04 December 2020: Original Paper
Predictors of Mortality During Initial Liver Transplant Hospitalization and Investigation of Causes of Death
Ni Gong1BE, Chao Jia2CDF, He Huang3CEF, Jing Liu4BCD, XueTing Huang4CF, Qiquan Wan4ADG*DOI: 10.12659/AOT.926020
Ann Transplant 2020; 25:e926020
Abstract
BACKGROUND: Liver transplant (LT) remains a life-saving procedure with a high mortality rate. The present study investigated the causes of death and sought to identify predictive factors of mortality during the initial LT hospitalization.
MATERIAL AND METHODS: We retrieved data on first-time adult recipients who underwent LT between November 2017 and October 2019 receiving grafts from donation after citizen’s death. The risk factors for mortality during the initial LT hospitalization were confirmed by univariate analysis. We also analyzed the causes of death.
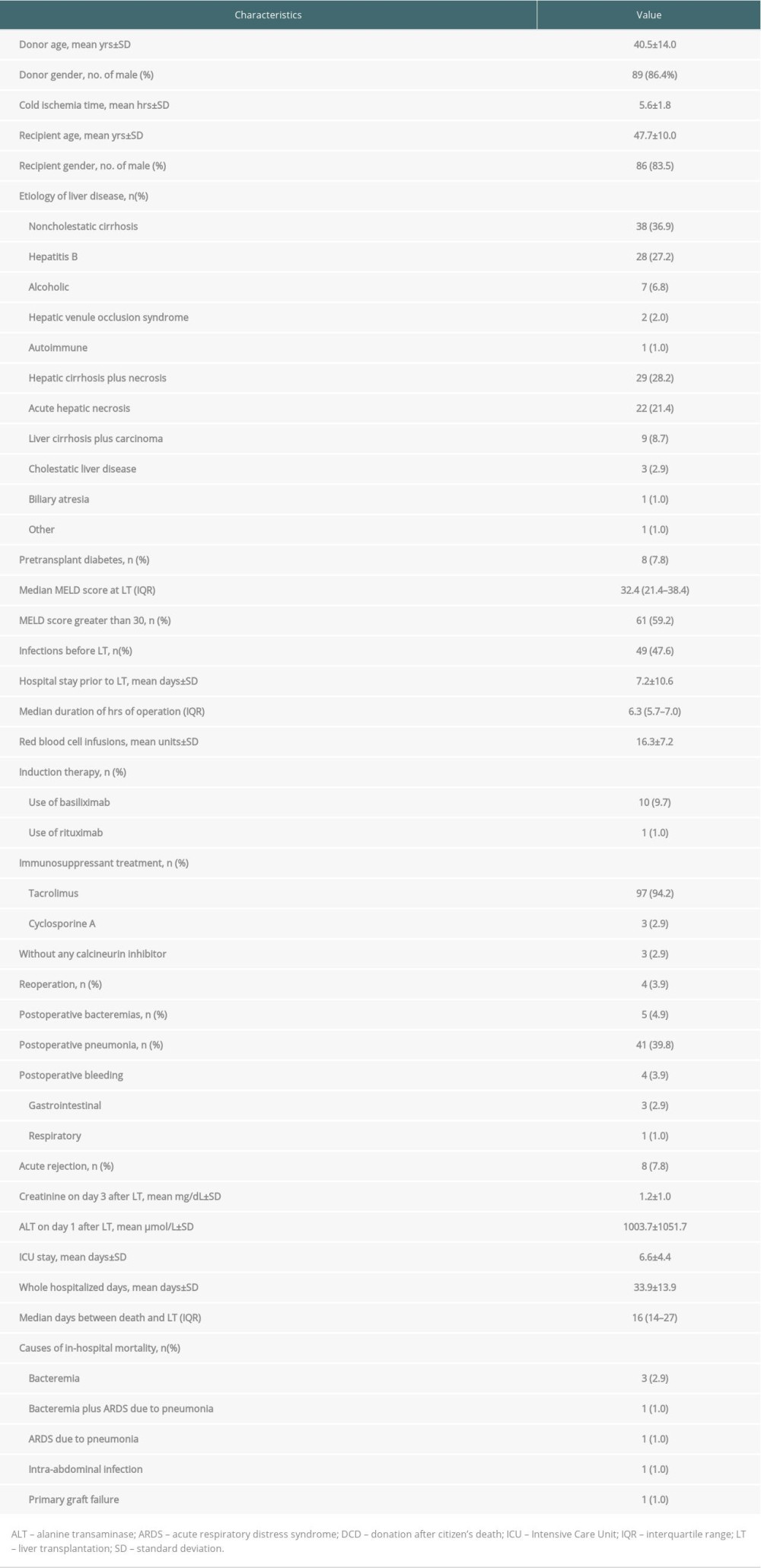
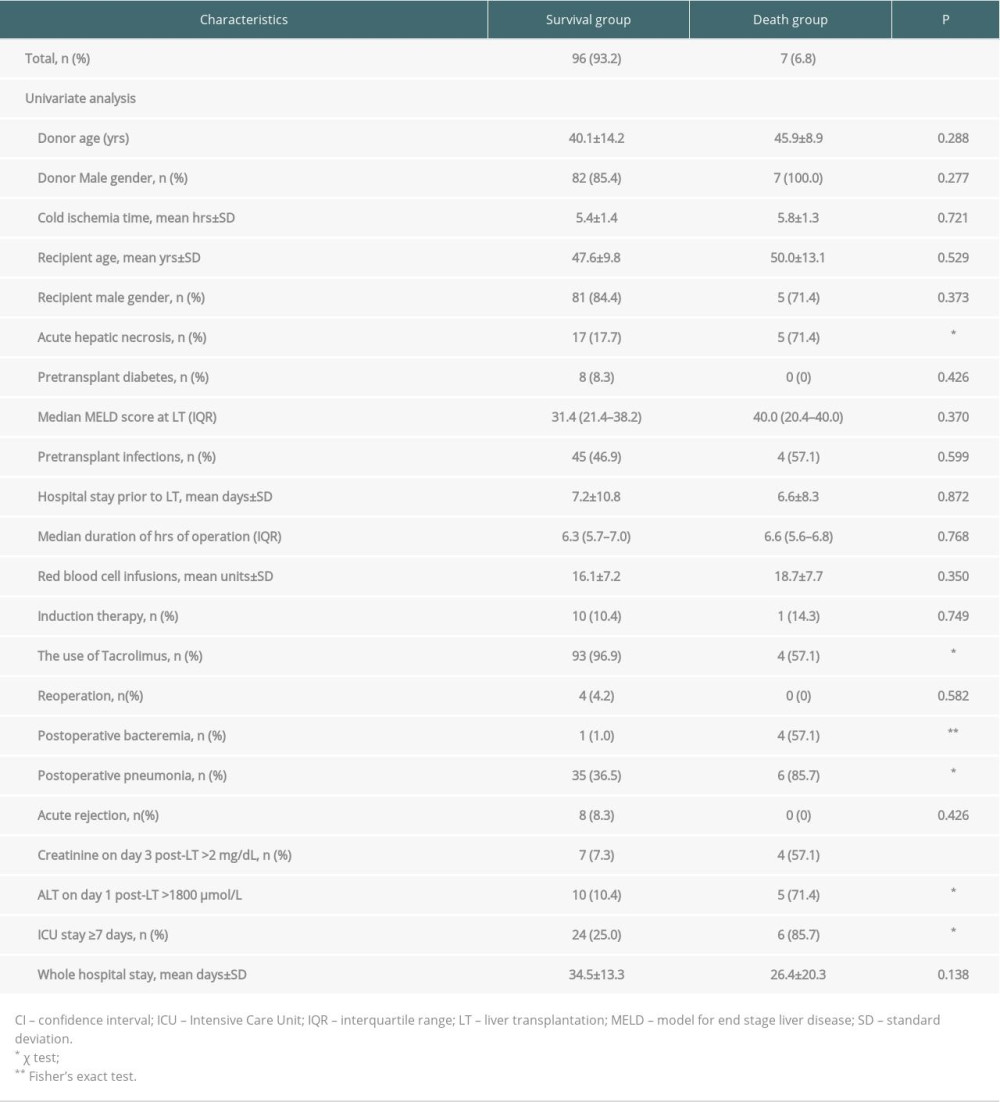
RESULTS: We enrolled 103 recipients, including 86 males and 17 females, with a mean age of 47.7 years. Thirty-eight (36.9%) recipients were labeled as non-cholestatic cirrhosis-related indications. Approximately 8% of all recipients had diabetes prior to LT. Induction therapy was used in 11 (10.7%) recipients, along with maintenance therapy. The median model for end-stage liver disease score at LT was 32.4 (21.4–38.4). The in-hospital mortality rate of LT recipients was 6.8% (7/103), and infections were responsible for most of the deaths (6/7). The 1 remaining death resulted from primary graft failure. Univariate analysis showed recipients with postoperative pneumonia (p<0.05), acute hepatic necrosis, and intensive care unit (ICU) stay ³7 days (both p<0.01), postoperative bacteremia, creatinine on day 3 after LT>2 mg/dL, and alanine transaminase on day 1 after LT >1800 µmol/L (all P<0.001) were much more likely to die.
CONCLUSIONS: In-hospital mortality of LT recipients was high, due in large part to infections. Acute hepatic necrosis, prolonged post-transplant ICU stays, certain types of postoperative infections, and postoperative liver and kidney dysfunction were potential risk factors for in-hospital mortality of LT recipients.
Keywords: Cause of Death, Liver Transplantation, Mortality, Risk Factors, End stage liver disease, Hospital Mortality, Hospitalization, Severity of Illness Index
Background
Liver transplantation (LT) is a life-sustaining but technically challenging procedure fraught with potential complications [1]. Although outcomes after LT have greatly improved with the development of surgical skills and perioperative care, immune-suppression management, and optimization in patient selection, post-LT 90-day, 1-year and 5-year mortality remain as high as 5%, 8.8–17% and 15–33%, respectively [1–10]. Rana et al. reported that mortality was highest within the first few days after transplantation in LT recipients, with the predominant contributors to death being technical factors [11]. Death related to cardiovascular/cerebrovascular/pulmonary/hemorrhage was the most frequent cause of death within the first 3 weeks after LT, and other causes of early death are operative complications, graft failure, and infections [2].
Given that the mortality rates are high and most previous studies have not specifically investigated the causes of death in LT recipients, the analyses of the timing, specific causes, and predictors of in-hospital death are of paramount importance to improve recipient quality of life [12–14]. We aimed to investigate the causes of death and the potential risk factors associated with mortality during the initial transplant hospitalization among LT recipients of grafts from donation after citizen’s death (DCD) in China.
Material and Methods
STUDY POPULATION:
A retrospective analysis was performed, and electronic medical records were reviewed to retrieve relevant data from the Third Xiangya Hospital, Central South University. Demographic, clinical, and laboratory data of first-time adult recipients undergoing LT from November 1, 2017 through October 31, 2019 were included to assess outcomes during initial LT hospitalization for all patients. Patients who underwent any type of prior transplants and required re-transplantation immediately following LT were excluded from this cohort. For antimicrobial prophylaxis, semi-synthetic penicillins/beta-lactamase inhibitors or a carbapenem were administered for 5 to 7 days following LT. The Ethics Committee of the Third Xiangya Hospital, Central South University approved this retrospective non-interventional study and waived the need for informed consent.
STUDY DESIGN AND DATA COLLECTION:
The enrolled patients were assigned to either the survival group or the mortality group. We analyzed the causes of and potential risk factors for mortality of recipients during their initial LT hospitalization. The immunosuppressive regimens comprised tacrolimus or cyclosporine and prednisone with/without mycophenolate mofetil or an additional monoclonal agent (basiliximab or rituximab). A few recipients did not receive any calcineurin inhibitors due to the severity of illness.
Clinical and laboratory data retrieved included demographics (age and sex), recipient pre-LT variables (LT indication, model for end-stage liver disease-MELD-score, diabetes, hospital stay, infections), donor-related variables (age, sex, cold ischemia time) and post-LT variables (primary graft dysfunction, type of immunosuppression, rejection episodes, bacteremias, pneumonias, reoperation, alanine transaminase at day 1 and creatinine at day 3 after LT, and ICU stay.
DEFINITION:
The definition of diabetes was that basal glycaemia was more than 126 mg/dl or 200 mg/dl any time during the day, in at least 3 consecutive tests, and/or antidiabetic drugs were needed [15]. Causes of death were stratified into primary graft failure, hemorrhage, infection, and pulmonary diseases [16]. Hospitalized days prior to LT was defined as hospitalization stays from the day of admission to the day of LT. Acute rejection was defined according to the Banff schema and was confirmed by biopsy [17]. MELD score was utilized to evaluate the severity of illness among LT candidates [18]. Bacteremia and pneumonia were defined in accordance with the criteria proposed by the Centers for Disease Control and Prevention [19].
STATISTICAL ANALYSIS:
Normally distributed quantitative data are described as means±(standard deviation). Non-normally distributed data are expressed as medians (1st–3rd quartile). Categorical variables are shown as frequencies and percentages. Univariate comparisons were made with the Mann-Whitney U test, the
Results
Over a 2-year period, we performed a total of 107 LTs using livers from DCD donors at our unit. Four patients were excluded: 1 recipient was under 18 years old at LT; 1 each underwent prior LT and kidney transplant; and 1 was retransplanted immediately after primary LT due to primary graft non-function. Thus, the sample comprised 103 adult patients (86 males and 17 females, mean age 47.7±10.0 years, range 19–73 years) who were divided into 1 of 2 groups: the survival (n=96 patients) group and the mortality group (n=7 patients).
Non-cholestatic cirrhosis was the predominant indication for LT (n=38, 36.9%), followed by hepatic cirrhosis in combination with necrosis (n=29, 28.2%), and acute hepatic necrosis (n=22, 21.4%). Eight (7.8%) patients were diagnosed with diabetes prior to LT. The median MELD score was 32.4 (interquartile range=21.4–38.4) and recipients with a MELD score greater than 30 constituted approximately 60% (61/103) of LT cases, with 42 (40.8%) of them having a MELD score greater than 35. Basiliximab or rituximab induction therapy was used in 11 (10.7%) recipients. Tacrolimus was prescribed in 94.2% of LT recipients. No calcineurin inhibitors were administrated in 3 patients due to the severity of illness. Mean hospital stay prior to LT, post-LT ICU, and whole hospital stays were 7.2±10.6, 6.6±4.4, and 33.9±13.9 days, respectively. Mean red blood cell infusion was 16.3±7.2 units during the initial LT hospitalization. In-hospital mortality was 6.8% (7 cases): 1 patient had bacteremia caused by
Table 2 shows that recipients in the mortality group were significantly more likely to have acute hepatic necrosis, postoperative pneumonia, and bacteremia (
Discussion
LT is a common solid-organ transplantation and is a life-saving treatment for acute liver failure, end-stage liver disease, and hepatocellular carcinoma [20]. We discovered that non-cholestatic cirrhosis (n=38, 36.9%), mainly due to hepatitis B, was the first indication for LT in our patients, which is in contrast to a Spanish study reporting that alcoholic cirrhosis remained a major indication for LT [21].
We found that the overall in-hospital mortality rate was 6.8%, in line with a recent study in which the overall in-hospital mortality rate was 6.3% [22]. We also found that the mortality rate of patients with acute hepatic necrosis was 22.7% following LT. Acute hepatic necrosis, characterized by hepatic encephalopathy and a bleeding tendency due to abrupt loss of liver function, has a high mortality rate and affects almost every organ system [23,24]. We discovered that the prevalence rate of acute hepatic necrosis was higher in the mortality versus survival group (71.4%
Longer ICU stay was previously reported to be significantly associated with mortality due to bacteremias and fungal infections in LT recipients [27–29]. We also found that length of ICU stay ≥7 days was associated with an increased mortality risk but not with length of the whole hospital stay, which was previously associated with mortality of LT recipients in a study of data from the United Network for Organ Sharing [2].
It was worth highlighting that in this study, creatinine >2 mg/dL on day 3 and alanine transaminase >1800 μmol/L on day 1 following LT were 2 potential risk factors for mortality. Our previous study and other studies found that kidney and liver dysfunctions were associated with mortality in LT recipients [2,7,30–32]. Highly standardized preoperative management strategies and improved surgical technique, therefore, are pivotal for reducing durations of operation, blood transfusions, and complications, thus further improving liver and kidney functions [1]. To prevent or reduce use of calcineurin inhibitors, which are associated with renal failure, early calcineurin inhibitors-free or calcineurin inhibitors minimization protocols should be considered as soon as possible in LT recipients with post-transplant impaired renal function [33].
The present study found that donor age and recipient age were not risk factors for mortality, in contrast with a previous study reporting that the risk of death among older recipients was about 3- to 4-fold higher than in younger ones [22]. The differences between these 2 studies are mainly related to the difference in sample size and age composition; our study had a smaller sample size and younger recipients. Other studies found combined recipient and donor age ≥120 years, or recipient and donor age, respectively, are among the strongest prognostic variables for post-transplant mortality in LT recipients [7,34]. Age >40 years is a donor characteristic that significantly increases risk of graft failure [33].
We observed that infections were the most common cause of death (n=6, 85.7%) in the present study, and 6 of the 7 deaths (85.7%) occurred within 30 days after LT. This finding agrees with the results of Watt et al. [35], who noted that infections were the leading cause of early death in LT recipients, but differs from other studies [2,36] reporting that early death was more likely related to graft or technical operative factors, and infection was the leading cause of death at 30–180 days after LT. The differences are mainly due to the different median MELD score at LT, which was 32.4 in our recipients but only about 20 in the studies by Baganate et al. and Piazza et al. [2,36]. Another reason might be that donor-derived infections are very common among LT recipients, and we previously reported that 3/67 LT recipients developed this type of infection in our center [37].
Most of the patients in our study who died had pre-LT infections. Our recipients with a pre-LT MELD score greater than 30 constituted approximately 60% of LT cases. These factors may have also contributed to the differences in findings between our study and other studies.
Patients who experienced a postoperative bacteremia or pneumonia were at a significantly higher risk for in-hospital mortality. Despite advances in surgical skills, perioperative management, immunosuppression, and prophylaxis, infections continue be a major cause of mortality after LT [38]. Bloodstream infection-associated mortality ranged from 10% to 52% in LT recipients [39]. Pneumonia, occurring in 8–23% of patients, led to a greater than 50% mortality rate in LT patients [38,40–42]. Infectious complications are a major cause of mortality, and prevention of infections is a cornerstone of treatment following LT [33]. Antimicrobial prophylaxis has contributed to increased patient survival [43]. It is of utmost importance to closely monitor post-LT patients with a suspected infection, and to apply effective preventive and prophylactic measures, including strict environmental infection control measures, appropriate isolation precautions, and hand hygiene practices. Studies have also shown a reduced risk of all kinds of infections with personalization of immune therapy, glycemic control, requirement of candidates to walk independently, and procalcitonin measurements for empiric antibiotic selection [44–46].
The use of tacrolimus was inversely correlated with a fatal outcome. The reason is not clear, but a possible explanation is that 3 of 7 patients assigned to the mortality group did not receive any calcineurin inhibitor due to the severity of infections, and it was thus plausible that the patients who did not use tacrolimus had a high mortality risk.
We examined a cohort of patients for whom the MELD score (international normalized ratio of prothrombin time, total bilirubin, and creatinine) was calculated to predict mortality among LT recipients and did not find an association between MELD score and mortality during the initial LT hospitalization. Although the MELD score accurately predicted pretransplant mortality, recent studies did not show its direct correlation with post-transplant mortality, which strongly agrees with our results [8,28,33,47,48]. Only a MELD score >35 provides a prediction of mortality following LT [49]. MELD does not reflect some manifestations of cirrhosis such as pulmonary complications of cirrhosis and hepatic encephalopathy, and miscellaneous liver diseases such as amyloidosis and primary hyperoxaluria [33]. Thus, MELD exception scores have been accepted and have triggered recent changes in allocation policy [8,33]. Serum sodium (MELD-Na), delta MELD, serum sodium, and age (integrated MELD) scores might be better predictors of mortality [50–52].
Cold ischemia time was not associated with mortality, in line with a recent report from Uruguay, which could be related to the short cold ischemia time (5.6±1.8 h) in our program [28]. Cold ischemia time, however, has been reported to be an independent transplant risk factor for graft loss and post-transplantation patient survival [33,34].
There are several limitations to this study. It was a single-center study; however, most of our findings were similar to those reported in the international literature. Other limitations include its retrospective design and limited sample size of patients who died, so that we could not consolidate the association between the statistically significant variables, identified by univariate comparisons, and in-hospital mortality by using a multivariable logistic regression model. The present study is of particular importance given that it is the first to define the mortality rate and explicitly characterize the risk factors for mortality during the initial LT hospitalization, which may facilitate quality improvement and help to minimize mortality. Larger prospective multicenter studies are warranted to provide a more comprehensive assessment of the causes of death and determine more specific risk factors for mortality.
Conclusions
In summary, our results should raise a red flag that extra attention must be paid to patients with acute hepatic necrosis as an indication for LT, post-LT infections, and kidney and liver dysfunction, as well as a prolonged ICU stay.
References
1. Balogh J, Gordon Burroughs S, Boktour M, Efficacy and cost-effectiveness of voriconazole prophylaxis for prevention of invasive aspergillosis in high-risk liver transplant recipients: Liver Transpl, 2016; 22; 163-70
2. Baganate F, Beal EW, Tumin D, Early mortality after liver transplantation: Defining the course and the cause: Surgery, 2018; 164; 694-704
3. Kim WR, Lake JR, Smith JM, OPTN/SRTR 2016 annual data report: Liver: Am J Transplant, 2018; 18(Suppl 1); 172-253
4. Ling Q, Dai H, Zhuang R, Predicting short-term survival after liver transplantation on 8 score systems: A national report from China Liver Transplant Registry: Sci Rep, 2017; 7; 42253
5. Fayek SA, Quintini C, Chavin KD, The current state of liver transplantation in the United States: Perspective from American Society of Transplant Surgeons (ASTS) Scientific Studies Committee and endorsed by ASTS Council: Am J Transplant, 2016; 16; 3093-104
6. Dutkowski P, Linecker M, DeOliveira ML, Challenges to liver transplantation and strategies to improve outcomes: Gastroenterology, 2015; 148; 307-23
7. Aloia TA, Knight R, Gaber AO, Analysis of liver transplant outcomes for United Network for Organ Sharing recipients 60 years old or older identifies multiple model for end-stage liver disease–independent prognostic factors: Liver Transpl, 2010; 16; 950-59
8. Kwong A, Kim WR, Lake JR, OPTN/SRTR 2018 Annual Data Report: Liver: Am J Transplant, 2020; 20(Suppl s1); 193-299
9. Cheng EY, Everly MJ, Trends of immunosuppression and outcomes following liver transplantation: An analysis of the United Network for Organ Sharing Registry: Clin Transpl, 2014; 13-26
10. Dutkowwsky P, Linecker M, De Oliveira ML, Challenges to liver transplantation and strategies to improve outcomes: Gastroenterology, 2015; 148; 307-23
11. Rana A, Gruessner A, Agopian VG, Survival benefit of solid-organ transplant in the United States: JAMA Surg, 2015; 150; 252-59
12. Bruns H, Lozanovski VJ, Schultze D, Prediction of postoperative mortality in liver transplantation in the era of MELD-based liver allocation: A multivariate analysis: PLoS One, 2014; 9; e98782
13. Sumskiene J, Kupcinskas L, Pundzius J, Prognostic factors for short and long-term survival in patients selected for liver transplantation: Medicina (Kaunas), 2005; 41; 39-46
14. Kim DJ, Lee SK, Jo JW, Prognosis after liver transplantation predicted by preoperative MELD score: Transplant Proc, 2006; 38; 2095-96
15. American Association of Clinical Endocrinologist/American College of Endocrinology (AACE/ACE), Obesity Task Force. AACE/ACE Position statement on the prevention, diagnosis, and treatment of obesity: Endocr Pract, 1998; 4; 297-330
16. United States Department of Health and Human Services, Organ Procurement and Transplantation Network: Standard Transplant Analysis Registry https://optn.transplant.hrsa.gov/data/requestdata/
17. , Banff schema for grading liver allograft rejection: An international consensus document: Hepatology, 1997; 25; 658-63
18. Axelrod DA, Schnitzler M, Salvalaggio PR, The economic impact of the utilization of liver allografts with high donor risk index: Am J Transplant, 2007; 7; 990-97
19. Horan TC, Andrus M, Dudeck M, CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting: Am J Infect Control, 2008; 36; 309-32
20. Department of Health, Human Services HRaS, Administration: OPTN/SRTR 2012 Annual Data Report, 2014, Rockville, MD, Organ Procurement and Transplantation Network (OPTN) and Scientific Registry of Transplant Recipients (SRTR)
21. Castelló B, Aguilera V, Blázquez MT, Post-transplantation outcome in non-alcoholic steatohepatitis cirrhosis: Comparison with alcoholic cirrhosis: Ann Hepatol, 2019; 18; 855-61
22. Gil E, Kim JM, Jeon K, Recipient age and mortality after liver transplantation: A population-based cohort study: Transplantation, 2018; 102; 2025-32
23. Grek A, Arasi L, Acute liver failure: AACN Adv Crit Care, 2016; 27; 420-29
24. Wang DW, Yin YM, Yao YM, Advances in the management of acute liver failure: World J Gastroenterol, 2013; 19; 7069-77
25. Olivari D, Mainardi V, Rando K, Risk factors of mortality after liver transplantation in Uruguay: Transplant Proc, 2018; 50; 499-502
26. Jo YY, Choi YS, Joo DJ, Pretransplant mortality predictors in living and deceased donor liver transplantation: J Chin Med Assoc, 2014; 77; 16-20
27. Shi SH, Kong HS, Xu J, Multidrug resistant gram-negative bacilli as predominant bacteremic pathogens in liver transplant recipients: Transpl Infect Dis, 2009; 11; 405-12
28. Liu X, Ling Z, Li L, Invasive fungal infections in liver transplantation: Int J Infect Dis, 2011; 15(5); e298-304
29. Singh N, Paterson DL, Gayowski T, Predicting bacteremia and bacteremic mortality in liver transplant recipients: Liver Transpl, 2000; 6; 54-61
30. Wan QQ, Ye QF, Ming YZ, The risk factors for mortality in deceased donor liver transplant recipients with bloodstream infections: Transplant Proc, 2013; 45; 305-7
31. Shi SH, Kong HS, Xu J, Multidrug resistant gram-negative bacilli as predominant bacteremic pathogens in liver transplant recipients: Transpl Infect Dis, 2009; 11; 405-12
32. Hong JC, Busuttil RW, Klintmalm GB, Outcome predictors in transplantation: Transplantation of the Liver Edition, 2015; 3, Philadelphia, Saunders
33. European Association for the Study of the Liver, EASL clinical practice guidelines: Liver transplantation: J Hepatol, 2016; 64; 433-85
34. Dutkowski P, Oberkofler CE, Slankamenac K, Are there better guidelines for allocation in liver transplantation? A novel score targeting justice and utility in the model for end-stage liver disease era: Ann Surg, 2011; 254; 745-53
35. Watt KD, Pedersen RA, Kremers WK, Evolution of causes and risk factors for mortality post-liver transplant: Results of the NIDDK long-term follow-up study: Am J Transplant, 2010; 10; 1420-27
36. Piazza NA, Singal AK, Frequency of cardiovascular events and effect on survival in liver transplant recipients for cirrhosis due to alcoholic or nonalcoholic steatohepatitis: Exp Clin Transplant, 2016; 14; 79-85
37. Ye QF, Zhou W, Wan QQ, Donor-derived infections among Chinese donation after cardiac death liver recipients: World J Gastroenterol, 2017; 23; 5809-16
38. Paterno F, Wilson GC, Wima K, Hospital utilization and consequences of readmissions after liver transplantation: Surgery, 2014; 156; 871-78
39. Shao M, Wan Q, Xie W, Bloodstream infections among solid organ transplant recipients: Epidemiology, microbiology, associated risk factors for morbility and mortality: Transplant Rev (Orlando), 2014; 28; 176-81
40. Hong SK, Hwang S, Lee SG, Pulmonary complications following adult liver transplantation: Transplant Proc, 2006; 38; 2979-81
41. Timothy S, Huprikar H: Transplant infections, 2016
42. Singh N, Gayowski T, Wagener M, Pulmonary infections in liver transplant recipients receiving tacrolimus. Changing pattern of microbial etiologies: Transplantation, 1996; 61; 396-401
43. Gavalda J, Vidal E, Lumbreras C, Infection prevention in solid organ transplantation: Enferm Infecc Microbiol Clin, 2012; 30; 27-33
44. Angarita SAK, Russell TA, Kaldas FM, Pneumonia after liver transplantation: Curr Opin Organ Transplant, 2017; 22; 328-35
45. Wallia A, Schmidt K, Johnson Oakes D, Glycemic control reduces infections in post-liver transplant patients: Results of a prospective, randomized study: J Clin Endocrinol Metab, 2017; 102(2); 451-59
46. Sato A, Kaido T, Iida T: Liver Transpl, 2016; 22; 436-45
47. Petrowsky H, Rana A, Kaldas FM, Liver transplantation in highest acuity recipients: Identifying factors to avoid futility: Ann Surg, 2014; 259; 186-94
48. Desai MN, Mange CK, Crawford M, Predicting outcome after liver transplantation: Utility of the model for end-stage liver disease and a newly derived discrimination function: Transplantation, 2004; 77; 99-106
49. Habib S, Berk B, Chang CC, MELD and prediction of post-liver transplantation survival: Liver Transpl, 2006; 12; 440-47
50. Kim WR, Biggins SW, Kremers WK, Hyponatremia and mortality among patients on the liver-transplant waiting list: N Engl J Med, 2008; 359; 1018-26
51. Huo TI, Wu JC, Lin HC, Evaluation of the increase in model for end-stage liver disease (DeltaMELD) score over time as a prognostic predictor in patients with advanced cirrhosis: Risk factor analysis and comparison with initial MELD and Child-Turcotte-Pugh score: J Hepatol, 2005; 42; 826-32
52. Merion RM, Wolfe RA, Dykstra DM, Longitudinal assessment of mortality risk among candidates for liver transplantation: Liver Transpl, 2003; 9; 12-18
In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588