30 May 2023: Case Report
Outcomes from a Single Transplant Center of 5 Pediatric Cases of Domino Liver Transplantation from Live Donors with Maple Syrup Urine Disease
Jin-Ping Zhang12ABCDEFG, Zhi-Jun Zhu12AF*, Li-Ying Sun123EFG, Lin WeiDOI: 10.12659/AOT.939893
Ann Transplant 2023; 28:e939893
Abstract
BACKGROUND: Maple syrup urine disease (MSUD) is a rare genetic deficiency of the branched-chain α-keto acid dehydrogenase (BCKAD) complex that breaks down amino acids, resulting in multi-organ failure. This report is of 5 pediatric cases of domino liver transplantation (DLT) from live donors with MSUD from a single transplant center in Beijing.
CASE REPORT: All MSUD donors were confirmed to have disease-causing mutations in BCKDHA (branched-chain keto acid dehydrogenase E1, α polypeptide) or BCKDHB (branched-chain keto acid dehydrogenase E1, β polypeptide) genes by peripheral blood whole-exon sequencing. Serum leucine and valine concentrations were significantly higher than normal values. Recipients ranged in age from 0.75 to 9 years old. Three patients underwent auxiliary liver transplantation, and the other children all underwent liver or partial liver transplantation. This case report was followed up for 25 to 79 months. The prognosis, growth, and development of patients were followed up. By the end of the last follow-up, all children had survived. All patients had normal serum leucine and valine concentrations after surgery. In case 1, portal vein stenosis post-operatively. In case 2, stenosis of hepatic artery and bile duct occurred. In case 5, hepatic artery and portal vein stenosis occurred, resulting in graft loss.
CONCLUSIONS: The findings from our center support the findings from other pediatric liver transplant centers that liver transplantation using MSUD donors can have successful outcomes without the development of MSUD in the recipient.
Keywords: Maple Syrup Urine Disease, Liver Transplantation, Pediatrics, Child, Humans, Infant, Child, Preschool, Living Donors, 3-Methyl-2-Oxobutanoate Dehydrogenase (Lipoamide), Leucine, Constriction, Pathologic, Valine
Background
The number of donors for liver transplantation is far less than the number of patients who need liver transplantation because of various liver diseases. This is a serious problem for every liver transplant doctor. Therefore, liver transplant surgeons have developed new ways to expand the liver pool, including split liver transplantation, living donor liver transplantation, broadening donor standards, and using donation after cardiac death (DCD). Futado et al first implemented and reported that patients with familial amyloid polyneuropathy (FAP) received the liver of DCD and transplanted their livers to patients with liver cancer in 1995 [1]. Since then, major transplantation centers have explored use of livers of patients with different inherited metabolic diseases as the donor liver for DLT or auxiliary liver transplantation, expanding the donor pool [2]. DLT involves using the diseased liver of one liver transplant recipient as a graft to another patient, passing it down like a domino [3]. Auxiliary liver transplantation involves the removal of part of the diseased liver and transplantation of a new part of the liver. The diseased liver remains in place to act as an auxiliary liver until the graft reaches sufficient volume [4]. However, with the passage of time, the diseased liver of several patients with genetic metabolic diseases, including FAP, as graft of domino liver transplantation, developed genetic metabolic diseases carried by the graft.
MSUD is an inherited autosomal recessive disease. This inherited disease is caused by loss of BCKAD complex’s quantity and activity. BCKAD complex is composed of decarboxylase (including E1 α polypeptide and E1 β polypeptide), dihydrolipoyl transacetylase (E2 polypeptide), and dihydrolipoamide dehydrogenase (E3 polypeptide). E1 α polypeptide is encoded by the BCKDHA gene. E1 β polypeptide is encoded by the BCKDHB gene. E2 polypeptide is encoded by the DBT (dihydrolipoamide branched-chain transacylase) gene. E3 is encoded by the DLD (dihydrolipoamide dehydrogenase) gene. At the same time, BCKAD complex’s function is regulated by 2 regulatory enzymes, BCKAD complex phosphorylase and BCKAD complex kinase. They regulate the activity of BCKAD complex through reversible phosphorylation and dephosphorylation. Loss of BCKAD complex’s quantity and activity prevents branched-chain amino acids from converting into their respective α-ketoacids by the enzyme branched-chain amino acid transamine, and from entering the pyruvate cycle and tricarboxylic acid cycle [2]. The mutation causes leucine, isoleucine, and valine, and their related α-ketoacids, to accumulate in the body. The disease is typically characterized by neonatal dysplasia, delayed developmental milestones, feeding difficulties, and a maple syrup smell in urine or cerumen [5]. Treatment includes close metabolic monitoring and dietary restriction of branched amino acids. If left untreated, irreversible neurological damage and metabolic disaster can occur. Good clinical outcomes can be expected if treatment is initiated early [6]. Patients need to follow a special diet for life to reduce their intake of branched-chain amino acids. This is a heavy financial burden for patients and their families [7]. Even with appropriate diet and drug treatment, elderly patients may have brain injury, chronic psychological burden, and death [8]. BCKAD complex is expressed and active in tissues of the whole body, mainly in skeletal muscle (about 60%), liver (about 10~15%), brain (about 10~20%), and kidney (about 10%) [9]. Liver transplantation is an internationally recognized treatment for the lack of BCKAD complex in patients with MSUD [10]. The liver of patients with MSUD cannot metabolize branched-chain amino acids, but other metabolic and synthetic functions are normal. Therefore, livers of patients with MSUD can be transplanted safely to patients without MSUD by domino liver transplantation, which is theoretically feasible. Using the livers of patients with MSUD as grafts for DLT can improve organ utilization and expand the donor pool. Here, we report that the livers of MSUD patients were transplanted to children with ornithine transcarboxylase deficiency (OTCD), Alagille syndrome, and biliary atresia. Therefore, this report is of 5 cases of DLT from live donors with MSUD from a single transplant center.
Case Series
ETHICS APPROVAL:
This study was approved by the Ethics Committee of the Beijing Friendship Hospital, Capital Medical University (No. 2018-P2111-01). Written informed consent was obtained from all participants.
PATIENT PERMISSION/CONSENT DECLARATIONS:
All patients signed surgical consent forms and agreed to participate in clinical observation and experiments.
DIAGNOSIS OF END-STAGE LIVER DISEASE IN THE RECIPIENTS:
All 3 OTCD children were confirmed by peripheral blood whole-exon gene test to have a pathogenic mutation of OTC (ornithine carbamoyl transferase) gene. Meanwhile, the patients suffered from recurrent coma caused by hyperammonemia. In case 3, pathogenic mutation of JAG1 (jagged 1) gene was confirmed by peripheral blood whole-exon sequencing. Table 2 shows the gene of the children with OTCD and Alagille syndrome, and Table 3 shows the Pediatric End-Stage Liver Disease score (PELD).
DIAGNOSIS OF MSUD IN THE DONOR:
All MSUD donors were confirmed to have disease-causing mutations in BCKDHA or BCKDHB genes by peripheral blood whole-exon sequencing. Serum leucine and valine concentrations were significantly higher than normal values. Table 4 shows the genes of the 4 MSUD children.
METHOD OF LIVER PROCURING:
Case 1 and Case 5 received liver from the same patient with MSUD and underwent auxiliary liver transplantation. Case 1 obtained the right half of the graft and Case 5 obtained the left half. The MSUD patient received liver from organ allocation and his liver was completely removed, including the inferior vena cava. Case 2 received the right half of the liver of the patient with MSUD and underwent auxiliary liver transplantation. The MSUD patient received the left lateral lobe liver donated by his father and underwent a living donor liver transplantation, whose liver was completely removed, except for the posterior inferior vena cava. Case 3 and Case 4 received the whole livers of patients with MSUD. The donor of Case 3 received the left half of liver from organ allocation. The donor of Case 4 received a whole liver from organ allocation.
METHOD OF DOMINO LIVER TRANSPLANTATION:
Case 4 underwent classic orthotopic liver transplantation, and the other children underwent piggyback liver transplantation. Case 2 underwent end-to-end biliary anastomosis, while the other children underwent Roux-en-Y operation to reconstruct the digestive tract, and the biliary tract of donor was anastomosed with the jejunum of the recipient. All patients except Case 4 received blood group-compatible liver transplantation.
IMMUNOSUPPRESSION:
Immunosuppression of Case 1, Case 2, Case 3, and Case 4 included mycophenolate mofetil (MMF)/tacrolimus/pred. Immunosuppression of Case 5 included cyclosporine/pred. Case 5 received cyclosporine instead of tacrolimus because tacrolimus does not reach a satisfactory blood concentration.
Results
Prognosis
CASE 1:
On the 6th day after the operation, B-ultrasound showed Case 1 had stenosis of the right portal vein of the transplanted liver, which was treated by portal vein balloon dilatation. Two months after the operation, calculi in the right ureter and right renal pelvis were developed. The calculi were removed by ureteroscopic laser lithotripsy. One year after the operation, the child’s growth and development had returned to normal (weight 32 kg, height 135 cm).
CASE 2:
On the second day after surgery, B-ultrasound showed Case 2 had arterial occlusion of the right half transplanted liver. We tried to use alprostadil to dilate the artery and warfarin as anticoagulant therapy. The hepatic artery of the right half of the transplanted liver formed collateral circulation on day 20 after surgery. At 34 days after surgery, the serum ammonia concentration decreased to normal. Two months after surgery, B-ultrasound showed an intrahepatic bile duct dilated in the left half liver, and cholangiography showed a bile duct in the left half liver was severely stenosed. Meanwhile, γ-glutamyl transpeptidase (GGT) and alkaline phosphatase (ALP) increased. We tried to use percutaneous transhepatic cholangial drainage (PTCD) as therapy, but it failed. Therefore, at 9 months after surgery, we had to perform a hilar cholangioplasty to re-anastomose the bile duct of the right half transplanted liver.
CASE 5:
On the third day after surgery, B-ultrasound showed Case 5 had hepatic artery occlusion in the left liver. We tried to use alprostadil to dilate the artery and unfractionated heparin for anticoagulant therapy. On the 14th day after surgery, B-ultrasound revealed the formation of collateral arteries in the left half transplanted liver. At 115 days after surgery, B-ultrasound showed slow hepatic portal vein flow in the transplanted left half liver (8.4 cm/s) combined with portal vein stenosis (0.2 cm). We had to perform an emergency portal venography and embolize the right posterior portal vein, but there was no obvious therapeutic effect. Two years after the operation, the child received DCD liver transplantation because of graft loss and recurrence of primary disease.
LABORATORY ANALYSIS:
After the operation, all 5 recipients recovered well in the early stage, and the branched-chain amino acids levels were normal. Figures 1 and 2 show the changes of serum ALT and total bilirubin in the 5 children before and after the operations. The serum ammonia concentration of 3 children with OTCD before and after operations is shown in Figure 3. By the end of the last follow-up on December 31, 2022, all children had survived, showing that liver grafts from children with MSUD can be used in domino liver transplantation.
Discussion
Since Professor Bruce implemented and reported the first case of using the liver of MSUD patients as the donor liver for domino liver transplantation in 2005 [11], a total of 54 patients [2,11–19] have been reported in the world and received the liver of maple syrup urine patients (Table 5). None of them reported that MSUD had been obtained from the liver of patients with MSUD, and there was no difference in the postoperative recovery and complications between the liver and the donor liver. The primary disease was relieved [17]. We reported the largest set of reported cases in China of DLT from live donors with MSUD. In the 5 children we reported, the same ALT and serum total bilirubin quickly returned to normal in the early postoperative period, and the blood ammonia of 3 children with OTCD also quickly decreased to normal. There was no restriction of diet after the operation. During the longest 5-year follow-up after surgery, there were no symptoms of MSUD or increase of serum branched-chain amino acids.
Generally, the liver of patients with genetic metabolic diseases was used as a domino liver donor, which was usually used for patients with advanced tumors and with relatively short life expectancy. However, according to our experience and reported cases, the liver of patients with MSUD can be used as a domino liver donor; this can improve recipient prognosis and solve the problem of patients without MSUD. The liver quality of patients with MSUD is excellent. It is safe to transplant the liver as a donor to patients with non-maple syrup urine disease donors.
However, the livers of patients with MSUD were used as grafts to DLT, which were procured in a different way from the common procurement from donation after cardiac death (DCD). For MSUD donor domino liver transplantation, we have some experience. Usually, when children with MSUD are undergoing liver transplantation, they are young and the proper hepatic artery is relatively slender. To ensure that children with MSUD retain an artery of sufficient length and relatively appropriate caliber to anastomose with the artery of graft, and to ensure an artery with sufficient length and relatively appropriate diameter can be used for anastomosis recipient’s artery without MSUD, the disconnected artery needs to be very carefully located so that livers of MSUD patients can be used as a graft to DLT. This requires close communication between doctors who operate on MSUD patients and those who perform domino liver transplantation. It is unclear how to reserve sufficient length outflow of MSUD patients and the graft while at the same time creating a sufficiently long outflow to accommodate the recipient’s inferior vena cava. Our rule of thumb is to keep the length of one artery and the outflow tract as long as possible in children with MSUD. When the artery of MSUD children’s liver is anastomosed with the recipient as the donor liver of domino, if the length of the artery and outflow tract is insufficient, angioplasty and reconstruction can be carried out in the background. The portal vein of the recipient without MSUD is usually selected as the vascular patch for outflow tract reconstruction, and the gastroduodenal artery of the recipient is usually anastomosed with the hepatic artery of the MSUD patients’ liver.
No cases of assisted liver transplantation have been reported in the past, but the cases we reported included 3 cases of assisted liver transplantation. Auxiliary liver transplantation was originally proposed as a treatment for reversible fulminant liver failure [20]. Auxiliary liver transplantation with a living related partial graft or a domino liver graft was initially introduced as a temporary or permanent support for patients, as it avoided small-for-size syndrome [21]. Our center since expanded the application of this auxiliary liver transplantation technique for use in the treatment of metabolic liver disease. We demonstrate that non-cirrhotic metabolic liver domino grafts can be used for auxiliary liver transplantation in patients diagnosed with alternative metabolic liver disease as a means of preserving the natural liver. Such a goal can be achieved by auxiliary liver transplantation through its ability to achieve a mutual compensation of metabolic defects between the residual natural liver and domino liver graft. In this way, it can ensure that a normal metabolic functional phenotype is achieved in the same recipient [22]. The recipient does not have 2 metabolic disorders after surgery [23], and 3 children with OTCD were given auxiliary liver transplantation using livers from MSUD. Because the 3 children’s PELD scores were very low, it was difficult for them to be assigned to livers in the Chinese organ allocation system, but the high levels of ammonia are damaging the children’s nervous systems. Family members cannot donate part of their livers. All 3 children were waiting for liver transplantation because of liver-related inherited metabolic diseases. One child had decreased blood flow in the artery and portal vein on one side of the graft after the operation. Although we tried many times to increase the blood flow on the donor side by embolizing the portal vein on the recipient side, the effect was not obvious. The child finally received DCD donor liver transplantation because of graft loss and recurrence of the primary disease. In the remaining 2 children, although the blood flow on one side of the graft did not decrease, we monitored the volume ratio of the graft to the recipient side by hepatobiliary dynamic phenomenon technology and found that the volume ratio of the graft was decreasing. The prognosis of these 2 children is also predictable. This is a difficult problem for all attempts to use the liver of patients with genetic and metabolic diseases as an auxiliary liver transplantation donor for the treatment of non-sclerosing liver disease. The competition of portal vein blood flow between donor and recipient livers is the main reason, and various centers are also trying to find solutions [24–29]. However, we believe that the graft atrophy and graft failure after DLT cannot be simply explained by the problem of blood runoff, which still has an unknown immune mechanism. The results of our clinical practice prove that auxiliary liver transplantation is feasible in DLT with MSUD patients as the donors. Use of auxiliary liver transplantation is very important in expanding the donor liver pool and shortening the waiting time.
The liver of MSUD patients has excellent curative effect as a donor liver for domino liver transplantation. It can be as practical as normal livers for non-MSUD patients. However, the vast majority of MSUD domino liver transplantation does not follow the conventional distribution system but is distributed according to size matching. Patients with low body weight (newborns or adolescents) and low scores of end-stage liver disease in children are preferred. Some scholars believe that the liver of MSUD patients should be uniformly distributed by the government [18] to make better use of this precious resource.
The number of cases reported in our case report is relatively small compared to other centers, but due to characteristics of our center, the operations are relatively more complex.
Conclusions
Our review of the literature and analysis of our reported cases show it is safe and reliable for non-MSUD patients to receive donor livers from MSUD patients, especially in whole-liver or segmental liver transplantation. The application of the liver of MSUD patients in DLT is of great significance to expand the liver source pool and reduce the mortality of patients on the waiting list. The donor livers of MSUD patients should be further studied as auxiliary domino liver donors to solve the problems of long-term graft atrophy and failure. Auxiliary liver transplantation can safely be used in domino liver transplantation from live donors with maple syrup urine disease. The findings from our center support the findings from other pediatric liver transplant centers that liver transplantation using MSUD donors can have successful outcomes without the development of MSUD in the recipient. These findings may allow expansion of the donor pool of liver grafts for pediatric liver transplantation.
Figures
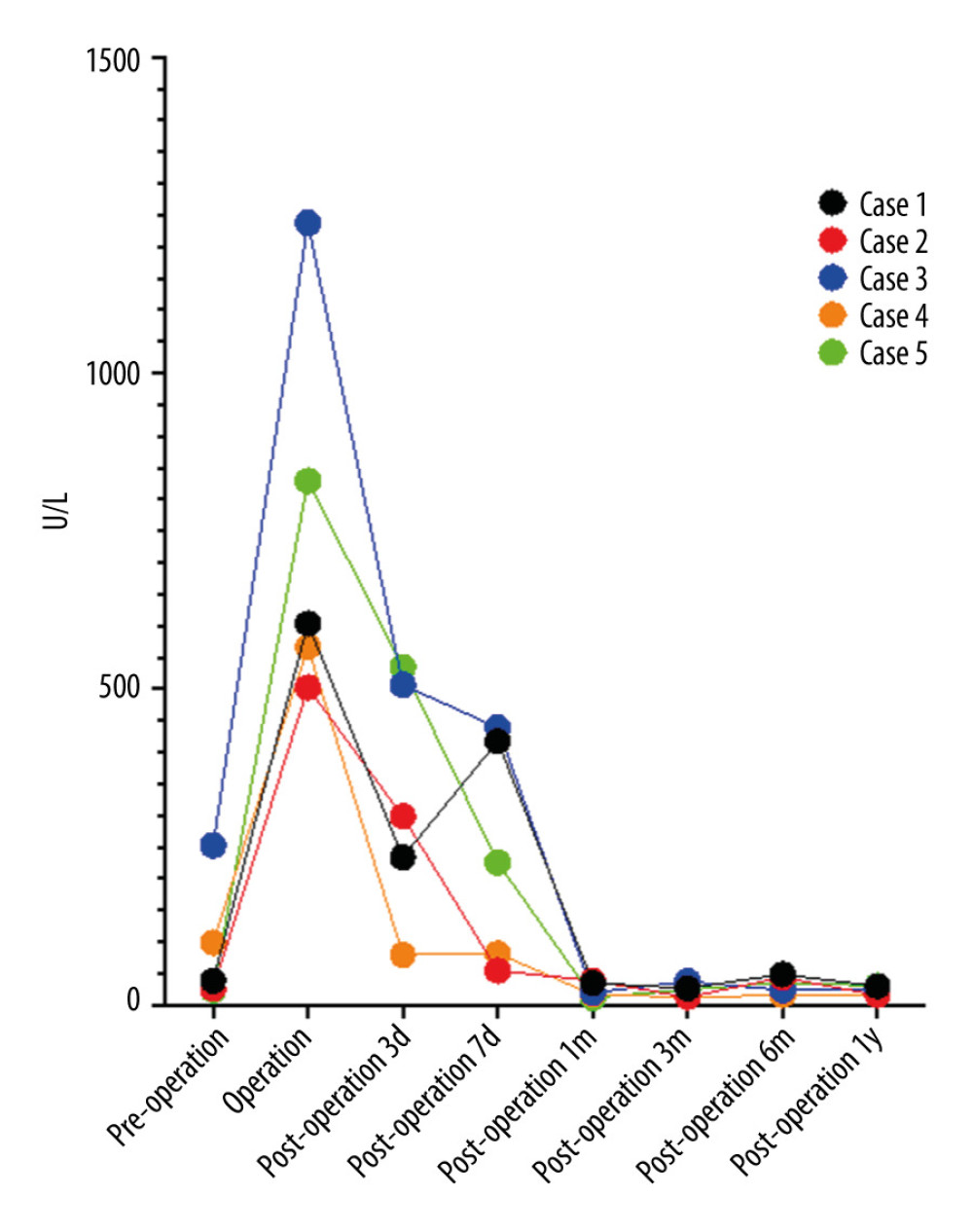 Figure 1. Serum alanine aminotransferase levels before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 1. Serum alanine aminotransferase levels before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.  Figure 2. Serum total bilirubin concentration before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 2. Serum total bilirubin concentration before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022. 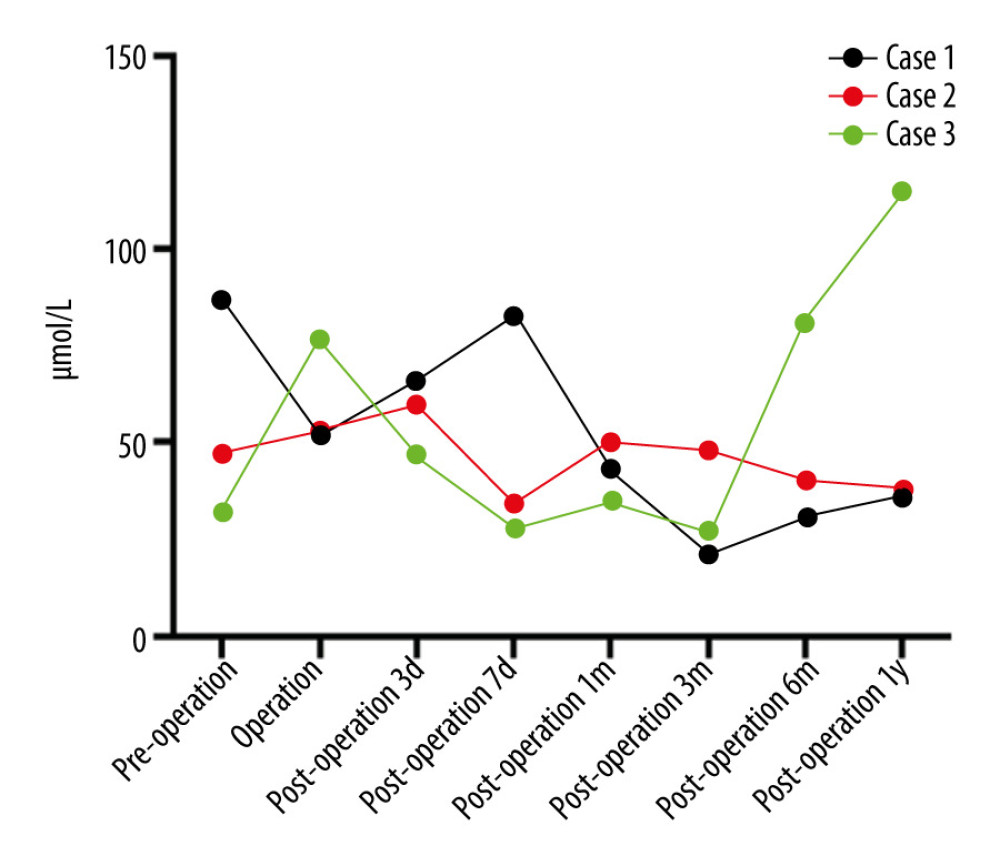 Figure 3. Serum ammonia concentration before and after liver transplantation in 3 children with OTCD. OTCD – ornithine transcarboxylase deficiency. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 3. Serum ammonia concentration before and after liver transplantation in 3 children with OTCD. OTCD – ornithine transcarboxylase deficiency. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022. References
1. Furtado A, Tomé L, Oliveira FJ, Sequential liver transplantation: Transplant Proc, 1997; 29(1–2); 467-68
2. Celik N, Squires JE, Soltys K, Domino liver transplantation for select metabolic disorders: Expanding the living donor pool: JIMD Rep, 2019; 48(1); 83-89
3. Marques HP, Barros I, Li J, Current update in domino liver transplantation: Int J Surg, 2020; 82S; 163-68
4. Lim C, Turco C, Balci D, Auxiliary liver transplantation for cirrhosis: From APOLT to RAPID: A scoping review: Ann Surg, 2022; 275(3); 551-59
5. Blackburn PR, Gass JM, Vairo FPE, Maple syrup urine disease: Mechanisms and management: Appl Clin Genet, 2017; 10; 57-66
6. Hassan SA, Gupta V, Maple syrup urine disease: StatPearls September 5, 2022, Treasure Island (FL), StatPearls Publishing
7. Packman W, Henderson SL, Mehta I, Psychosocial issues in families affected by maple syrup urine disease: J Genet Couns, 2007; 16(6); 799-809
8. Strauss KA, Carson VJ, Soltys K, Branched-chain α-ketoacid dehydrogenase deficiency (maple syrup urine disease): Treatment, biomarkers, and outcomes: Mol Genet Metab, 2020; 129(3); 193-206
9. Suryawan A, Hawes JW, Harris RA, A molecular model of human branched-chain amino acid metabolism: Am J Clin Nutr, 1998; 68(1); 72-81
10. Wendel U, Saudubray JM, Bodner A, Schadewaldt P, Liver transplantation in maple syrup urine disease: Eur J Pediatr, 1999; 158(2); 60-64
11. Barshop BA, Khanna A, Domino hepatic transplantation in maple syrup urine disease: N Engl J Med, 2005; 353(22); 2410-11
12. Badell IR, Hanish SI, Hughes CB, Domino liver transplantation in maple syrup urine disease: A case report and review of the literature: Transplant Proc, 2013; 45(2); 806-9
13. Feier FH, Miura IK, Fonseca EA, Successful domino liver transplantation in maple syrup urine disease using a related living donor: Braz J Med Biol Res, 2014; 47(6); 522-26
14. Shimizu S, Sakamoto S, Fukuda A, Surgical technique and the long-term outcomes of pediatric living donor domino liver transplantation from patients with maple syrup urine disease: Pediatr Transplant, 2022; 26(2); e14174
15. Mohan N, Karkra S, Rastogi A, Living donor liver transplantation in maple syrup urine disease – case series and world’s youngest domino liver donor and recipient: Pediatr Transplant, 2016; 20(3); 395-400
16. Roda KMO, Vincenzi R, Fonseca EA, Domino liver transplant in maple syrup urine disease: Technical details of cases in which the first surgery involved a living donor: Transplantation, 2019; 103(3); 536-43
17. Celik N, Kelly B, Soltys K, Technique and outcome of domino liver transplantation from patients with maple syrup urine disease: Expanding the donor pool for live donor liver transplantation: Clin Transplant, 2019; 33(11); e13721
18. Herden U, Grabhorn E, Santer R, Surgical aspects of liver transplantation and domino liver transplantation in maple syrup urine disease: Analysis of 15 donor-recipient pairs: Liver Transpl, 2019; 25(6); 889-900
19. Shimizu S, Sakamoto S, Fukuda A, Surgical technique and the long-term outcomes of pediatric living donor domino liver transplantation from patients with maple syrup urine disease: Pediatr Transplant, 2022; 26(2); e14174
19. Min X, Chao S, Hong Q, Clinical analysis of domino liver transplantation in children: Electronic Journal of Practical Organ Transplantation, 2020; 8(6); 451-56
20. Terpstra OT, Auxiliary liver grafting: A new concept in liver transplantation: Lancet, 1993; 342(8874); 758
21. Inomata Y, Kiuchi T, Kim I, Auxiliary partial orthotopic living donor liver transplantation as an aid for small-for-size grafts in larger recipients: Transplantation, 1999; 67(10); 1314-19
22. Qu W, Wei L, Zhu ZJ, Considerations for use of domino cross-auxiliary liver transplantation in metabolic liver diseases: A review of case studies: Transplantation, 2019; 103(9); 1916-20
23. Zhu ZJ, Wei L, Qu W, First case of cross-auxiliary double domino donor liver transplantation: World J Gastroenterol, 2017; 23(44); 7939-44
24. Kim BS, Joo SH, Lee SH, Auxiliary partial orthotopic liver transplantation for adult onset type II citrullinemia: J Korean Surg Soc, 2011; 80(Suppl 1); S51-S54
25. Kasahara M, Takada Y, Kozaki K, Functional portal flow competition after auxiliary partial orthotopic living donor liver transplantation in noncirrhotic metabolic liver disease: J Pediatr Surg, 2004; 39(7); 1138-41
26. Kasahara M, Takada Y, Egawa H, Auxiliary partial orthotopic living donor liver transplantation: Kyoto University experience: Am J Transplant, 2005; 5(3); 558-65
27. Rela M, Bharathan A, Palaniappan K, Portal flow modulation in auxiliary partial orthotopic liver transplantation: Pediatr Transplant, 2015; 19(3); 255-60
28. Shanmugam NP, Perumalla R, Gopinath R, Auxiliary liver transplantation: A form of gene therapy in selective metabolic disorders: J Clin Exp Hepatol, 2011; 1(2); 118-20
29. Broering DC, Walter J, Bassas AF, Overcoming the portal steal phenomenon in auxiliary partial orthotopic liver transplantation by modulation of the venous outflow of the native liver: Liver Transpl, 2005; 11(9); 1140-43
Figures
 Figure 1. Serum alanine aminotransferase levels before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 1. Serum alanine aminotransferase levels before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022. Figure 2. Serum total bilirubin concentration before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 2. Serum total bilirubin concentration before and after liver transplantation. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022. Figure 3. Serum ammonia concentration before and after liver transplantation in 3 children with OTCD. OTCD – ornithine transcarboxylase deficiency. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022.
Figure 3. Serum ammonia concentration before and after liver transplantation in 3 children with OTCD. OTCD – ornithine transcarboxylase deficiency. Figure was created using Prism 9 for mac OS Version 9.4.1 (458), July 18, 2022. Tables
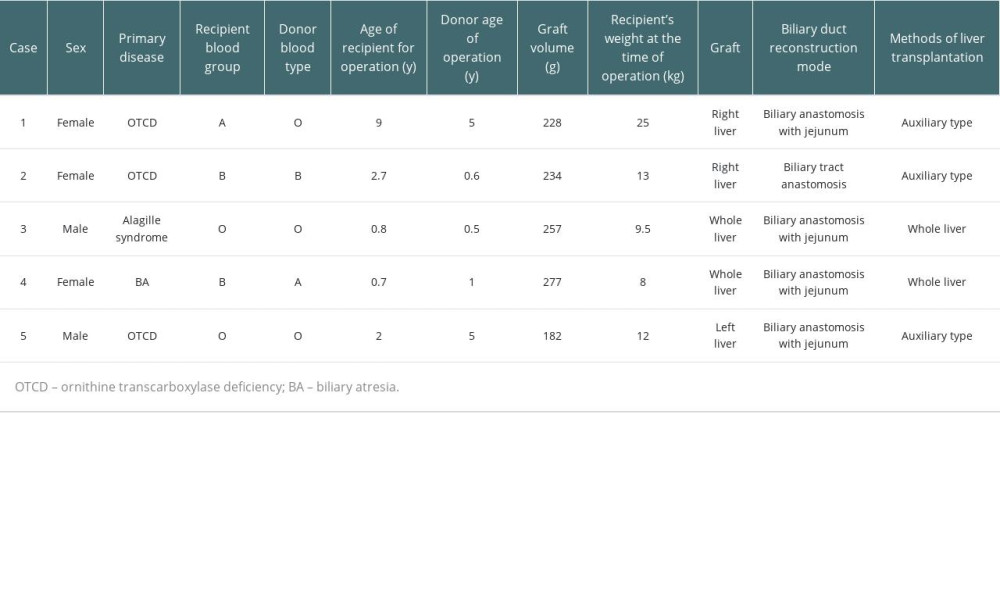 Table 1. General conditions of 5 children.
Table 1. General conditions of 5 children.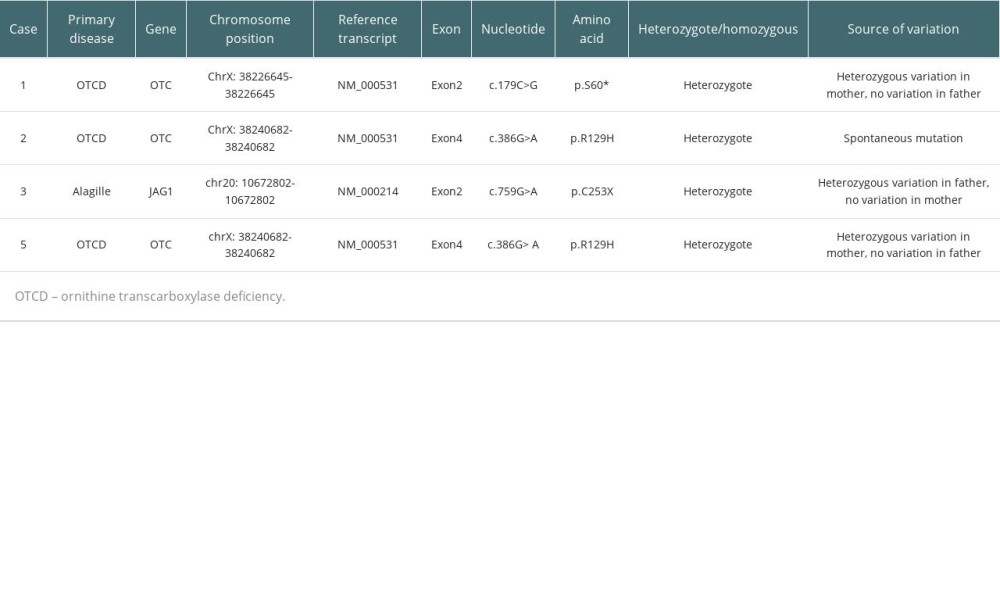 Table 2. Genes of the 3 OTCD children and Alagille child.
Table 2. Genes of the 3 OTCD children and Alagille child.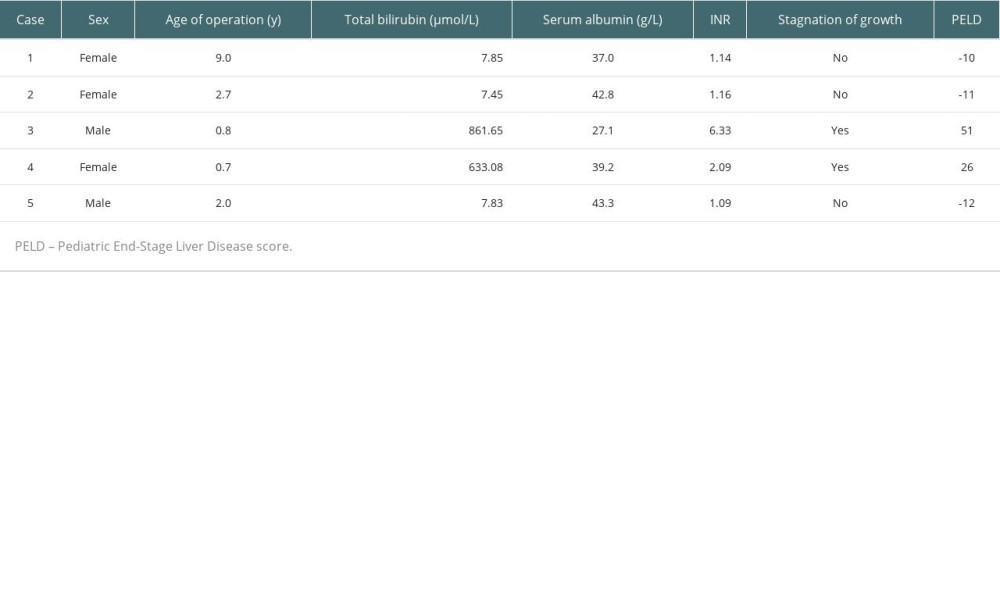 Table 3. Pediatric End-Stage Liver Disease scores of 5 children.
Table 3. Pediatric End-Stage Liver Disease scores of 5 children.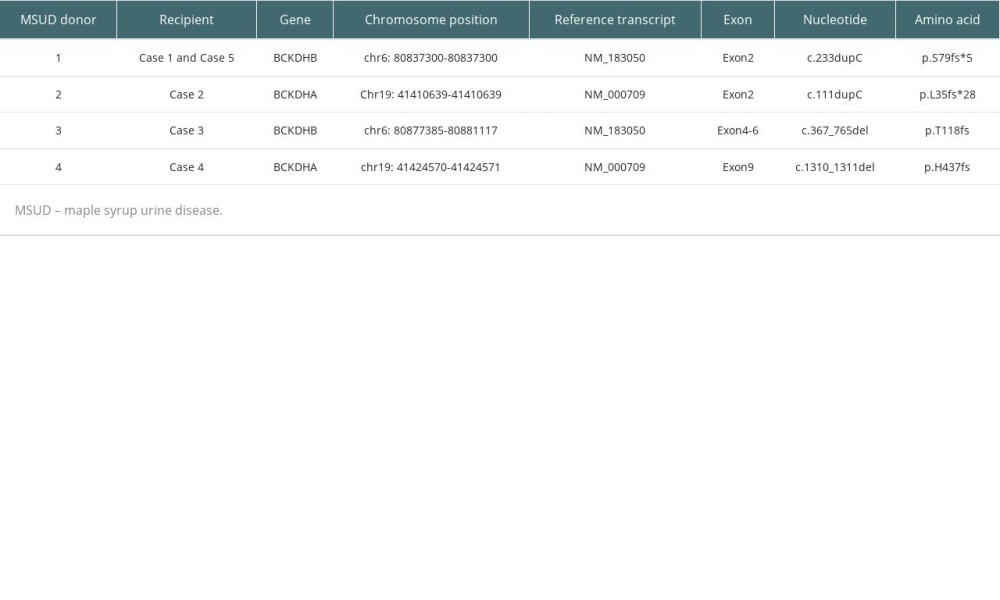 Table 4. Genes of the 4 MSUD children.
Table 4. Genes of the 4 MSUD children. Table 5. DLT from live donors with MSUD.
Table 5. DLT from live donors with MSUD. Table 1. General conditions of 5 children.
Table 1. General conditions of 5 children. Table 2. Genes of the 3 OTCD children and Alagille child.
Table 2. Genes of the 3 OTCD children and Alagille child. Table 3. Pediatric End-Stage Liver Disease scores of 5 children.
Table 3. Pediatric End-Stage Liver Disease scores of 5 children. Table 4. Genes of the 4 MSUD children.
Table 4. Genes of the 4 MSUD children. Table 5. DLT from live donors with MSUD.
Table 5. DLT from live donors with MSUD. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








