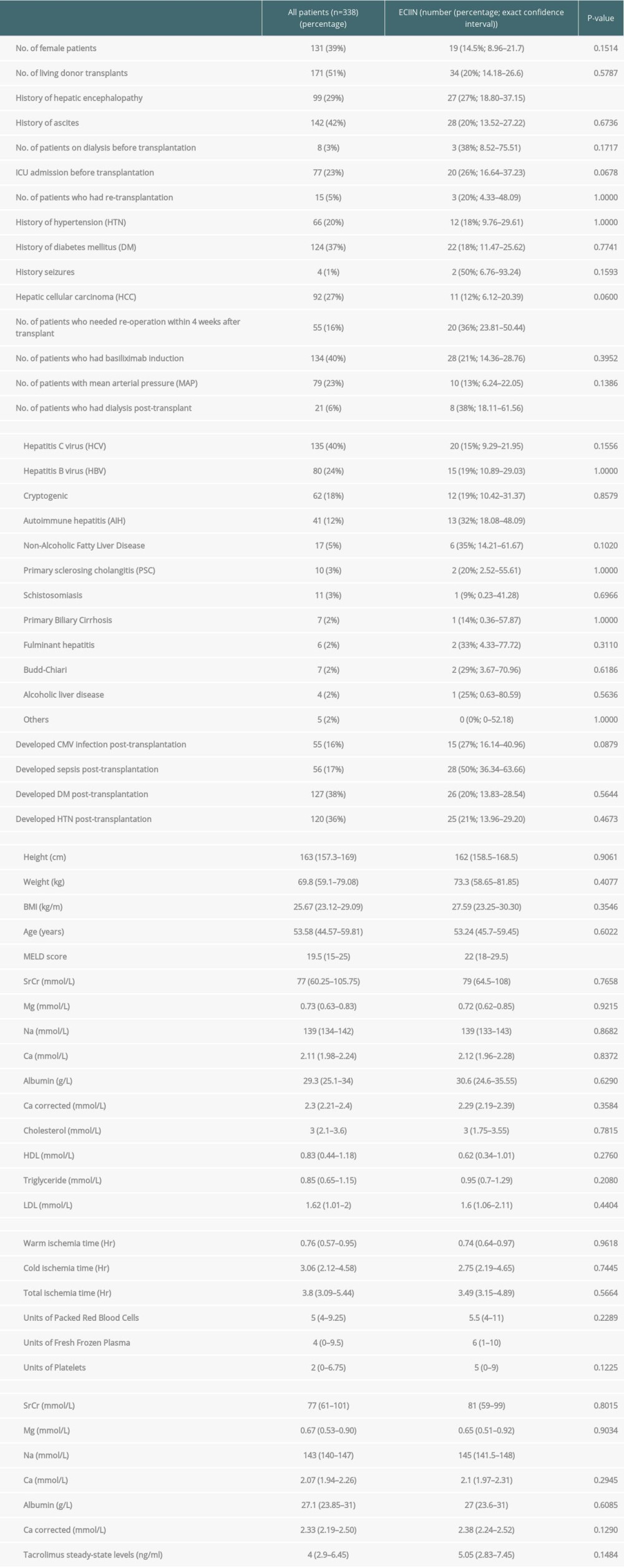 Table 2. Incidence and types of ECIIN.
Table 2. Incidence and types of ECIIN.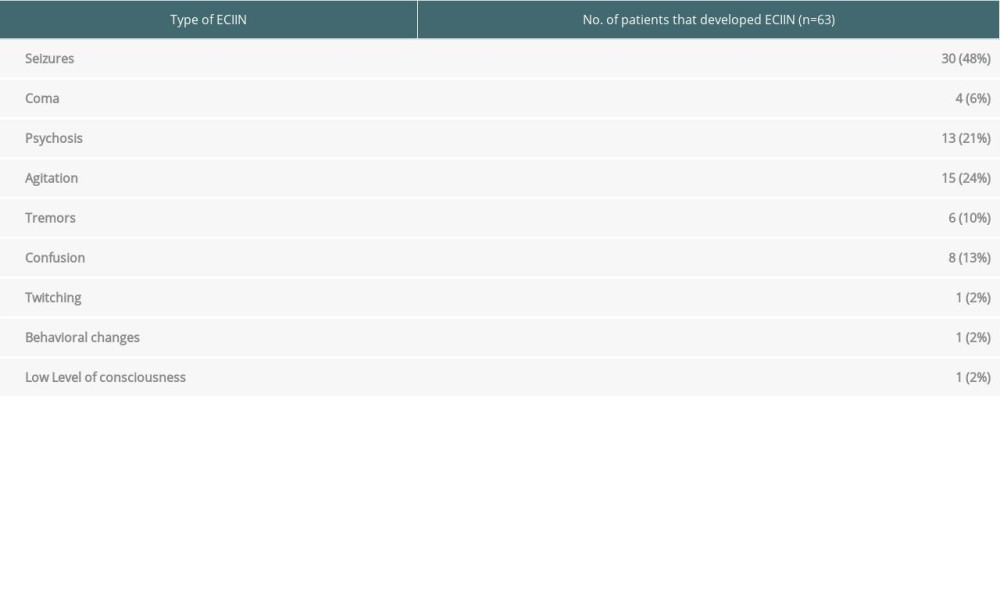 Table 3. Management for liver transplant recipients who developed ECIIN.
Table 3. Management for liver transplant recipients who developed ECIIN.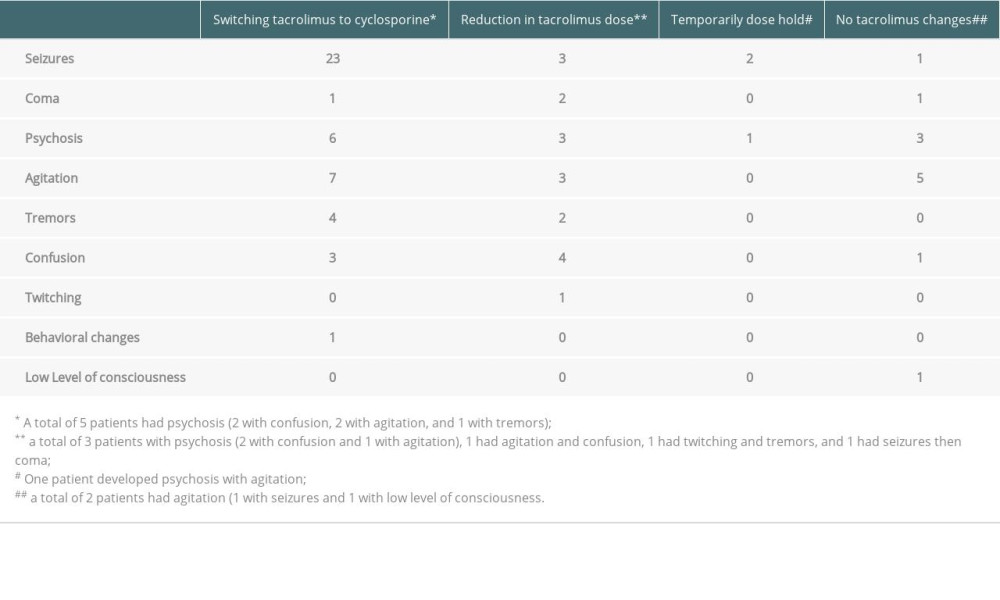 Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN.
Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN.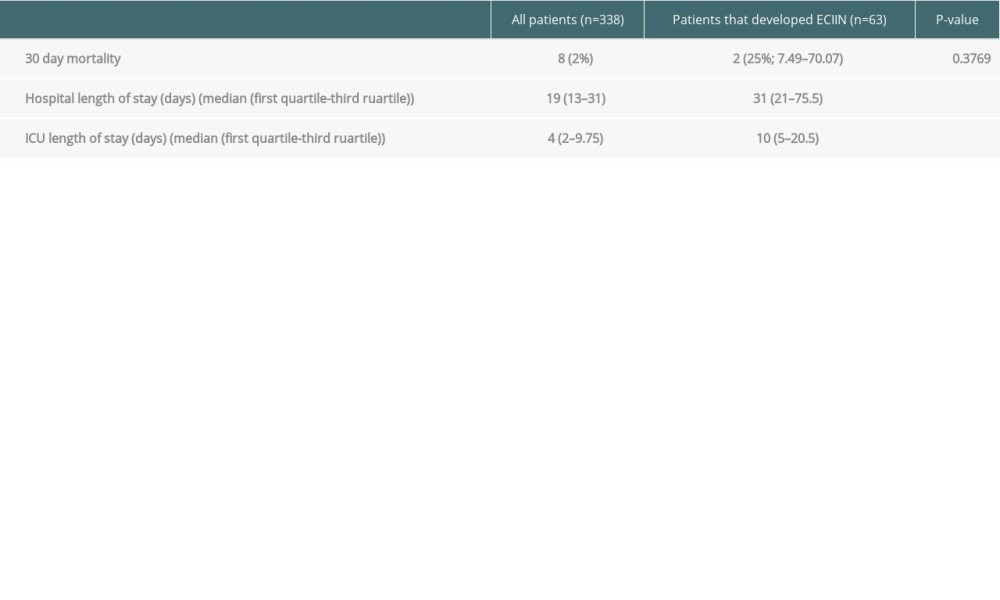 Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN.
Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN.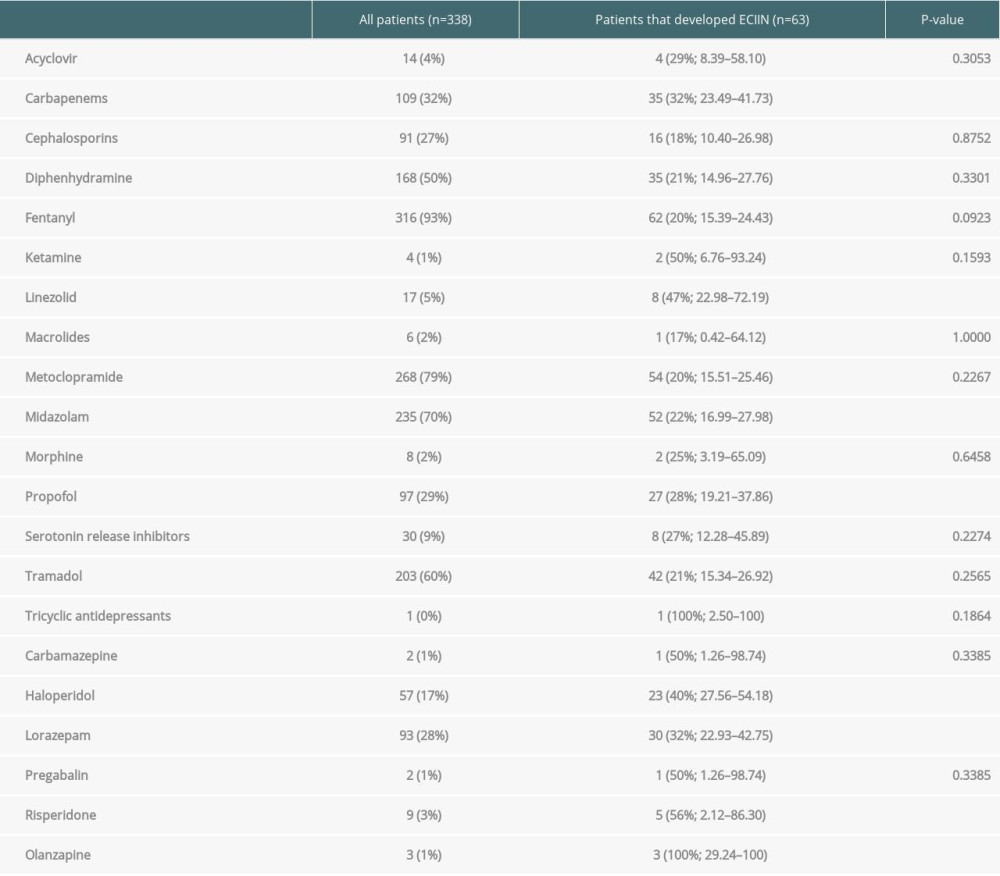
References
1. Lock JF, Malinowski M, Schwabauer E, Initial liver graft function is a reliable predictor of tacrolimus trough levels during the first post-transplant week: Clin Transplant, 2011; 25(3); 436-43
2. Neves DB, Rusi MB, Diaz LG, Salvalaggio P, Primary graft dysfunction of the liver: Definitions, diagnostic criteria and risk factors: Einstein (Sao Paulo), 2016; 14(4); 567-572
3. Senzolo M, Ferronato C, Burra P, Neurologic complications after solid organ transplantation: Transpl Int, 2009; 22(3); 269-78 [Erratum in: Transpl Int. 2009;22(4):507]
4. Mueller AR, Platz KP, Bechstein WO, Neurotoxicity after orthotopic liver transplantation. A comparison between cyclosporine and FK506: Transplantation, 1994; 58(2); 155-70
5. Balderramo D, Prieto J, Cárdenas A, Navasa M, Hepatic encephalopathy and post-transplant hyponatremia predict early calcineurin inhibitor-induced neurotoxicity after liver transplantation: Transpl Int, 2011; 24(8); 812-19
6. Hinchey J, Chaves C, Appignani B, A reversible posterior leukoencephalopathy syndrome: N Engl J Med, 1996; 334(8); 494-500
7. Lewis MB, Howdle PD, Neurological complications of liver transplantation in adults: Neurology, 2003; 61; 1174
8. Marco S, Cecilia F, Patrizia B, Neurologic complications after solid organ transplantation: Transpl Int, 2009; 22(3); 269-78
9. Bechstein WO, Neurotoxicity of calcineurin inhibitors: Impact and clinical management: Transpl Int, 2000; 13(5); 313-26
10. Abouljoud MS, Anil Kumar MS, Brayman KL, Neoral rescue therapy in transplant patients with intolerance to tacrolimus: Clin Transplant, 2002; 16; 168-72
11. Luzzi S, Demasi M, Lucifero AG, An underrated complication of the organs’ transplantations: Ann Ital Chir, 2020; 91; 298-309
12. Lué A, Martinez E, Navarro M, Donor age predicts calcineurin inhibitor induced neurotoxicity after liver transplantation: Transplantation, 2019; 103(8); e211-15
13. Braksick SA, Nasr DM, Neurological emergencies from prescription drugs: Neurological Emergencies, 2020; 301-18
14. Michels M, Sonai B, Dal-Pizzol F, Polarization of microglia and its role in bacterial sepsis: J Neuroimmunol, 2017; 303; 90-98
15. Provenzani A, Santeusanio A, Mathis E, Pharmacogenetic considerations for optimizing tacrolimus dosing in liver and kidney transplant patients: World J Gastroenterol, 2013; 19(48); 9156-73
16. Almeman AA, Major CYP450 polymorphism among Saudi patients: Drug Metab Lett, 2021; 14(1); 17-24
17. DiMartini A, Fontes P, Dew MA, Age, model for end-stage liver disease score, and organ functioning predict posttransplant tacrolimus neurotoxicity: Liver Transpl, 2008; 14(6); 815-22
18. Sastre L, Crespo G, Neurotoxicity after liver transplantation: Does donor age matter?: Transl Gastroenterol Hepatol, 2021; 6; 66
19. Dong Y, Xu Q, Li R, CYP3A7, CYP3A4, and CYP3A5 genetic polymorphisms in recipients rather than donors influence tacrolimus concentrations in the early stages after liver transplantation: Gene, 2022; 809; 146007
20. de Sousa Arantes Ferreira G, de Conde Watanabe AL, Carvalho Trevizoli N, Tacrolimus-associated psychotic disorder: A report of 2 cases: Transplant Proc, 2020; 52; 1350-53
Figures
 Figure 1. Time-to-event analysis for liver transplant recipients who developed ECIIN using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc.
Figure 1. Time-to-event analysis for liver transplant recipients who developed ECIIN using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. Figure 2. (A) Distribution of Wilcoxon score for ICU length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. (B) Distribution of Wilcoxon score for hospital length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc.
Figure 2. (A) Distribution of Wilcoxon score for ICU length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. (B) Distribution of Wilcoxon score for hospital length of stay using SAS Institute, Inc. 2015. SAS/IML® 14.1 User’s Guide. Cary, NC: SAS Institute, Inc. Tables
 Table 1. Baseline characteristics and confounders associated with the development of early calcineurin inhibitor-induced neurotoxicity (ECIIN) using univariate analysis.
Table 1. Baseline characteristics and confounders associated with the development of early calcineurin inhibitor-induced neurotoxicity (ECIIN) using univariate analysis. Table 2. Incidence and types of ECIIN.
Table 2. Incidence and types of ECIIN. Table 3. Management for liver transplant recipients who developed ECIIN.
Table 3. Management for liver transplant recipients who developed ECIIN. Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN.
Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN. Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN.
Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN. Table 1. Baseline characteristics and confounders associated with the development of early calcineurin inhibitor-induced neurotoxicity (ECIIN) using univariate analysis.
Table 1. Baseline characteristics and confounders associated with the development of early calcineurin inhibitor-induced neurotoxicity (ECIIN) using univariate analysis. Table 2. Incidence and types of ECIIN.
Table 2. Incidence and types of ECIIN. Table 3. Management for liver transplant recipients who developed ECIIN.
Table 3. Management for liver transplant recipients who developed ECIIN. Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN.
Table 4. Secondary endpoints for liver transplant recipients who developed ECIIN. Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN.
Table 5. Neurotoxic agents used for liver transplant recipients who developed ECIIN. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








