07 June 2022: Original Paper
Phosphatidylethanol (PEth) for Monitoring Sobriety in Liver Transplant Candidates: Preliminary Results of Differences Between Alcohol-Related and Non-Alcohol-Related Cirrhosis Candidates
Jan-Paul Gundlach1EF*, Felix Braun1AD, Finn Mötter1B, Alexander Bernsmeier1CDE, Pablo Barrio2E, Nicola Ehmke1B, Rainer Günther3B, Holger Hinrichsen4A, Thomas Becker1AD, Wolfgang WeinmannDOI: 10.12659/AOT.936293
Ann Transplant 2022; 27:e936293
Abstract
BACKGROUND: Monitoring sobriety is mandatory for liver transplant (LT) candidates with alcohol-related cirrhosis in Germany. Prior to listing, abstinence of 6 months is required. However, little is known about biomarker performance in alcohol-related cirrhosis. Routine testing of ethyl glucuronide in urine (uEtG) or hair (hEtG) is prone to manipulation or is unfeasible in anuria. Phosphatidylethanol (PEth) in dried-blood spots is a promising alternative. We compared PEth with routine parameters and self-reports in alcohol-related and non-alcohol-related cirrhosis at our transplant center.
MATERIAL AND METHODS: All patients received self-report questionnaires (AUDIT & TLFB). Blood, urine and hair samples, as well as PEth dried-blood spots were drawn at baseline. In addition, survival analyses were conducted.
RESULTS: Out of 66 patients, 53 were listed for LT and 13 were candidates not listed so far. An alcohol-use disorder was found in 25 patients. Positive results for uEtG, hEtG, and PEth were found in 5/65, 9/65, and 34/66 cases, respectively. PEth positivity was found in 52% of patients with alcohol-related cirrhosis, while 53% of patients with other liver diseases were positive. While uEtG, hEtG, and TLFB correlated with higher PEth values, active waiting list status was significantly correlated with negative PEth values. During the mean follow-up of 41.15 months, 23 patients were transplanted (34.9%). None of the biomarkers significantly predicted survival.
CONCLUSIONS: PEth can importantly assist abstinence monitoring in LT candidates due to its high validity and objectivity. The high percentage of patients with alcohol consumption in the non-alcoholic liver disease cohort underscores the importance of testing all transplant candidates.
Keywords: Ethyl Glucuronide, Liver Diseases, Alcoholic, Liver Transplantation, Phosphatidylethanol, Alcohol Drinking, Glycerophospholipids, Humans, Liver Cirrhosis, Alcoholic
Background
LIVER TRANSPLANTATION IN ALCOHOL-RELATED LIVER DISEASE:
Alcohol-related liver disease accounted for the majority of liver transplantations (LT) in Germany in 2019 [1]. Alcoholic steatohepatitis (ASH) induces fibrosis and can progress to liver cirrhosis, while alcohol abstinence can improve ASH with regressive fibrosis. In end-stage cirrhosis, LT is a well-established and valuable procedure [2], but relapse into drinking can limit graft survival [3]. Also, the donor organ shortage limits the access to a graft. Therefore, the gap between need and demand requires allocation rules, transparency, and fairness [4].
Alcohol-related cirrhosis has been intensively debated as an eligible indication over the last 30 years. The conflict of accepting these patients for LT was concordantly solved by the introduction of the 6-month rule [5]. This rule has 2 intentions. First, it enables identification of candidates that recover under sobriety. Second, it allows proof of compliance and medical adherence prior to LT and is a selection tool for candidacy [6]. Of note, recently, similar relapse rates among patients undergoing early LT with sobriety less than 6 months versus standard LT with ALD were demonstrated [7]. Taking all that into account, proper abstinence monitoring has become paramount among LT evaluation procedures. While questionnaires and self-reports are always valuable in such undertakings, they yield relevant bias that might be overcome by biological markers of alcohol consumption [8,9].
MONITORING SOBRIETY:
Routinely performed laboratory tests are available for determination of alcohol consumption. Indirect markers such as gamma-glutamyl-transpeptidase (μGT), mean corpuscular volume (MCV), and carbohydrate-deficient transferrin (CDT) are well known to have limitations in various settings. Direct ethanol metabolites are important as they are biomarkers with high sensitivity and specificity. Most frequently, ethyl glucuronide (EtG), phosphatidylethanol (PEth), and, to a lesser extent, ethyl sulfate (EtS), are used in various settings [10]. EtG and EtS are metabolites of ethanol. Depending on the amount of alcohol consumed and time after consumption, EtG is still detectable in the body after completion of alcohol elimination [11–13], but its detection is limited to 130 h after ethanol intake [14]. Although EtG concentration in urine (uEtG) is regularly determined [15], its detection can be hindered by oligo-anuria in advanced liver failure [16]. The criterion standard is hairEtG (hEtG) [17], which has been proposed to be highly specific and useful for the monitoring of alcohol use before LT [18]. However, hair and urine probes can be influenced by various factors and can be subject to manipulation [19–21].
A promising alternative is PEth. The precursors are naturally prevalent phosphatidylcholine homologues – PEth16: 0/18: 1 and 16: 0/18: 2 – which are the most prevalent ones in human blood [22]. PEth is formed after ingestion of alcohol and can be detected up to 12 days after a single drinking event [23]. PEth has been employed in numerous settings [24], including LT [24–26] and is recommended in various contexts in the 2020 German evidence- and consensus-based S3 guideline on alcohol [27]. Furthermore, it is reported to have higher sensitivity compared to uETG and EtS [28].
ALCOHOL TESTING IN NON-ALCOHOL-RELATED LIVER DISEASE:
However, some concerns remain in the current literature. First, head-to-head comparisons among direct alcohol biomarkers remain scarce in transplant candidate studies. For example, only 2 previous studies, with limited sample size, have directly compared PEth to EtG in transplant candidates [24,26]. Therefore, new studies are needed to identify the analytic ability of diverse direct alcohol biomarkers. Second, while alcohol-related liver disease (ALD) has historically accounted for the majority (up to 30%) of LT worldwide [29], nowadays, the spectrum of LT indications is broad and includes progressive and irreversible acute or chronic liver diseases or genetic diseases that are life-threatening or extremely limiting quality of life and can be cured by LT. For example, estimates in the US suggest NASH (non-alcoholic steatohepatitis) has become the second leading cause of LT, surpassing ALD, accounting for 13% of the total transplantations [30]. European reports describe a similar increase over the last years [31]. Therefore, more information is needed regarding diagnostic performance of alcohol biomarkers in these populations. While alcohol abstinence is not mandatory in many guidelines for patients with non-alcohol-related cirrhosis awaiting LT, we believe alcohol monitoring with direct alcohol biomarkers could provide significant information all through the peri-transplantation procedures. Two reasons for this can be argued. First, alcohol use might have gone undetected, and second, similar to alcohol-related cirrhosis, significant and lasting life-style changes must be achieved to avoid disease recurrence and graft loss. In that sense, alcohol biomarkers could also be considered as indictors of such endeavors.
In this study we analyzed the diagnostic performance of PEth in the evaluation of candidates awaiting LT and compared the results with uETG, hETG, modified time line follow back (TLFB) questionnaire [32], and the Alcohol Use Disorders Identification Test (AUDIT) [33]. The 2 methods are recommended by various international guidelines, such as the Australian, the NHS, and the German guideline on alcohol use disorders [34]. We further compared the results between alcohol-related and non-alcohol-related candidates.
Material and Methods
DETERMINATION OF ETG IN HAIR (HETG):
The technique utilized for extraction of the hair specimen has been demonstrated before [35]. Briefly, OASIS Max Columns (Waters S.A.S., Saint-Quentin, France) was used for phase extraction, after the hair probes were cleaned with water, acetone, pulverization, and incubation for 2 h in an ultrasonic bath. Probes were derivatized with heptafluorobutyric. In negative chemical ionization, anhydride analysis was conducted by the use of GC-MS/MS. Documented ion changeovers were m/z 596/213 (quantifier) and 397/213 (qualifier) for EtG, and m/z 601/213 for EtG-D5. The detection limit (LOD) was 0.05 pg/mg hair and the lower limit of quantification (LLOQ) was set at 0.2 pg/mg hair [36]. EtG and EtS were analyzed according to a published LC-MS/MS procedure using the QTrap 3200 LC-MS/MS system [37].
DETERMINATION OF PHOSPHATIDYLETHANOL (PETH) IN DRIED BLOOD SPOTS (DBS) PREPARED FROM VENOUS BLOOD:
Analyses of PEth 16: 0/18: 1 and PEth 16: 0/18: 2 were conducted in DBS. Internal standards were D5-PEth 16: 0/18: 1 and D5-PEth 16: 0/18: 2. Deuterated standards were established at our research center from phosphatidylcholine 16: 0/18: 1 and phosphatidylcholine 16: 0/18: 2 and D6-ethanol metabolized by phospholipase D as previously described [23]. DBS were prepared as follows: 20 μL of whole blood were placed on filter cards (GR2261004, PKI 226 Bioanalysis Card, Perkin Elmer, Rodgau, Germany) and dehydrated for at least 3 h. Then, PEth was separated from DBS using 0.5 mL methanol for 10 min. The supernatant was moved to a vial and evaporated to dryness in a 50°C current of nitrogen. The extract was resuspended in 0.2 mL mobile phase A (10 mM ammonium acetate/30: 70 v/v acetonitrile). Next, 80 μL eluate were inserted in the online-SPE-LC-MS/MS appliance.
We modified an earlier-described PEth analysis technique validated for whole blood samples [23] for the use of DBS by standardization at 20–2000 ng/mL with a QTrap 3200 tandem mass spectrometer, which includes a turbo ionspray source (Sciex, Toronto, Canada). The limit of quantitation (LoQ) for PEth 16: 0/18: 1 and PEth 16: 0/18: 2 was 20 ng/mL. Probes were trapped by using a Synergi Polar-RP column (20×2 mm, 5 μm). Then, the 2 probes in the eluate were divided with a Luna RP-C5 column (50×2 mm, 5 μm; Phenomenex, Brechbühler, Schlieren, Switzerland) for differential extraction.
STATISTICAL ANALYSIS:
Statistical analysis was performed with SPSS (Version 25.0, SPSS Inc., Chicago, IL, USA). Collected data are represented as value, percentage, or mean with standard deviation. PEth findings were dichotomized into positive and negative results. The association of examined parameters and EtG test results with a positive PEth finding was tested by Pearson and Spearman correlation coefficient. Independency and influence of factors were determined by binary logistic regression analysis. Significant values were then entered into multiple binary logistic regression analysis to perform multivariate testing. The impact of tested parameters concerning the expression of PEth values was tested by linear regression analysis. The association of PEth and other biomarkers’ positivity with survival was estimated with Kaplan-Meier survival curves and the log-rank test. A
Results
BASELINE ASSESSMENTS:
In total, 66 patients (31 female; mean age 53.29±12.0; range 21–74 years) were enrolled in the study. The diagnoses included cases as follows (Figure 1): 21 alcohol-related cirrhosis, 11 hepatocellular carcinoma (HCC, 4 of them with alcohol-related cirrhosis), 8 primary sclerosing cholangitis (PSC), 6 cryptogenic cirrhosis, 5 hepatitis C virus (HCV), 3 autoimmune hepatitis (AIH), 3 NASH, 3 primary biliary cirrhosis (PBC), 2 polycystic liver disease (PLD), 2 portal vein thrombosis (PVT), 1 Abernethy-syndrome, and 1 Caroli-syndrome. In total, 25 of 66 patients had the diagnosis of an alcohol use disorder. Patient characteristics and laboratory measurement values are listed in Table 1.
COMPARISON OF SOBRIETY TEST RESULTS:
In our cohort, 5/65 (7.7%) of patients were positive for uEtG, 9/65 (13.8%) were positive for hEtG, and 34/66 (51.5%) were positive for PEth (Table 1). Except for 1 patient with a positive hEtG, all other patients with positive uEtG or hEtG test values also tested positive for PEth. Alcohol consumption was stated in the TLFB questionnaire by 25 patients and 33 patients had ≥1 point in the AUDIT. Table 2 depicts the percentage of positive biomarkers according to the presence of alcoholic- or non-alcohol-related cirrhosis. Regarding PEth, results showed a similar percentage of positives between the groups (52.0% vs 51.2%).
Of the 25 patients with ALD, 11 patients were listed for LT with an active status (“transplantable”, status T). All of these patients were negative for uEtG, which is mandatory according to the German LT guideline. However, 1 of these patients tested positive for hEtG and 4 were positive for PEth. These findings prompted a new interrogation, but no waiting list withdrawal. According to the AUDIT results, 9 patients had ≥1 point and 1 patient reported alcohol consumption 6 months ago in the TLFB. This particular patient was negative for all 3 biomarkers. All other patients did not report alcohol consumption in the TLFB.
Similarly, from a total of 6 patients listed with ALD and inactive status (“not transplantable”, NT), none were positive for uEtG, but 5 were positive for PEth and 3 for hEtG. All patients had ≥1 points in the AUDIT and 2 had a positive self-report for alcohol consumption within the last 2 weeks and 5 patients more than 2 weeks ago. The uEtG-, hEtG-, and PEth-negative patients reported alcohol consumption more than 6 months ago.
Non-listed patients presenting with alcohol-related cirrhosis for evaluation (n=8) had a 12.5% positive rate for both uEtG and hETG, while PEth was positive in 50.0% of the cases. All except 1 patient had ≥1 points in the AUDIT and 4 out of 8 patients reported alcohol consumption more than 1 week and up to 6 months ago. The patient with positive uEtG, hEtG, and PEth stated 66 g/d alcohol consumption within the last month. Importantly, the uEtG test result was below the cut-off (0.5 mg/L) of the German guideline for LT candidates.
Patients with other liver diseases listed for LT had a high rate of positive PEth results (Figure 2). In total, 18/34 (52.9%) patients had a positive PEth, 1/33 (3.0%) had a positive hEtG, and 3/33 (9.1%) had a positive uEtG. All of these patients had 0 points in the AUDIT, while 3 stated alcohol consumption within the last 2 weeks. Another 7 patients stated alcohol consumption more than 2 weeks ago in the TLFB. Out of the 3 patients with alcohol intake within the last 2 weeks, 2 were positive for uEtG and PEth and 1 was positive for hEtG. The patients with alcohol intake more than 6 months ago were negative for all biomarkers.
Out of 5 patients admitted for LT with non-alcohol-related diagnosis, only 1 had ≥1 point in the AUDIT and none stated alcohol consumption within the last 2 weeks. One patient reported alcohol consumption within the last month and 2 stated alcohol consumption more than 6 months ago. However, 1 patient (20.0%) was positive for uEtG and 3 patients (60.0%) were positive for hEtG and PEth.
FOLLOW-UP DATA:
The mean follow-up time was 41.15 months (±20.04 months, range 0–61 months). During that time, 23 patients were transplanted (34.9%). Six of 23 patients died after LT. These deaths were caused by ARDS after repeated LT for primary nonfunction (PNF) on postoperative day (pod) 2, gastrointestinal bleeding on pod 193, HCC recurrence on pod 314, alcohol relapse and pancreatitis on pod 871, and 2 patients died for unknown reasons at home on pod 163 and 519, respectively. Out of those 6 patients, 2 patients tested PEth-positive at baseline and both patients had a relapse after LT, while their indication for LT was ALD. However, both patients complied with 6 months of abstinence (uEtG analysis) and psychiatric evaluations before LT. On note, all patients with ALD were offered the option of a psychiatric referral and participation in self-help groups after LT.
Another 10 non-transplant patients (15.2%) died during the observational period: 1 patient with decompensation after TIPS-thrombosis, 3 with HCC progression, and 6 with unknown reasons. Four of the dead patients had positive PEth at baseline and none had a documented relapse during the follow-up. Out of 25 patients with ALD at baseline, 3 patients had been abstinent for less than 6 months. One of these patients was successfully transplanted 2 years later, another patient had a follow-up of 5 years without LT, and the third patient, who was PEth-positive at baseline, died 3 months after study inclusion.
We did not find any significant association between positive test results and overall survival for PEth (
CORRELATION OF PETH VALUES:
PEth values were correlated with other biomarkers and self-reported alcohol consumption. In the correlation and regression analyses, uEtG and hEtG showed a significant positive correlation with the biomarker PEth (P<0.026 and P<0.018 respectively). However, diagnosis of alcohol-related cirrhosis, AUDIT >1 point, or TLFB results were not significantly associated with a positive test result (Table 3).
The influence of tested parameters on the height of PEth values were tested by linear regression analysis. Positive uEtG and hEtG test results significantly predicted higher PEth values. Alcohol consumption over the last week, last 2 weeks, last month and last 6 months correlated with higher PEth values. This was even intensified if alcohol intake was admitted within 2 weeks (P<0.001, R2=0.501) and 4 weeks (P=0.001, R2=0.160). The active waiting list status (T) correlated significantly with negative PEth values (Table 4).
Discussion
RATIONALE FOR MONITORING SOBRIETY IN NON-ALCOHOL-RELATED LIVER DISEASE:
Two reasons could explain why monitoring consumption in non-alcohol-related diseases is important. First, heavy alcohol use might have gone undetected in non-ALD populations, given that alcohol use disorders frequently go unnoticed in medical settings and also due to the stigma associated with the disease, which makes its disclosure difficult [38]. Second, ALD and other non-alcoholic conditions such as NASH share significant underlying necessities, mainly related to life-style changes required to avoid disease recurrence and graft loss [39,40]. Therefore, direct alcohol biomarkers could also be viewed as life-style change indicators that could help both professionals and patients to monitor and sustain those changes.
SUPERIOR PETH SENSITIVITY:
This and similar studies suggest PEth could have some advantages and/or give complementary information regarding alcohol intake over other direct alcohol biomarkers [22,23,26,28]. In the present study, PEth values were found to be positive 3.5 times more often than any of the other metabolites, indicating that PEth might be more sensitive than uEtG and hEtG in the detection of ethanol use in the study setting. Various reasons could explain this increased sensitivity. Abstinence of several days may result in negative uEtG but still be PEth-positive due to differences in half-life of the markers [14,22,23]. Consequently, announcing a urine control may result in short-term abstinence before possible urine sampling and lead to negative uEtG concentrations. Compared to hEtG, it must be noted that hair analysis does not cover the last 2 weeks before cutting the hair, since incorporation and growth needs 10–14 days. Hair treatment and other conditions might lower hEtG concentrations, while blood used for PEth detection is unlikely to be manipulated. Previous studies conducted in similar populations have also shown greater sensitivity for PEth [24,26].
PETH CORRELATES WITH ALCOHOL AMOUNT INGESTED:
Another interesting feature of PEth previously suggested in the literature is its correlation with the amount of ethanol ingested. It has actually been proposed as a marker of heavy vs non-heavy alcohol intake [41]. While an exact cut-off is difficult to find [42], our regression analysis did find a correlation between the amount of self-reported alcohol ingestion and PEth values. Results from our regression analysis also suggest PEth correlate well with other clinical variables, such as self-reports and also with other direct biomarkers.
BIOMARKER LIMITATIONS:
Among the limitations of this study are the following: 1) First of all, this was a bullet-point analysis and analyzed LT candidates in our center based on their biomarker parameters at a single time point. A longitudinal view in different centers would be useful for further analysis. In addition, centers experienced in early LT below 6 months of sobriety [6] could add value in this regard. 2) Furthermore, the self-assessment is subjective by nature and might be influenced by memory bias. 3) Our cohort does not represent participants with extremely high alcohol consumption, as these patients are not transplant candidates per se.
While the uEtG cut-off is defined as 0.5 mg/L in the guideline of the German Medical Association (artificially increased value through, for example, the consumption of sauerkraut), comparable cut-off values for PEth are still under discussion. PEth is highly specific and sensitive, but without a defined cut-off it is difficult to judge non-intended and intended alcohol consumption in a cohort of LT patients (with various diagnoses of liver disease), although a value of 0 ng/mL should be advised considering the impact of alcohol on liver function after LT. The long half-life found in the terminal elimination phase (>8 days) explains the long period until PEth values in the blood drop below about 100 ng/mL PEth 16: 0/18: 1, which can take several weeks after drinking, depending on the amount consumed before alcohol cessation [43]. In clinical use, non-intentional consumption has been examined less than intentional intake (ie, excessive alcohol consumption). So far, only cut-off values for a lower limit of the PEth 16: 0/18: 1 concentration (35 ng/mL) and a limit to differentiate between non-intended and excessive alcohol consumption have been suggested (210 ng/mL) in Swedish studies [44,45]. A further lowering of the limit value appears to be possible only after future studies with an analysis of low alcohol consumption and addressing of false-positive test results.
Conclusions
Taken together, all these findings suggest the high validity of PEth as an alcohol biomarker in the peri-transplantation period. While uEtG is the current criterion standard in transplantation guidelines in many countries, accumulating data indicate that PEth could significantly add value and objectivity to this complex process. Alcohol consumption after LT impairs graft survival. In the non-alcoholic liver disease cohort, there is a high percentage of patients with alcohol consumption. Therefore, our data underscore the importance of testing all transplant candidates.
Figures
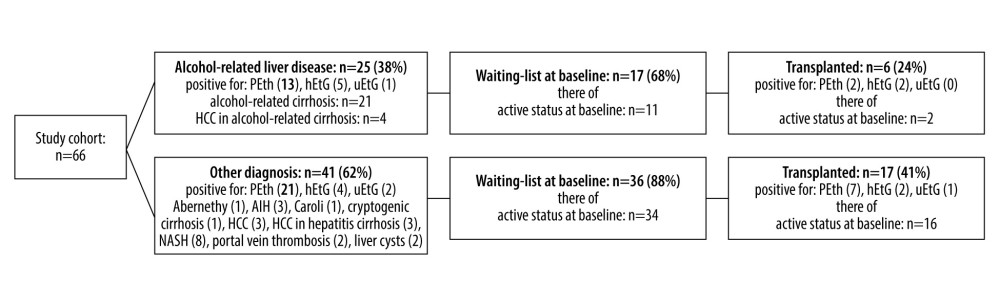 Figure 1. Schematic study group presentation divided into alcohol-related and non-alcohol-related diagnoses of liver disease. In addition to the breakdown of the diagnoses, the proportion of positive biomarkers is shown. In flow direction, listing for liver transplantation as well as the number of performed liver transplantations is displayed. Waiting list status is specified for active listing in contrast to inactive status (“not transplantable”). Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA.
Figure 1. Schematic study group presentation divided into alcohol-related and non-alcohol-related diagnoses of liver disease. In addition to the breakdown of the diagnoses, the proportion of positive biomarkers is shown. In flow direction, listing for liver transplantation as well as the number of performed liver transplantations is displayed. Waiting list status is specified for active listing in contrast to inactive status (“not transplantable”). Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA. 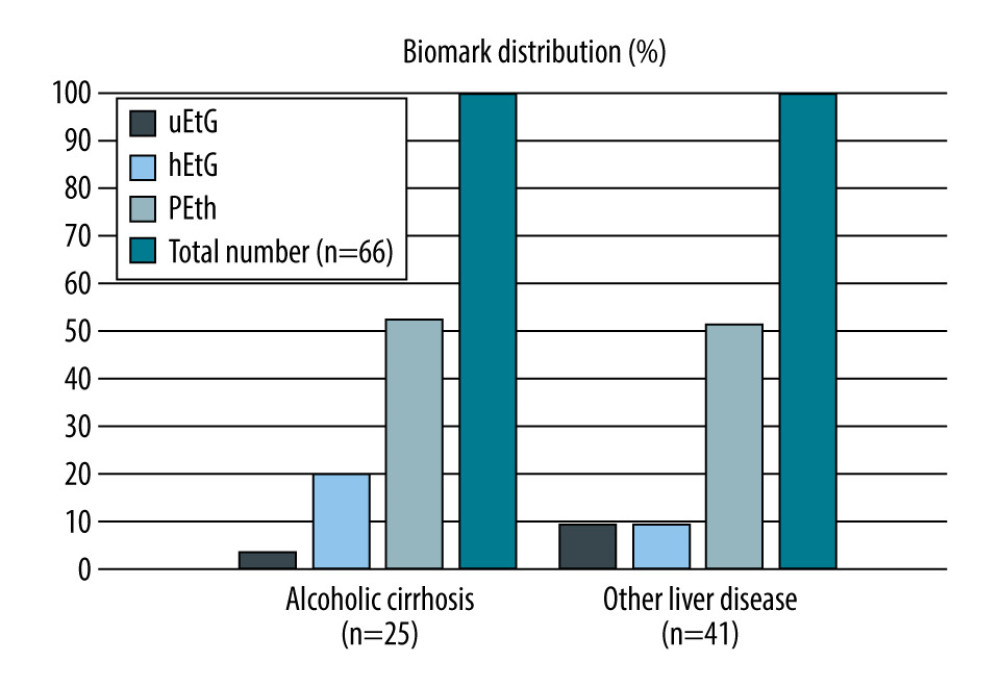 Figure 2. Overview of positive biomarker test results in patients with alcohol-related cirrhosis (n=25) or other liver diseases (n=41) in percent. uEtG and hEtG were tested in 65 patients, respectively. For PEth (n=66), patients with liver diseases other than alcohol-related cirrhosis showed a positive test result in over 50% of the cases, which is comparable to patients with alcohol-related cirrhosis. Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA.
Figure 2. Overview of positive biomarker test results in patients with alcohol-related cirrhosis (n=25) or other liver diseases (n=41) in percent. uEtG and hEtG were tested in 65 patients, respectively. For PEth (n=66), patients with liver diseases other than alcohol-related cirrhosis showed a positive test result in over 50% of the cases, which is comparable to patients with alcohol-related cirrhosis. Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA. Tables
Table 1. Patient characteristics, biomarker values, and follow-up data, showing the number of patients (n), range, mean value and standard deviation (SD) for demographic parameters, expression of liver values, and AUDIT results, as well as number of positive test results, range, mean value and SD for uEtG, hEtG, PEth (16: 0/18: 2), and realized liver transplantations (LT) within the follow-up period for the total group as well as for the biomarker groups.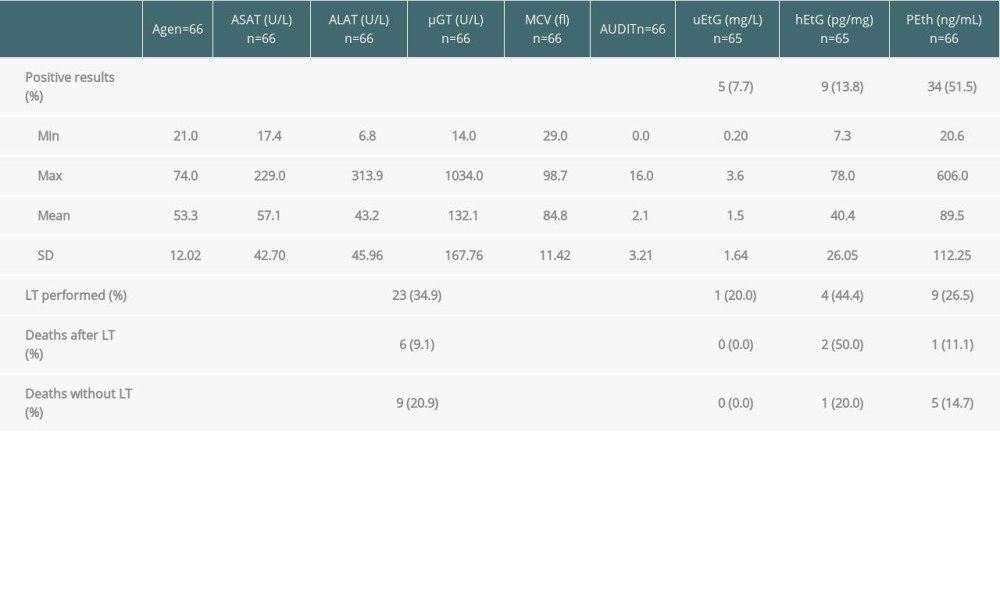 Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires.
Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires.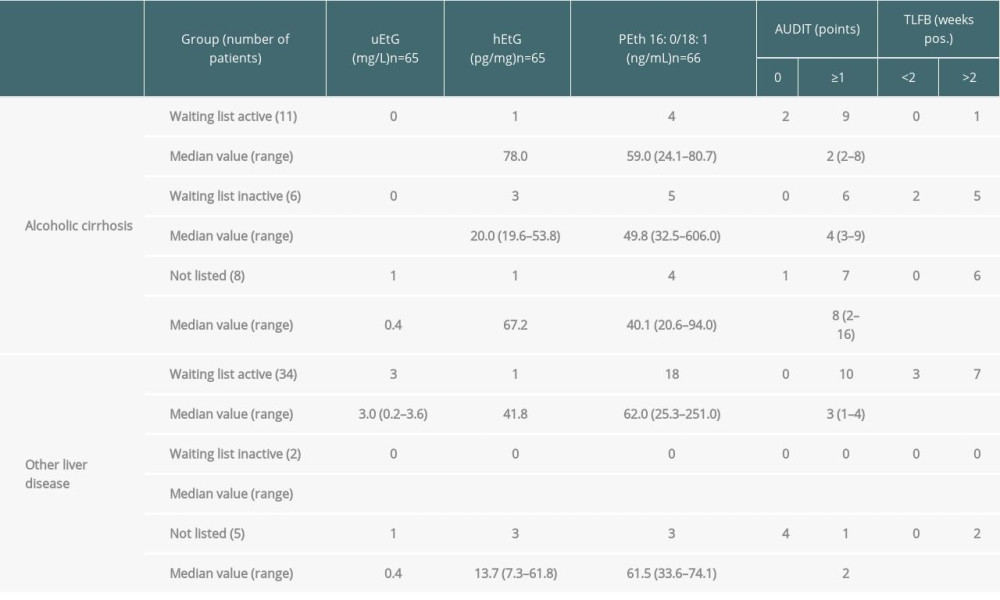 Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant.
Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant.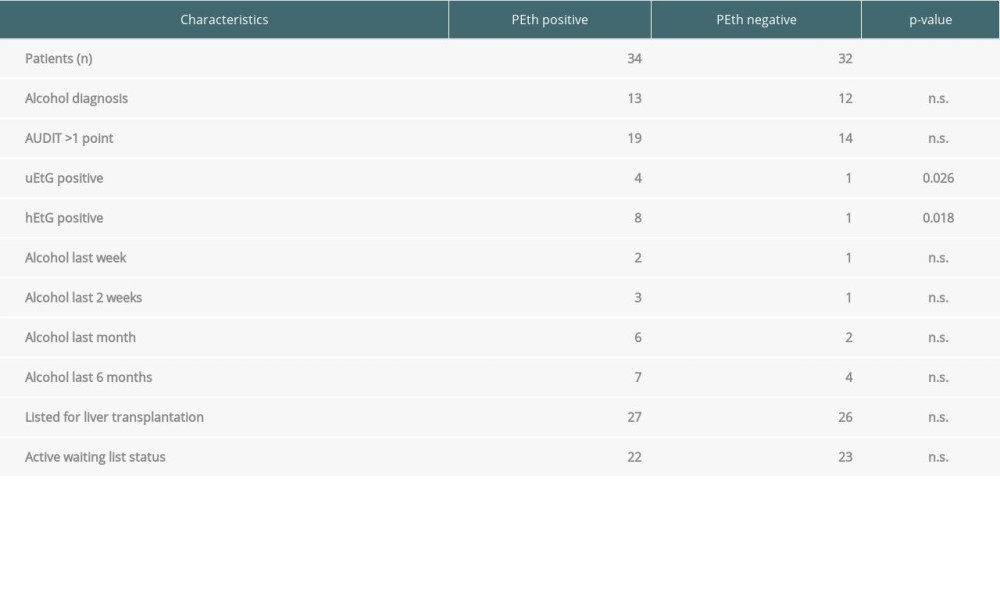 Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown.
Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown.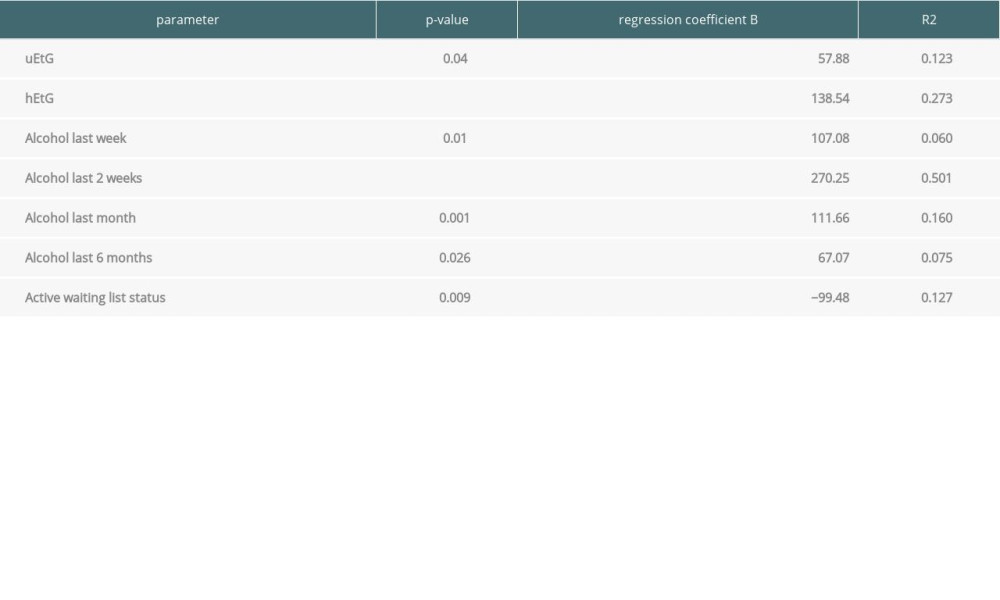
References
1. : Indications for Liver Transplantation in Germany in 2019 https://www.dso.de/DSO-Infografiken/Indikationen_LeberTX_2019.png
2. Jaurigue MM, Cappell MS, Therapy for alcoholic liver disease: World J Gastroenterol, 2014; 20(9); 2143-58
3. Chuncharunee L, Yamashiki N, Thakkinstian A, Sobhonslidsuk A, Alcohol relapse and its predictors after liver transplantation for alcoholic liver disease: A systematic review and meta-analysis: BMC Gastroenterol, 2019; 19(1); 150
4. Gallegos-Orozco JF, Charlton MR, Alcoholic liver disease and liver transplantation: Clin Liver Dis, 2016; 20(3); 521-34
5. Shawcross DL, O’Grady JG, The 6-month abstinence rule in liver transplantation: Lancet, 2010; 376(9737); 216-17
6. Braun F, Selection of alcohol-dependent candidates for liver transplantation: SUCHT, 2018(64); 157-64
7. Herrick-Reynolds KM, Punchhi G, Greenberg RS, Evaluation of early vs standard liver transplant for alcohol-associated liver disease: JAMA Surg, 2021; 156(11); 1026-34
8. Zemore SE, The effect of social desirability on reported motivation, substance use severity, and treatment attendance: J Subst Abuse Treat, 2012; 42(4); 400-12
9. Barrio P, Teixidor L, Rico N, Urine ethyl glucuronide unraveling the reality of abstinence monitoring in a routine outpatient setting: A cross-sectional comparison with ethanol, self-report and clinical judgment: Eur Addict Res, 2016; 22(5); 243-48
10. Niemela O, Biomarker-based approaches for assessing alcohol use disorders: Int J Environ Res Public Health, 2016; 13(2); 166
11. Wurst FM, Kempter C, Seidl S, Alt A, Ethyl glucuronide – a marker of alcohol consumption and a relapse marker with clinical and forensic implications: Alcohol Alcohol, 1999; 34(1); 71-77
12. Borucki K, Schreiner R, Dierkes J, Detection of recent ethanol intake with new markers: Comparison of fatty acid ethyl esters in serum and of ethyl glucuronide and the ratio of 5-hydroxytryptophol to 5-hydroxyindole acetic acid in urine: Alcohol Clin Exp Res, 2005; 29(5); 781-87
13. Halter CC, Dresen S, Auwaerter V, Kinetics in serum and urinary excretion of ethyl sulfate and ethyl glucuronide after medium dose ethanol intake: Int J Legal Med, 2008; 122(2); 123-28
14. Armer JM, Gunawardana L, Allcock RL, The performance of alcohol markers including ethyl glucuronide and ethyl sulphate to detect alcohol use in clients in a community alcohol treatment programme: Alcohol Alcohol, 2017; 52(1); 29-34
15. van de Luitgaarden IAT, Beulens JWJ, Schrieks IC, Urinary ethyl glucuronide can be used as a biomarker of habitual alcohol consumption in the general population: J Nutr, 2019; 149(12); 2199-205
16. Heinemann ML, Elsing L, Kaiser T, Challenges of LC-MS/MS ethyl glucuronide analysis in abstinence monitoring of liver transplant candidates: Clin Chem Lab Med, 2020; 58(8); 1265-70
17. Biondi A, Freni F, Carelli C, Ethyl glucuronide hair testing: A review: Forensic Sci Int, 2019; 300; 106-19
18. Sterneck M, Yegles M, Rothkirch von G, Determination of ethyl glucuronide in hair improves evaluation of long-term alcohol abstention in liver transplant candidates: Liver Int, 2014; 34(3); 469-76
19. Moeller KE, Lee KC, Kissack JC, Urine drug screening: Practical guide for clinicians: Mayo Clin Proc, 2008; 83(1); 66-76
20. Petzel-Witt S, Pogoda W, Wunder C, Influence of bleaching and coloring on ethyl glucuronide content in human hair: Drug Test Anal, 2018; 10(1); 177-83
21. Eisenbeiss L, Binz TM, Baumgartner MR, Cheating on forensic hair testing? Detection of potential biomarkers for cosmetically altered hair samples using untargeted hair metabolomics: Analyst, 2020; 145(20); 6586-99
22. Gnann H, Thierauf A, Hagenbuch F, Time dependence of elimination of different PEth homologues in alcoholics in comparison with social drinkers: Alcohol Clin Exp Res, 2014; 38(2); 322-26
23. Schröck A, Hernandez Redondo A, Martin Fabritius M, Phosphatidylethanol (PEth) in blood samples from “driving under the influence” cases as indicator for prolonged excessive alcohol consumption: Int J Legal Med, 2016; 130(2); 393-400
24. Andresen-Streichert H, Beres Y, Weinmann W, Improved detection of alcohol consumption using the novel marker phosphatidylethanol in the transplant setting: Results of a prospective study: Transpl Int, 2017; 30(6); 611-20
25. Fleming MF, Smith MJ, Oslakovic E, Phosphatidylethanol detects moderate-to-heavy alcohol use in liver transplant recipients: Alcohol Clin Exp Res, 2017; 41(4); 857-62
26. Barrio P, Gual A, Lligona A, Phosphatidylethanol for monitoring alcohol use in liver transplant candidates: An observational study: J Clin Med, 2020; 9(9); 3060
27. Kiefer F, Hoffmann SGuideline Group AUD, German guidelines on screening, diagnosis and treatment of alcohol use disorders – update 2021: Eur Addict Res, 2022 [Online ahead of print]
28. Schröck A, Wurst FM, Thon N, Weinmann W, Assessing phosphatidylethanol (PEth) levels reflecting different drinking habits in comparison to the alcohol use disorders identification test – C (AUDIT-C): Drug Alcohol Depend, 2017; 178; 80-86
29. Burroughs AK, Sabin CA, Rolles K, 3-month and 12-month mortality after first liver transplant in adults in Europe: Predictive models for outcome: Lancet, 2006; 367(9506); 225-32
30. Charlton MR, Burns JM, Pedersen RA, Frequency and outcomes of liver transplantation for nonalcoholic steatohepatitis in the United States: Gastroenterology, 2011; 141(4); 1249-53
31. Haldar D, Kern B, Hodson J, Outcomes of liver transplantation for non-alcoholic steatohepatitis: A European Liver Transplant Registry study: J Hepatol, 2019; 71(2); 313-22
32. Sobell MB, Sobell LC, Klajner F, The reliability of a timeline method for assessing normal drinker college students’ recent drinking history: Utility for alcohol research: Addict Behav, 1986; 11(2); 149-61
33. Conigrave KM, Saunders JB, Whitfield JB, Diagnostic tests for alcohol consumption: Alcohol Alcohol, 1995; 30(1); 13-26
34. , German S3 guideline: Screening, diagnosis and treatment of alcohol use disorders: AWMF-Register Nr 076-001, 2021
35. Kerekes I, Yegles M, Coloring, bleaching, and perming: Influence on EtG content in hair: Ther Drug Monit, 2013; 35(4); 527-29
36. Cappelle D, Neels H, Yegles M, Gas chromatographic determination of ethyl glucuronide in hair: Comparison between tandem mass spectrometry and single quadrupole mass spectrometry: Forensic Sci Int, 2015; 249; 20-24
37. Hernández Redondo A, Schröck A, Kneubuehl B, Weinmann W, Determination of ethyl glucuronide and ethyl sulfate from dried blood spots: Int J Legal Med, 2013; 127(4); 769-75
38. Volkow ND, Stigma and the toll of addiction: N Engl J Med, 2020; 382(14); 1289-90
39. Nobili V, Carter-Kent C, Feldstein AE, The role of lifestyle changes in the management of chronic liver disease: BMC Med, 2011; 9; 70
40. Burra P, Becchetti C, Germani G, NAFLD and liver transplantation: Disease burden, current management and future challenges: JHEP Rep, 2020; 2(6); 100192
41. Viel G, Boscolo-Berto R, Cecchetto G, Phosphatidylethanol in blood as a marker of chronic alcohol use: A systematic review and meta-analysis: Int J Mol Sci, 2012; 13(11); 14788-812
42. Helander A, Hermansson U, Beck O, Dose-response characteristics of the alcohol biomarker phosphatidylethanol (PEth) – a study of outpatients in treatment for reduced drinking: Alcohol Alcohol, 2019; 54(6); 567-73
43. Luginbühl M, Weinmann W, Butzke I, Pfeifer P, Monitoring of direct alcohol markers in alcohol use disorder patients during withdrawal treatment and successive rehabilitation: Drug Test Anal, 2019; 11(6); 859-69
44. Helander A, Hansson TNational harmonization of the alcohol biomarker PEth: Lakartidningen, 2013; 110(39–40); 1747-48 [in German]
45. Isaksson A, Walther L, Hansson T, High-throughput LC-MS/MS method for determination of the alcohol use biomarker phosphatidylethanol in clinical samples by use of a simple automated extraction procedure-preanalytical and analytical conditions: J Appl Lab Med, 2018; 2(6); 880-92
Figures
 Figure 1. Schematic study group presentation divided into alcohol-related and non-alcohol-related diagnoses of liver disease. In addition to the breakdown of the diagnoses, the proportion of positive biomarkers is shown. In flow direction, listing for liver transplantation as well as the number of performed liver transplantations is displayed. Waiting list status is specified for active listing in contrast to inactive status (“not transplantable”). Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA.
Figure 1. Schematic study group presentation divided into alcohol-related and non-alcohol-related diagnoses of liver disease. In addition to the breakdown of the diagnoses, the proportion of positive biomarkers is shown. In flow direction, listing for liver transplantation as well as the number of performed liver transplantations is displayed. Waiting list status is specified for active listing in contrast to inactive status (“not transplantable”). Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA. Figure 2. Overview of positive biomarker test results in patients with alcohol-related cirrhosis (n=25) or other liver diseases (n=41) in percent. uEtG and hEtG were tested in 65 patients, respectively. For PEth (n=66), patients with liver diseases other than alcohol-related cirrhosis showed a positive test result in over 50% of the cases, which is comparable to patients with alcohol-related cirrhosis. Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA.
Figure 2. Overview of positive biomarker test results in patients with alcohol-related cirrhosis (n=25) or other liver diseases (n=41) in percent. uEtG and hEtG were tested in 65 patients, respectively. For PEth (n=66), patients with liver diseases other than alcohol-related cirrhosis showed a positive test result in over 50% of the cases, which is comparable to patients with alcohol-related cirrhosis. Figure prepared with PowerPoint 2019, Microsoft Corporation, Redmond/Washington, USA. Tables
 Table 1. Patient characteristics, biomarker values, and follow-up data, showing the number of patients (n), range, mean value and standard deviation (SD) for demographic parameters, expression of liver values, and AUDIT results, as well as number of positive test results, range, mean value and SD for uEtG, hEtG, PEth (16: 0/18: 2), and realized liver transplantations (LT) within the follow-up period for the total group as well as for the biomarker groups.
Table 1. Patient characteristics, biomarker values, and follow-up data, showing the number of patients (n), range, mean value and standard deviation (SD) for demographic parameters, expression of liver values, and AUDIT results, as well as number of positive test results, range, mean value and SD for uEtG, hEtG, PEth (16: 0/18: 2), and realized liver transplantations (LT) within the follow-up period for the total group as well as for the biomarker groups. Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires.
Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires. Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant.
Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant. Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown.
Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown. Table 1. Patient characteristics, biomarker values, and follow-up data, showing the number of patients (n), range, mean value and standard deviation (SD) for demographic parameters, expression of liver values, and AUDIT results, as well as number of positive test results, range, mean value and SD for uEtG, hEtG, PEth (16: 0/18: 2), and realized liver transplantations (LT) within the follow-up period for the total group as well as for the biomarker groups.
Table 1. Patient characteristics, biomarker values, and follow-up data, showing the number of patients (n), range, mean value and standard deviation (SD) for demographic parameters, expression of liver values, and AUDIT results, as well as number of positive test results, range, mean value and SD for uEtG, hEtG, PEth (16: 0/18: 2), and realized liver transplantations (LT) within the follow-up period for the total group as well as for the biomarker groups. Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires.
Table 2. Results of positive biomarkers uEtG, hEtG, and PEth and patient self-assessment with the AUDIT and TLFB questionnaires. Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant.
Table 3. Frequency table of patient characteristics with positive or negative test results for PEth. Displayed are numbers of positive findings for alcohol diagnosis, >1 point in the AUDIT, self-reported alcohol consumption, and waiting list status (T and NT). Significance was determined by results of binary logistic regression analyses, P<0.05 was regarded as statistically significant. Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown.
Table 4. Influences on the height of PEth values. The results of simple linear regression analysis regarding the height of PEth values are displayed. P values <0.05 are regarded as statistically significant. Unstandardized regression coefficient B to determine effect direction and R2 as measure of goodness of fit is shown. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








