21 April 2023: Original Paper
Converting from Tenofovir Disoproxil Fumarate to Tenofovir Alafenamide in Patients with Hepatitis B Following Liver Transplantation
Chih-Hsien Cheng1ABCDE*, Hao-Chien Hung1BC, Jin-Chiao Lee1BC, Yu-Chao Wang1CD, Tsung-Han Wu1CD, Chen-Fang Lee1ABC, Ting-Jung Wu1BC, Hong-Shiue Chou1BC, Kun-Ming ChanDOI: 10.12659/AOT.938731
Ann Transplant 2023; 28:e938731
Abstract
BACKGROUND: Taiwan has a high prevalence of hepatitis B virus (HBV) infection. HBV-related end-stage liver disease is the leading cause of liver transplantation (LT). Tenofovir alafenamide (TAF) is a recently approved agent for the treatment of chronic HBV infection that improves renal profiles compared with tenofovir disoproxil fumarate (TDF) in phase III trials. This study aimed to assess the outcomes of TAF treatment in LT recipients.
MATERIAL AND METHODS: This retrospective study analyzed 17 LT recipients who underwent treatment with TDF and TAF. Changes in baseline renal function were compared.
RESULTS: Seventeen LT recipients received TDF for ≥48 weeks and were switched to TAF. During TDF treatment, estimated glomerular filtration rate (eGFR) (using the Modification of Diet in Renal Disease [MDRD] formula) decreased significantly at weeks 24 and 48. At week 48, only 2 patients (11.8%) displayed improved renal function, whereas the other patients showed decreased eGFR ranging from 5.48% to 62.84%. After switching to TAF, the median eGFR increased by 3.01% at week 24 and decreased by 0.31% at week 48. Seven patients (47%) showed improved renal function at week 48 after TAF treatment.
CONCLUSIONS: Switching from TDF to TAF was associated with fewer short-term renal impairment while maintaining the antiviral efficacy in LT recipients.
Keywords: Hepatitis B, Liver Transplantation, Humans, Tenofovir, Retrospective Studies, Alanine, Adenine, Antiviral Agents, Hepatitis B, Chronic, Hepatitis B virus
Background
Taiwan is an endemic country for hepatitis B virus (HBV) infection, and despite vaccination, screening, and treatment programs, HBV-related end-stage liver diseases are still the leading reason for liver transplantation (LT). After transplantation, the prevention of HBV recurrence is critical, and most prophylactic regimens consist of anti-hepatitis B immunoglobulin (HBIg) and anti-HBV nucleoside/nucleotide analogs.
Tenofovir alafenamide (TAF) has recently been approved for the treatment of chronic HBV. TAF is a phosphonate prodrug of tenofovir disoproxil fumarate (TDF) that was developed to improve TDF antiviral activity and safety profiles. Compared with TDF, TAF has greater plasma stability, which enables the intracellular delivery of lower doses of the active metabolite tenofovir diphosphate. Clinical trials in patients with chronic HBV demonstrated good efficacy and improved renal safety profiles [1,2]. However, only a few real-world studies have reported the effects of TAF in transplant patients. In this study, we analyzed the effects of changing the treatment in LT recipients from TDF to TAF and compared the changes in renal function over time.
Material and Methods
STATISTICAL ANALYSIS:
Continuous data are expressed as the median and interquartile range. Significant differences were determined using the Mann-Whitney U test, one-way ANOVA, or Wilcoxon paired tests using GraphPad Prism software (GraphPad Software, San Diego, CA, USA). A probability value of
Results
PATIENTS:
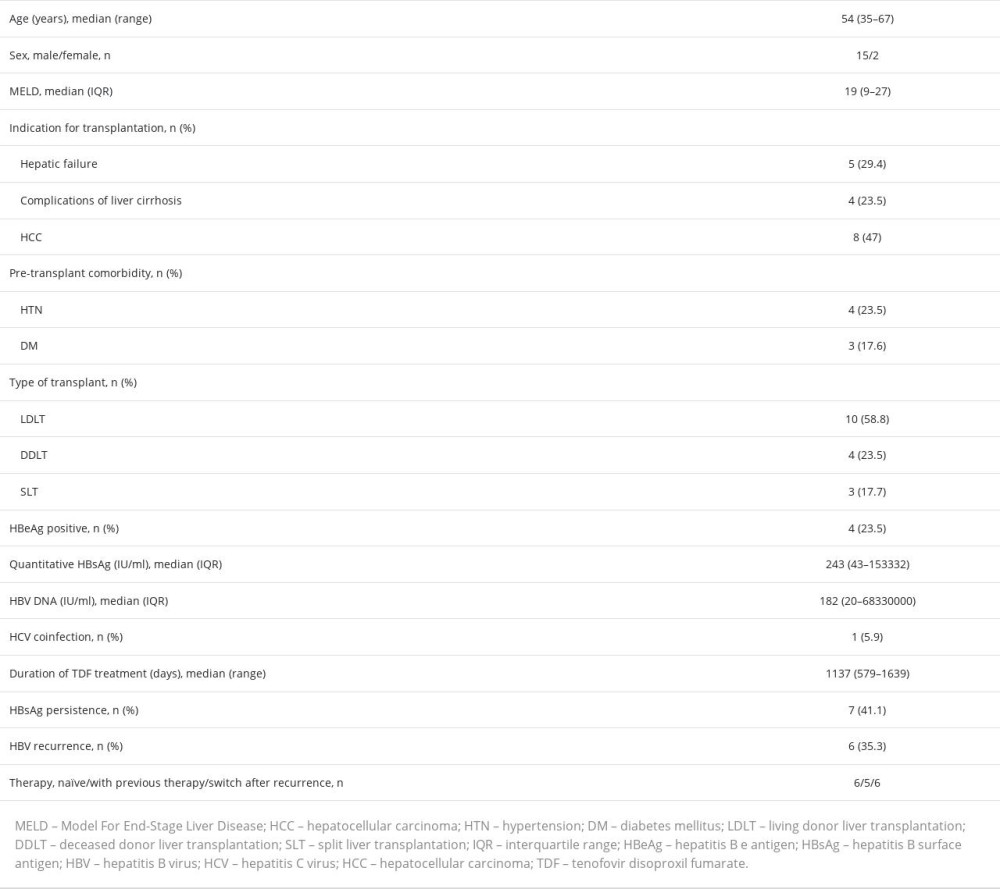
Seventeen patients were included in this study (Table 1). Ten patients received living donor LTs, 4 received deceased donor LTs, and 3 received split LTs. At the time of transplantation, the median Model of End-Stage Liver Disease score was 19 (interquartile range [IQR] 9–27). The median duration of TDF treatment was 1137 (range 579–1639) days.
Tacrolimus and mycophenolate mofetil were administered as maintenance immunosuppressive agents. Three patients also received everolimus. Blood levels of tacrolimus did not change significantly before and after switching from TDF to TAF (median level: 4.75 and 4.70 ng/mL, respectively;
RENAL FUNCTION CHANGES DURING THERAPY WITH TDF AND TENOFOVIR ALAFENAMIDE:
During TDF therapy, the renal function significantly decreased at weeks 12 and 24 (Figure 1). The median creatinine levels increased from 0.8 mg/dL at baseline to 1.14 mg/dL and 1.10 mg/dL at 12 and 24 weeks, respectively (p<0.001 and p<0.0001). The median eGFR decreased from 101.28 mL/min/1.73 m2 to 73.04 mL/min/1.73 m2 and 71.63 mL/min/1.73 m2 at 12 and 24 weeks, respectively (p<0.001 and p<0.0001). Compared with the baseline levels, the median eGFR decreased by 13.43% at week 24 and by 17.66% at week 48. After 24 weeks, renal function stabilized with no further significant changes. At week 48, only 2 patients (11.8%) displayed improved renal function, whereas the other patients showed decreased eGFR, ranging from 5.48% to 62.84%.
After switching to TAF, the median creatinine improved at week 12 and went from 1.18 mg/dL to 1.09 mg/dL (p<0.05; Figure 2). The median eGFR increased from 67.62 mL/min/1.73 m2 at baseline to 72.77 mL/min/1.73 m2 at week 12 (not significant) and decreased to 69.22 mL/min/1.73 m2 and 67.11 mL/min/1.73 m2 at week 24 and 48, respectively (p<0.05). The median eGFR increased by 3.01% at week 24 and decreased by 0.31% at week 48. After the initial 12 weeks, a small improvement in renal function was noted, which subsequently decreased over time. At week 48, renal function showed a further significant decrease, but the overall level remained similar to the baseline levels (Figure 3). At week 48, 7 patients (47%) showed improved renal function. In the remaining patients, eGFR decreased from 0.31% to 15.67%.
STRATIFICATION BY STAGES OF CKD:
Before TDF therapy, 10 patients (58.8%) were in stage 1, 6 (35.3%) were in stage 2, and 1 (5.9%) was in stage 3a. Only patients with stage 1 disease exhibited a significant decline in renal function. At week 48, the median percent change in eGFR was −23.8%, −10.8%, and −8.25% for stage 1, 2, and 3a patients, respectively. Upon initiating therapy with TAF, the majority of patients were in stage 2 (10 patients, 58.8%). Four patients (23.5%) were in stage 1, and 1 patient (5.9%) each had stages 3a, 3b, and 4 disease. At week 48, the median eGFR change was −12.62% and +17% for stage 1 and 2 patients, respectively (Figure 4A).
Compared with TDF, TAF had fewer detrimental effects on renal function in patients with worse CKD stages (Figure 4B). Interestingly, 2 patients with stage 3b and 4 disease showed substantial improvement in renal function even after TAF therapy. The stage 3a patient showed +7.96% in eGFR; the stage 3b patient, +26.86%; and the stage 4 patient, +23.95% (Figure 4C).
ANTIVIRAL EFFICACY:
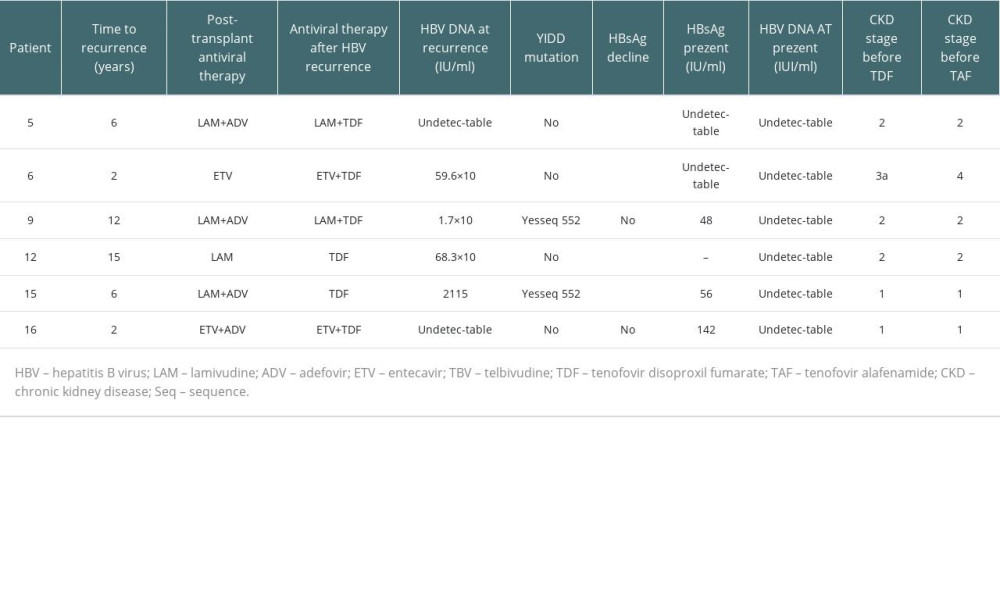
Six patients started TDF as a naïve treatment, 5 had a treatment history with other antivirals, and 6 were administered TDF due to HBV reactivation. Among the patients with HBV reactivation, 3 showed a decrease in HBsAg levels >1 log at 48 weeks with TDF, 1 showed a decrease in HBsAg levels >1 log at 72 weeks, and at level remained the same in 2 (both patients had a YIDD motif mutation, and the other remained negative for HBV DNA; Table 2). The antiviral efficacy remained unchanged after switching to TAF.
Discussion
TDF is a widely used first-line HBV treatment; however, experience with HIV patients has indicated a potential risk of proximal tubular dysfunction, proteinuria, and Fanconi syndrome [6]. TAF has greater plasma stability than TDF, enables more deficient delivery to hepatocytes, and allows treatment at lower doses [7]. Moreover, switching from TDF to TAF improved eGFR and proteinuria in patients with impaired renal function [8]. Although renal dysfunction is less common in patients with chronic hepatitis B [9], it remains a frequent complication in LT patients [10]. TAF has been available in Taiwan since April 2017 and was added to the Taiwanese National Health Insurance Program reimbursement list in June 2019. Data on the use of TAF in LT recipients are scarce. Therefore, we assessed the impact of TAF compared with TDF on renal parameters in this specific high-risk population.
After treatment with TDF, patients showed an early decline in renal function at weeks 12 and 24. The median eGFR declined by 13.4% at week 24. From weeks 24 to 48, no further significant changes were noted in creatinine or eGFR, but the median eGFR declined by 17.6% compared with baseline at the end of week 48. After switching to TAF, the main changes were observed during the first 12 and 24 weeks. However, during this time, renal function improved. At week 24, the median eGFR increased by 3%. After 48 weeks of treatment, a slow decline was noted in renal function, which remained similar to the baseline. A recent study by Saab et al showed that LT recipients treated with TAF did not display any significant changes in renal function over time, whereas those treated with other antiviral agents showed a significant increase in serum creatinine [11]. Sinakos et al compared LT recipients who switched to TAF due to low GFR (<60 mL/min/1.73 m2) with those who remained on TDF; they observed that switching from TDF to TAF in patients who had already developed CKD led to a stabilization of renal function, although without a robust improvement [12]. In another study, Sripongpun et al reported that switching from other antivirals to TAF resulted in 73% improvement in renal function in LT patients [13]. In our study, the initial improvement in eGFR was not maintained until week 48; however, the adverse impact was less significant than that of TDF. These results are in accordance with those of studies in patients with chronic hepatitis B or HIV, in which long-term treatment with TAF for 96 or 120 weeks stabilized or improved renal function [14,15].
When we stratified the patients according to the CKD stage, the most significant decline in eGFR was observed in stage 1 patients, both with TDF or TAF. However, these patients did not progress to stage 2 after 48 weeks. Notably, patients with poor renal function appeared to benefit the most from TAF. Three patients in stages 3 and 4 showed a marked improvement of >20% in renal function. Preliminary data from a phase II study comparing TDF with TAF in LT recipients with CKD stage 2 or greater showed that switching to TAF resulted in a trend toward improved renal function (median change: −0.07 for TAF vs −0.02 mg/dL for TDF;
The antiviral effects of TDF and TAF are comparable. Neither treatment-naïve nor treatment-experienced patients experienced HBV recurrence. Thus, antiviral effects were maintained when therapy was switched from TDF to TAF. Among patients with HBV recurrence, 4 of 6 patients experienced an HBsAg >1 log reduction with TDF but did not show any further decline with TAF. Treatment of patients with HBV recurrence after LT can be challenging. Salvage therapy may include other nucleo(t)-side inhibitors, either as monotherapy or in combination [17,18]. So far, resistance to TDF and TAF appears uncommon, and in vitro studies have shown the potent antiviral activity of TAF against adefovir-, lamivudine-, and entecavir-resistant HBV isolates [19]. Therefore, TDF and TAF with better safety profiles are reasonable therapeutic options for this group of patients.
Despite the widespread use of TAF in patients with chronic hepatitis, only a few studies have analyzed its administration in LT patients. Spripongpun et al administered TAF to 11 LT recipients for at least 48 weeks and observed no changes in the serum levels of immunosuppressants. A significant reduction was noted in ALT levels, and all patients had undetectable serum HBV DNA throughout the study [13]. Saab et al reported no HBsAg seroconversion in 30 LT recipients receiving TAF monotherapy. Rashidi-Alavijeh et al also observed no seroconversion in 22 LT patients, with only 1 HBV DNA-positive patient after 1 year of TAF therapy. The effectiveness of TAF is being evaluated in prospective randomized trials [20,21], which may provide further evidence to guide clinical practice in transplant patients, particularly in those who require higher immunosuppressive drug levels.
Our study has several limitations. As this was a retrospective study, we did not adjust for comorbidities that can affect renal function, independent of the type of antiviral treatment. Additionally, we did not include a control group of patients who continued their treatment with TDF. Although the serum phosphorus levels did not change significantly during the 48 weeks of observation (data not shown), the changes in bone mineral density were not measured, and the impact of TAF on bone mineralization in LT recipients was not evaluated. Additionally, renal function was assessed using the MDRD formula, although the same results were obtained using the CKD-EPI equation (Supplementary Figure 1) [22]. Additional studies using radionuclide techniques (51Cr-EDTA and 99mTc-DPTA) would improve the validity of our findings. Furthermore, the small sample size precludes conclusions regarding the effect on patients with advanced CKD stages or antiviral efficacy against drug-resistant HBV strains. Finally, the short study period was insufficient to evaluate long-term safety and efficacy. Unlike nontransplant patients, the effect of treatment may be hampered by long-term immunosuppressive therapy.
Conclusions
Switching from TDF to TAF was associated with fewer short-term renal impairments while maintaining the antiviral efficacy in LT recipients.
Figures
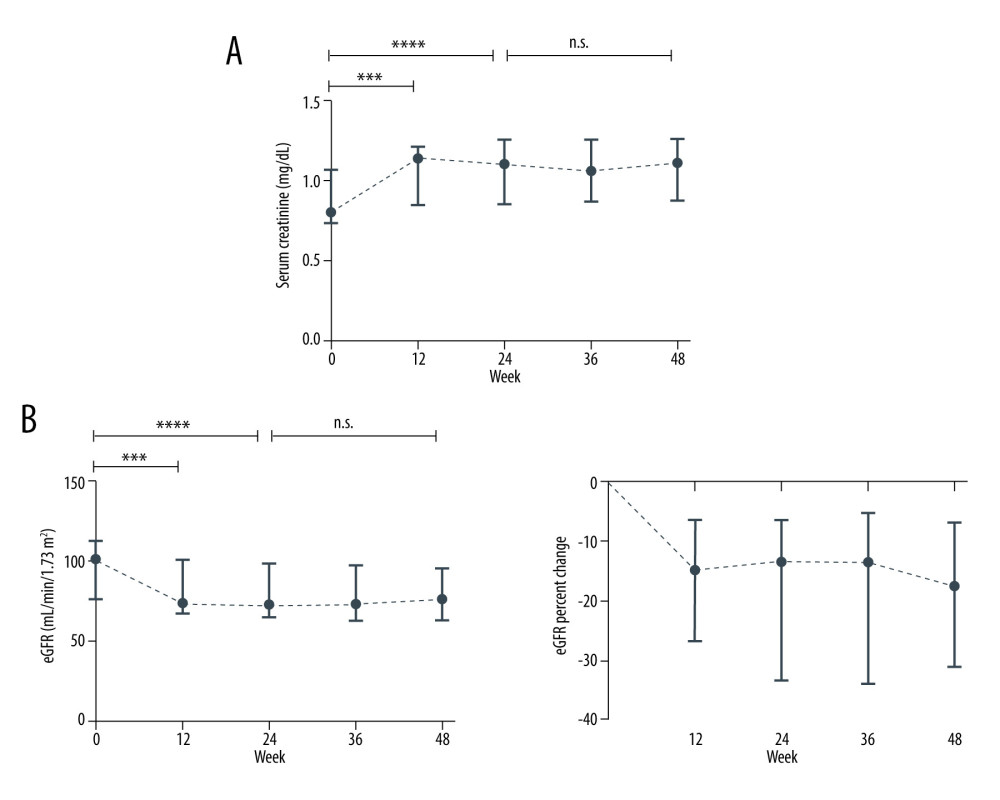 Figure 1. Renal function change during therapy with TDFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 1. Renal function change during therapy with TDFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). 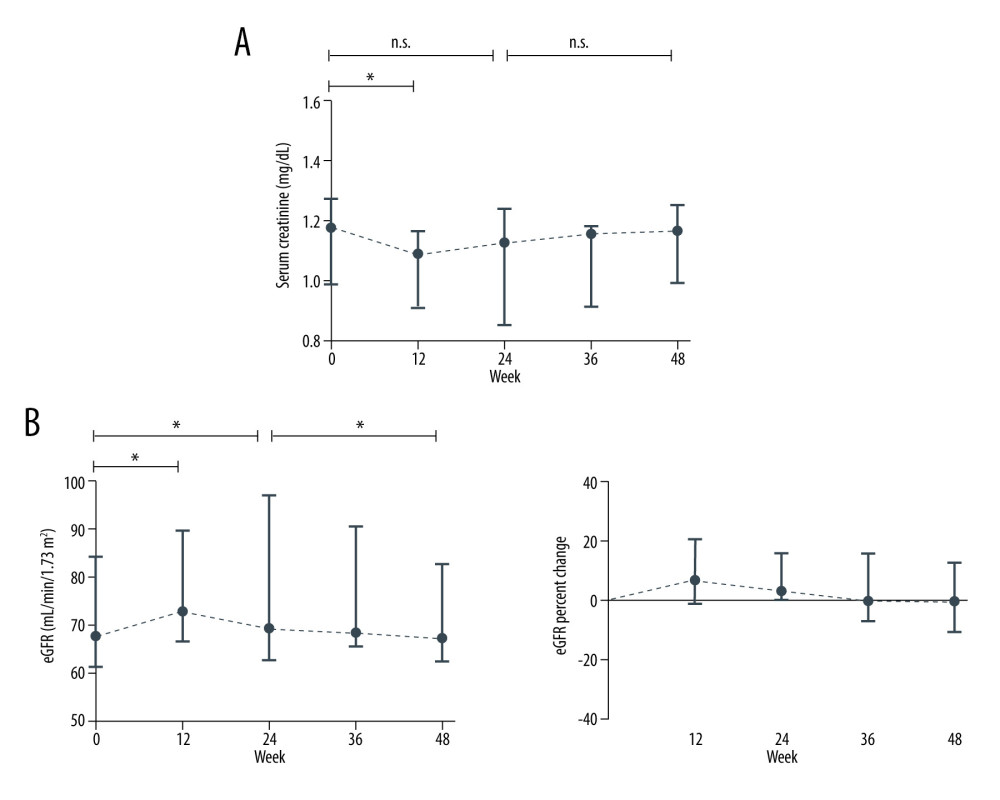 Figure 2. Renal function change after switching over to TAFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 2. Renal function change after switching over to TAFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). 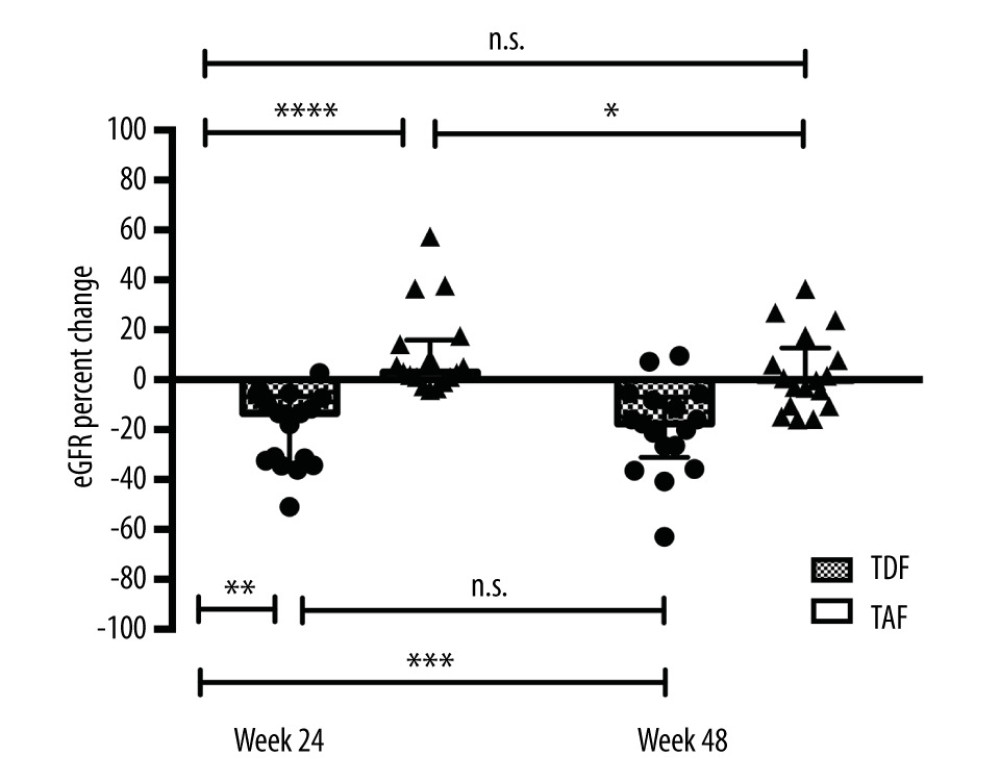 Figure 3. Renal function change during therapy with TDF and TAFMedian (IQR) eGFR percentage change from baseline at weeks 24 and 48 during TDF and TAF treatment. n.s. not significant, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 3. Renal function change during therapy with TDF and TAFMedian (IQR) eGFR percentage change from baseline at weeks 24 and 48 during TDF and TAF treatment. n.s. not significant, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). 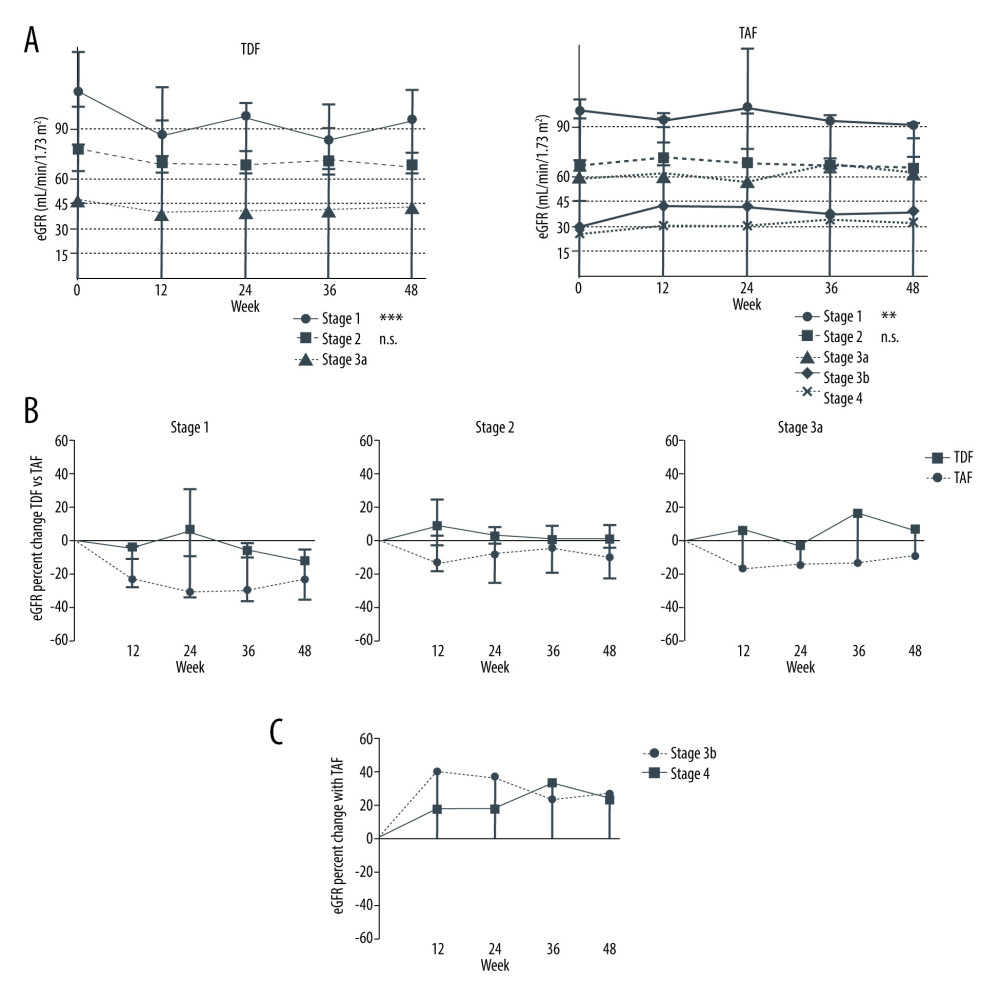 Figure 4. Renal function change during therapy with TDF and TAF after stratifying by stages of chronic kidney disease(A) Median and IQR eGFR and (B) eGFR percent change from baseline at weeks 12, 24, 36 and 48 during TDF and TAF therapy. (C) eGFR percent change of stage 3b and 4 patients during TAF. n.s. – not significant, *** p<0.001 (one-way ANOVA).
Figure 4. Renal function change during therapy with TDF and TAF after stratifying by stages of chronic kidney disease(A) Median and IQR eGFR and (B) eGFR percent change from baseline at weeks 12, 24, 36 and 48 during TDF and TAF therapy. (C) eGFR percent change of stage 3b and 4 patients during TAF. n.s. – not significant, *** p<0.001 (one-way ANOVA). References
1. Agarwal K, Brunetto M, Seto WK, 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection: J Hepatol, 2018; 68; 672-81
2. Chan HL, Fung S, Seto WK, Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of HBeAg-positive chronic hepatitis B virus infection: A randomised, double-blind, phase 3, non-inferiority trial: Lancet Gastroenterol Hepatol, 2016; 1; 185-95
3. Lee WC, Chou HS, Wu TH, Low-dose anti-hepatitis B immunoglobulin regimen as prophylaxis for hepatitis B recurrence after liver transplantation: Transpl Infect Dis, 2019; 21; e13190
4. Levey AS, Coresh J, Greene T, Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate: Ann Intern Med, 2006; 145; 247-54
5. Levey AS, Coresh J, Balk E, National Kidney Foundation practice guidelines for chronic kidney disease: Evaluation, classification, and stratification: Ann Intern Med, 2003; 139; 137-47
6. Hall AM, Hendry BM, Nitsch D, Connolly JO, Tenofovir-associated kidney toxicity in HIV-infected patients: A review of the evidence: Am J Kidney Dis, 2011; 57; 773-80
7. Babusis D, Phan TK, Lee WA, Mechanism for effective lymphoid cell and tissue loading following oral administration of nucleotide prodrug GS-7340: Mol Pharm, 2013; 10; 459-66
8. Surial B, Ledergerber B, Calmy A, Changes in renal function after switching from TDF to TAF in HIV-infected individuals: A prospective cohort study: J Infect Dis, 2020; 222; 637-45
9. Heathcote EJ, Marcellin P, Buti M, Three-year efficacy and safety of tenofovir disoproxil fumarate treatment for chronic hepatitis B: Gastroenterology, 2011; 140; 132-43
10. Weber ML, Ibrahim HN, Lake JR, Renal dysfunction in liver transplant recipients: Evaluation of the critical issues: Liver Transpl, 2012; 18; 1290-301
11. Saab S, Song D, Challita YP, Long-term outcomes with oral therapy in liver transplant recipients with hepatitis B: Clin Transplant, 2019; 33; e13740
12. Sinakos E, Panas P, Fragkou N, Tenofovir alafenamide prophylaxis post-liver transplantation: A real-world study in patients with chronic kidney disease: Acta Gastroenterol Belg, 2022; 85; 331-37
13. Sripongpun P, Mannalithara A, Kwo PY, Kim WR, Potential benefits of switching liver transplant recipients to tenofovir alafenamide prophylaxis: Clin Gastroenterol Hepatol, 2020; 18; 747-49
14. Chan HL, Fung S, Seto WK, Improved bone and renal safety of switching from tenofovir disoproxil fumarate to tenofovir alafenamide: Preliminary results from 2 phase 3 studies in HBeAg-positive and HBeAg-negative patients with chronic hepatitis B (abstract no. PS-041 plus oral presentation): J Hepatol, 2017; 66(Suppl 1); S25
15. Gupta SK, Post FA, Arribas JR, Renal safety of tenofovir alafenamide vs. tenofovir disoproxil fumarate: A pooled analysis of 26 clinical trials: Aids, 2019; 33; 1455-65
16. Gane E, George B, Munn S, Evaluation of renal and bone safety in post-liver transplant patients with chronic kidney disease receiving tenofovir alafenamide for HBV prophylaxis (abstract no. PS-336): J Hepatol, 2018; 68(Suppl 1); S514
17. Neff GW, O’Brien CB, Nery J, Outcomes in liver transplant recipients with hepatitis B virus: Resistance and recurrence patterns from a large transplant center over the last decade: Liver Transpl, 2004; 10; 1372-78
18. Akyildiz M, Karasu Z, Zeytunlu M, Adefovir dipivoxil therapy in liver transplant recipients for recurrence of hepatitis B virus infection despite lamivudine plus hepatitis B immunoglobulin prophylaxis: J Gastroenterol Hepatol, 2007; 22; 2130-34
19. Liu Y, Miller MD, Kitrinos KM, Tenofovir alafenamide demonstrates broad cross-genotype activity against wild-type HBV clinical isolates and maintains susceptibility to drug-resistant HBV isolates in vitro: Antiviral Res, 2017; 139; 25-31
20. Hospital TVG: Tenofovir alafenamide switching therapy in kidney or liver transplant recipients with chronic HBV infection, 2021 https://ClinicalTrials.gov/show/NCT05410496
21. University F: Tenofovir alafenamide for HBV prophylaxis in post orthotopic liver transplant, 2021 https://ClinicalTrials.gov/show/NCT05063071
22. Levey AS, Stevens LA, Schmid CH, A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12
Figures
 Figure 1. Renal function change during therapy with TDFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 1. Renal function change during therapy with TDFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). Figure 2. Renal function change after switching over to TAFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 2. Renal function change after switching over to TAFMedian (IQR) of (A) creatinine (mg/dL) and (B) eGFR (mL/min/1.73 m2 by the MDRD formula) levels at baseline, at weeks 12, 24, 36, and 48. n.s. not significant, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). Figure 3. Renal function change during therapy with TDF and TAFMedian (IQR) eGFR percentage change from baseline at weeks 24 and 48 during TDF and TAF treatment. n.s. not significant, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001 (Wilcoxon paired test).
Figure 3. Renal function change during therapy with TDF and TAFMedian (IQR) eGFR percentage change from baseline at weeks 24 and 48 during TDF and TAF treatment. n.s. not significant, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001 (Wilcoxon paired test). Figure 4. Renal function change during therapy with TDF and TAF after stratifying by stages of chronic kidney disease(A) Median and IQR eGFR and (B) eGFR percent change from baseline at weeks 12, 24, 36 and 48 during TDF and TAF therapy. (C) eGFR percent change of stage 3b and 4 patients during TAF. n.s. – not significant, *** p<0.001 (one-way ANOVA).
Figure 4. Renal function change during therapy with TDF and TAF after stratifying by stages of chronic kidney disease(A) Median and IQR eGFR and (B) eGFR percent change from baseline at weeks 12, 24, 36 and 48 during TDF and TAF therapy. (C) eGFR percent change of stage 3b and 4 patients during TAF. n.s. – not significant, *** p<0.001 (one-way ANOVA). In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588