30 October 2020: Meta-Analysis
Comparison of Surgical Techniques in Living Donor Nephrectomy: A Systematic Review and Bayesian Network Meta-Analysis
Qi Xiao1ABCDEFG, Biqi Fu2BCF, Keqin Song1BC, Sufen Chen1BC, Jianfeng Li1BCDF, Jiansheng Xiao1ABCFG*DOI: 10.12659/AOT.926677
Ann Transplant 2020; 25:e926677
Abstract
BACKGROUND: The aim of this study was to compare and evaluate surgical techniques used for living donor nephrectomy (LDN).
MATERIAL AND METHODS: We performed a meta-analysis to compare 4 surgical techniques: open LDN (OLDN), laparoscopic LDN (LLDN), hand-assisted LLDN (HALLDN), and robot-assisted LLDN (RLDN).
RESULTS: No significant differences were found among these surgical techniques in terms of BMI, donor postoperative complications, 1-year graft survival, and DGF. Compared to the OLDN, the other 3 surgical techniques preferred to harvest the left kidney. When the right kidney was chosen as a donor, OLDN was the first-choice surgical technique. EBL was significantly lower in the HALLDN, LLDN, and RLDN groups when compared to the OLDN group. However, operative time and WIT were significantly shorter in the OLDN group. The RLDN group had an increased rate of donor intraoperative complications and a significantly lower VAS on day 1. The OLDN group required more morphine intake than the LLDN group. The length of hospital stay was significantly longer and AR was significantly higher in the OLDN group than in the LLDN and HALLDN groups.
CONCLUSIONS: There are no significant differences in donor postoperative complications, recipient DGF, and graft survival among the 4 surgical techniques. OLDN reduces WIT and operation time, but increases EBL and AR. RLDN and LLDN reduce the length of hospital stay, morphine intake, and VAS, and thus accelerate recovery. However, RLDN is associated with increased intraoperative complications.
Keywords: Hand-Assisted Laparoscopy, Laparoscopy, Living Donors, Robotics, Bayes Theorem, Kidney, Kidney Transplantation, Nephrectomy, Network Meta-Analysis
Background
Kidney transplantation remains the treatment of choice for patients with end-stage kidney failure. Kidneys for transplantation are obtained either from a deceased donor or a living donor. The relative shortage of deceased donor kidneys has led to long waiting times for a kidney transplant, so living donor kidney transplantation is a more realistic option for patients. Living donor kidneys not only expand the donor pool, but offer better graft function and have longer graft survival than kidneys from deceased donors [1]. However, since living kidney donors are healthy individuals, it is of the utmost importance that the safety of donors is ensured so they can resume their normal activities as soon as possible [2].
The first open living donor nephrectomy (OLDN) was reported by Murray et al. in 1954, and since then has been a standard procedure for live kidney donation for many years [3]. However, postoperative pain, scarring, and other discomforts and complications associated with large flank incisions were found in many donors undergoing OLDN surgery. To solve these problems, laparoscopic living donor nephrectomy (LLDN) was introduced in 1995 by Ratner and colleagues to replace the OLDN approach [4]. Compared to the OLDN approach, LLDN is associated with less postoperative pain, shorter hospital stay, better cosmetic results, and quicker recovery [5]. Several technical modifications of laparoscopic surgery have been made, including hand-assisted laparoscopic surgery, robot-assisted laparoscopic surgery, retroperitoneoscopic access, laparoscopic single-site surgery, and natural orifice transluminal endoscopic surgery. The first hand-assisted laparoscopic living donor nephrectomy (HALLDN) was performed in 1998 [6]. HALLDN makes surgical dissection more efficient because of the multiple ways that hands and instruments can be used, significantly increasing the technical capability and resulting in a faster procedure and a shorter operative time. This helps surgeons feel the consistency of kidney tissues and take full advantage of the OLDN approach [7]. In 2002, Horgan [8] first reported robot-assisted laparoscopic living donor nephrectomy (RLDN). Compared to the standard laparoscopic surgery, this robotic system provides 3-dimensional vision with increased precision, thus enhancing the ability of surgeons to perform complex tasks in a laparoscopic environment [9]. Each of these laparoscopic surgery modifications has its own specific technical advantages.
Many studies have used the standard pairwise meta-analyses to compare surgical techniques available in living donor nephrectomy (LDN) for kidney transplantation. However, an insurmountable limitation of these meta-analyses is that only 2 surgical approaches can be directly compared. Surgeons face huge challenges in selecting the best surgical strategy. To address this important issue, we performed a network meta-analysis (NMA), which allows simultaneous comparisons of all surgical approaches in LDN. Another advantage of using NMA is that it enables the integration of both direct and indirect evidence from clinical trials and allows indirect comparisons of a variety of treatments that have not previously been directly compared in head-to-head studies. To guide the selection of surgical procedures for LDN, we systematically reviewed and summarized the NMA results of different surgical techniques.
Material and Methods
LITERATURE SEARCH STRATEGY:
This study was conducted in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. PubMed, EMBASE, and Cochrane Library databases were searched without any language restrictions. The following searching keywords were used in combination: living donor, nephrectomy, kidney transplantation, laparoscopy, robotics, and hand-assisted laparoscopy. All abstracts, clinical studies, and citations were reviewed. To try to collect more clinical trials, a manual search was also done to identify additional publications of relevant studies. The latest literature search date for this study was July 22, 2020. The literature search was done independently by 2 authors (B.F. and K.S.).
DATA EXTRACTION AND QUALITY ASSESSMENT:
Two authors (Q.X. and S.C.) independently extracted the following data from collected studies: first author, publication year, country, study design, mean age and standard deviation, sex, inclusion and exclusion criteria, total number of patients, and number of subjects undergoing each type of surgical method. Any disagreements between the reviewers occurring during analysis of outcomes of interest were resolved through discussion with the other authors (J. L. and J.X.). If the continuity data were provided as median and range, we estimated the mean difference (MD) and standard deviation (SD) based on the formula of Hozo et al. [10]. Study quality was evaluated using the Cochrane risk of bias assessment tool for random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. Studies were judged as low risk of bias, high risk of bias, or unclear risk of bias. Review Manager software version 5.3 was used to plot the quality assessment.
INCLUSION CRITERIA:
Articles were included only if they satisfied the following criteria: (1) adults undergoing nephrectomy for live organ donation; (2) surgical techniques involved: OLDN, including mini-open approach, transperitoneal approach, and extraperitoneal approach; LLDN, including conventional laparoscopic surgery, standard laparoscopic surgery, laparoendoscopic single-site surgery, and retroperitoneoscopic surgery; RLDN, including robotic-assisted retroperitoneoscopic surgery, and robotic-assisted laparoscopic surgery; and HALLDN, including hand-assisted retroperitoneoscopic surgery and hand-assisted laparoscopic surgery; (3) comparative studies evaluating 2 or more surgical techniques (OLDN, LLDN, RALLDN and/or HALLDN); and (4) studies that had at least 1 of the outcomes of interest of our study (see below).
EXCLUSION CRITERIA:
Articles were not included if they met the following criteria: (1) studies that did not meet the inclusion criteria; (2) children and patients undergoing nephrectomy for cancer or a benign kidney disease; and (3) a publication that was an abstract, case report, review, editorial, or letter, or that reported incomplete data, duplicate data, or experiments on non-human animals.
OUTCOMES OF INTEREST:
The outcomes of the study include the side of nephrectomy (right or left kidney), body mass index (BMI), warm ischemia time (WIT), estimated blood loss (EBL), operation time, intraoperative and postoperative donor complications, visual analogue scale (VAS), morphine intake on day 1, length of hospital stay, delayed graft function (DGF), acute rejection (AR), and 1-year graft survival.
STATISTICAL ANALYSIS:
For each outcome of interest, a network plot was drawn using STATA 14.0 software. The consistency test of all the surgical techniques was performed to assess their interest outcomes. In the network plot, nodes represent surgical techniques and connecting lines represent the evidence of direct comparison between the 2 groups of surgical techniques. The area of the nodes represents the cumulative number of enrolled patients for each intervention, and the width of the lines is proportional to the number of trials that have compared paired surgical treatments. In the consistency analysis,
We used a Bayesian approach utilizing the “GEMTC” package to analyze data by a random-effects or fixed-effects model using the Markov chain Monte Carlo chain. Three parallel chains were simultaneously run with different initial values. A total of 20 000 interactions were performed for each chain, and the first 5000 were used for a “burn-in” cycle to eliminate the impact of initial values. For binary data, odd ratios (OR) with 95% credible intervals (CrI) were estimated for the comparisons. When the 95% CrI of OR did not contain the value 1, it was regarded as indicating a statistically significant difference between 2 groups. For continuous outcomes, the MD with 95% CrI was calculated. If the 95% CrI of MD did not contain the value 0, it was considered to be significantly different. The ranking probability of each type of surgery technique for each outcome of interest was calculated. Cumulative ranking was estimated based on the surface under the cumulative ranking curve (SUCRA) to evaluate the ranking probabilities of each type of surgery technique. The results of ranking probabilities are shown in Table 1.
Results
LITERATURE SEARCH AND STUDY CHARACTERISTICS:
A total of 1545 publications were retrieved using the aforementioned search criteria. An additional 17 records were obtained from other sources, including the reference lists of the retrieved articles. After removing redundant ones, 1031 publications were further reviewed. Extensive screening identified 47 publications, as shown in Figure 1 [11–57]. Hamidi 2009 [13] and Oyen 2005 [14], Nicholson 2010 [16] and Nicholson 2011 [17], Simforoosh 2005 [18] and Simforoosh 2012 [19], and Waller 2001 [20] and Waller 2002 [21] reported the same donor datasets, so we combined their outcomes of interest into 4 studies. Finally, 43 studies were included. Among these included studies, 31 were non-RCTs (25 retrospective and 6 prospective) and 12 were RCTs.
The characteristics of the included studies are summarized in Table 2, including the surgical techniques compared and patient demographics in individual studies. A total of 6772 patients were included in the selected studies. Thirty-four studies were two-arm trials and 9 were three-arm trials involving different types of surgeries for kidney transplantation. Of all patients, 2003 were treated with OLDN, 2710 with LLDN, 1809 with HALLDN, and 250 with RALLDN. A wide geographic distribution of patients was seen, with patients mainly from North America, South America, Europe, Oceania, and Asia. The network plots of comparisons between different surgical approaches with their corresponding sample sizes are shown in Figure 2, in which the cumulative number of enrolled patients for each intervention and the number of trials comparing each pair of treatments are indicated by the node area and the line width, respectively.
RISK OF BIAS IN INCLUDED STUDIES:
The risk of bias in the included studies was evaluated by use of the Cochrane Collaboration’s risk of bias assessment tool. When evaluating selection bias, we found that 10 of the included studies reported sufficient details to evaluate sequence generation and allocation concealment. Of the included studies, only 2 mentioned the use of blinding of participants and personnel and/or blinding of outcome assessment, but none of them reported incomplete outcome data or no selective outcome reporting; therefore, the attrition bias of the included studies was evaluated as low risk. Other sources of bias were identified as unclear risk in all articles, because there were too few available details to make a decision (Figure 3).
CONSISTENCY ANALYSIS:
We used node-splitting models and heat plots for direct and indirect comparisons to evaluate the consistency of outcomes of interest. The data showed that all P values were >0.05, indicating that there was no evidence of a significant inconsistency in the network (Figure 4).
OUTCOMES OF INTEREST:
The results of all pairwise comparisons of the different surgical techniques for the outcomes of interest are displayed in Table 3. Table 1 shows the results of the ranking probabilities for all interventions based on the SUCRA value for each outcome of interest.
DONOR DEMOGRAPHICS:
There was no significant difference in BMI among the OLDN, HALLDN, LLDN, and RLDN groups. Compared with the OLDN group, the other 3 groups preferred to harvest the left kidney. When we chose the right kidney as a donor, OLDN was the first choice for treatment compared to HALLDN, LLDN, and RLDN; however, there was no significant difference in selecting the side of nephrectomy among the HALLDN, LLDN, and RLDN groups.
DONOR OPERATIVE PARAMETERS:
EBL was significantly lower in the HALLDN, LLDN, and RLDN groups when compared to the OLDN group. OLDN ranked first for high probability of EBL. However, operative time and WIT were significantly shorter in the OLDN group compared to the other 3 groups. Compared to the LLDN and RLDN groups, the HALLDN group had a shorter operative time and WIT. HALLDN and OLDN ranked third and fourth, respectively, with high probabilities for operation time and WIT. The rate of intraoperative complications was higher in the RLDN group, but there was no significant difference in the postoperative complications. RLDN ranked first for donor intraoperative complications. We performed a subgroup analysis of intraoperative and postoperative complications according to the Clavien scale. In terms of the donor intraoperative complication, most of the reported data were Clavien scale III and IV. The rate of intraoperative complications (III–IV) was higher in the RLDN group, but there was no significant difference in the postoperative complications (I–II and III–IV). The RLDN group had a significantly lower VAS on day 1 when compared to the OLDN, HALLDN, and LLDN groups. The OLDN group required more morphine intake than the LLDN group. The length of hospital stay was significantly longer in the OLDN group. OLDN ranked first for probabilities for the VAS on day 1, morphine intake, and duration of hospital stay.
RECIPIENT PARAMETERS:
There were no significant differences in 1-year graft survival and DGF among the 4 surgical methods. The OLDN group had a significantly higher AR when compared to the LLDN and HALLDN groups; it ranked first for AR with a high probability. There was no significant difference in AR between the HALLDN and LLDN groups.
Discussion
LDN is a unique surgical procedure because healthy people take surgical risks for the benefits of patients. It is very important to choose the best surgical approach to obtain a living donor kidney. There are 2 major issues to keep in mind: (1) donor mortality and morbidity risks could be minimized by carefully selecting safe surgical techniques, which are negatively associated with the surgical skills and experience of the transplant center and operating surgeons; and (2) maintain the optimal function of the donor kidney to ensure the recipient gets the best results after kidney transplantation [58].
In this meta-analysis, we found that there was no significant difference in BMI among the donors enrolled for LDN with different surgical techniques. The left kidney was preferable in LDN, especially when using the RLDN approach. Left kidney donor nephrectomy is technically easier to perform due to a longer renal vein, which provides implantation advantages. In addition, using the transperitoneal approach in right kidney RLDN is more difficult because the presence of the liver complicates the dissection [59,60].
WIT and operation time in the LLDN and RLDN groups were longer than those in the HALLDN and OLDN groups, but there were no significant differences in WIT and operation time between the RLDN and LLDN groups. This is most likely due to the rapid extraction of the kidney through the hand port after vascular ligation in the manual-assisted approach, while the LLDN and RLDN approaches require a bag removal and incision [61]. Our data show that EBL was significantly lower in the HALLDN, LLDN, and RLDN groups compared to the OLDN group, which may be due to the use of finer instruments, magnified view, and multi-angle vision in the laparoscopic surgery [62]. The included studies used the VAS to assess pain on the first day after surgery. We found that the RLDN group had the lowest VAS score among all 4 interventions. Moreover, the OLDN group required more morphine intake than the LLDN group, indicating that RLDN and LLDN reduce donor postoperative pain. RLDN ranked last in VAS and morphine intake. One possible reason for the reduced pain in the robotic surgery is the robotic arms, which rotate around the port site and move at a fixed remote center; therefore, there is less leverage and pressure around the port, resulting in less trauma to the abdominal tissue around the port [24]. Accordingly, relief of pain leads to an earlier recovery. Therefore, we further analyzed the length of hospital stay and found that the OLDN group had a significantly longer hospital stay than the other 3 groups. However, the rate of intraoperative complications was higher in the RLDN group, but there was no significant difference in postoperative complications among all groups. Intraoperative complications were mainly attributable to vascular injury in addition to other causes, such as instrument failure, improper use, and visceral injury [25–27]. The presence of intraoperative complications reflects a less experienced surgeon but is expected to diminish as the surgeon gains experience and develops surgical expertise. Using cadavers for training can quickly improve the surgeon’s surgical skills, and development of the surgical training model may also help improve the RLDN learning curve; for example, by using robot-assisted partial nephrectomy training models. In terms of recipient outcomes, the increase in WIT and operation time does not translate into the incidence of DGF or affect 1-year graft survival because the recipient DGF and graft survival were not different among the 4 surgical methods. Nevertheless, we found that the OLDN technique caused significantly higher AR compared to LLDN and HALLDN, but there was no significant difference in AR between the HALLDN and LLDN groups. We believe that this is related to a central bias effect, because we did not include information on immunosuppression in the meta-analysis, and they may have used a more active approach to immunosuppression than other centers.
To the best of our knowledge, this study is the first to use the NMA method to comparatively assess 4 surgical approaches in LDN. By including direct and indirect evidence, NMA improved the estimation precision of effects of treatments, and increased the analytical power when compared to a pairwise meta-analysis, which uses only direct evidence. However, several limitations should be noted in this study. First, a fundamental limitation of our study is the small number of randomized controlled trials eligible for inclusion, as well as the limited number of participants in each study. Second, the retrospective design inherently introduces a selection bias in the study population. Third, the immunosuppression factor was not included in this meta-analysis.
Conclusions
In summary, this NMA study found that there was no significant difference in donor postoperative complications, recipient DGF, and graft survival among the 4 surgical techniques. OLDN and HALLDN reduce WIT and operation time, but OLDN increases EBL and AR. RLDN and LLDN reduces the length of hospital stay, morphine intake, and VAS, and thus accelerate recovery. However, RLDN leads to increased intraoperative complications due to the learning curve. We performed a subgroup analysis of intraoperative complications according to the Clavien scale. The rate of intraoperative complications (III–IV) was higher in the RLDN group. Most intraoperative complications are due to uncontrolled bleeding due to intraoperative vascular injury or splenic tear, and surgeons can significantly reduce the incidence of such complications by training in the model. The expected benefits of individual patients should be considered when selecting a surgical method in LDN. Given the limitations of the included studies, more high-quality direct evidence and comparisons of multiple interventions are needed to support our findings.
Figures
 Figure 1. Flow diagram of study selection for inclusion.
Figure 1. Flow diagram of study selection for inclusion.  Figure 2. Network plots for different outcomes of interest of different surgical approaches. The area of the nodes represents the cumulative number of enrolled patients for each intervention and the width of the lines represents the number of trials comparing each pair of treatments. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. (1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival.
Figure 2. Network plots for different outcomes of interest of different surgical approaches. The area of the nodes represents the cumulative number of enrolled patients for each intervention and the width of the lines represents the number of trials comparing each pair of treatments. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. (1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival. 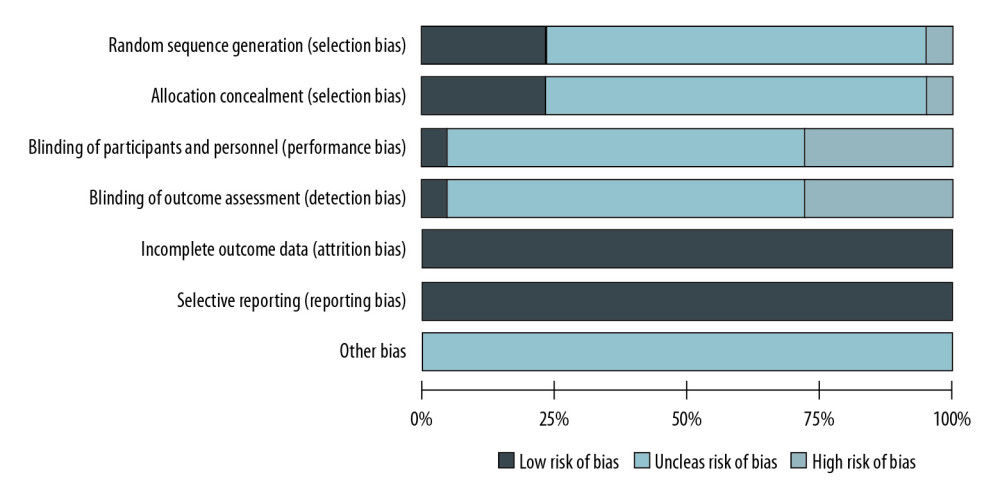 Figure 3. Assessment of study quality using the Cochrane Collaboration’s risk of bias tool.
Figure 3. Assessment of study quality using the Cochrane Collaboration’s risk of bias tool.  Figure 4. The results of consistency analysis by node-splitting approach and the heat plots between the direct and indirect evidence comparisons among all outcomes of different surgical approaches. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. 1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival.
Figure 4. The results of consistency analysis by node-splitting approach and the heat plots between the direct and indirect evidence comparisons among all outcomes of different surgical approaches. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. 1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival. References
1. Lee LY, Pham TA, Melcher ML, Living kidney donation: Strategies to increase the donor pool: Surg Clin North Am, 2019; 99(1); 37-47
2. LaPointe Rudow D, Warburton KM, Selection and postoperative care of the living donor: Med Clin North Am, 2016; 100(3); 599-611
3. Murray G, Holden R, Transplantation of kidneys, experimentally and in human cases: Am J Surg, 1954; 87(4); 508-15
4. Ratner LE, Ciseck LJ, Moore RG, Laparoscopic live donor nephrectomy: Transplantation, 1995; 60(9); 1047-49
5. Wilson CH, Sanni A, Rix DA, Laparoscopic versus open nephrectomy for live kidney donors: Cochrane Database Syst Rev, 2011(11); CD006124
6. Wolf JS, Tchetgen MB, Merion RM, Hand-assisted laparoscopic live donor nephrectomy: Urology, 1998; 52(5); 885-87
7. Özdemir-van Brunschot DM, Koning GG, van Laarhoven KC, A comparison of technique modifications in laparoscopic donor nephrectomy: A systematic review and meta-analysis: PLoS One, 2015; 10(3); e0121131
8. Horgan S, Vanuno D, Benedetti E, Early experience with robotically assisted laparoscopic donor nephrectomy: Surg Laparosc Endosc Percutan Tech, 2002; 12(1); 64-70
9. Giacomoni A, Concone G, Di Sandro S, The meaning of surgeon’s comfort in robotic surgery: Am J Surg, 2014; 208(5); 871-72
10. Hozo SP, Djulbegovic B, Hozo I, Estimating the mean and variance from the median, range, and the size of a sample: BMC Med Res Methodol, 2005; 5; 13
11. Brook NR, Harper SJ, Bagul A, Laparoscopic donor nephrectomy yields kidneys with structure and function equivalent to those retrieved by open surgery: Transplant Proc, 2005; 37(2); 625-26
12. Brook NR, Gibbons N, Nicol DL, Open and laparoscopic donor nephrectomy: Activity and outcomes from all Australasian transplant centers: Transplantation, 2010; 89(12); 1482-88
13. Hamidi V, Andersen MH, Oyen O, Cost effectiveness of open versus laparoscopic living-donor nephrectomy: Transplantation, 2009; 87(6); 831-38
14. Øyen O, Andersen M, Mathisen L, Laparoscopic versus open living-donor nephrectomy: Experiences from a prospective, randomized, single-center study focusing on donor safety: Transplantation, 2005; 79(9); 1236-40
15. Kok NF, Lind MY, Hansson BM, Comparison of laparoscopic and mini incision open donor nephrectomy: Single blind, randomised controlled clinical trial: BMJ, 2006; 333(7561); 221
16. Nicholson ML, Kaushik M, Lewis GR, Randomized clinical trial of laparoscopic versus open donor nephrectomy: Br J Surg, 2010; 97(1); 21-28
17. Nicholson ML, Elwell R, Kaushik M, Health-related quality of life after living donor nephrectomy: A randomized controlled trial of laparoscopic versus open nephrectomy: Transplantation, 2011; 91(4); 457-61
18. Simforoosh N, Basiri A, Tabibi A, Comparison of laparoscopic and open donor nephrectomy: A randomized controlled trial: BJU Int, 2005; 95(6); 851-55
19. Simforoosh N, Basiri A, Shakhssalim N, Long-term graft function in a randomized clinical trial comparing laparoscopic versus open donor nephrectomy: Exp Clin Transplant, 2012; 10(5); 428-32
20. Waller JR, Veitch PS, Nicholson ML, Laparoscopic live donor nephrectomy: A comparison with the open operation: Transplant Proc, 2001; 33(7–8); 3787-88
21. Waller JR, Hiley AL, Mullin EJ, Living kidney donation: A comparison of laparoscopic and conventional open operations: Postgrad Med J, 2002; 78(917); 153-57
22. Wolf JS, Merion RM, Leichtman AB, Randomized controlled trial of hand-assisted laparoscopic versus open surgical live donor nephrectomy: Transplantation, 2001; 72(2); 284-90
23. Yadav K, Aggarwal S, Guleria S, Comparative study of laparoscopic and mini-incision open donor nephrectomy: Have we heard the last word in the debate?: Clin Transplant, 2016; 30(3); 328-34
24. Bhattu AS, Ganpule A, Sabnis RB: J Endourol, 2015; 29(12); 1334-40
25. Cohen AJ, Williams DS, Bohorquez H, Robotic-assisted laparoscopic donor nephrectomy: Decreasing length of stay: Ochsner J, 2015; 15(1); 19-24
26. Geffner S, Klaassen Z, Tichauer M, Robotic-assisted laparoscopic donor nephrectomies: Early experience and review of the literature: J Robot Surg, 2011; 5(2); 115-20
27. Janki S, Klop KWJ, Hagen SM, Robotic surgery rapidly and successfully implemented in a high-volume laparoscopic center on living kidney donation: Int J Med Robot, 2017; 13(2); rcs.1743
28. Liu XS, Narins HW, Maley WR, Robotic-assistance does not enhance standard laparoscopic technique for right-sided donor nephrectomy: JSLS, 2012; 16(2); 202-7
29. Luke PP, Aquil S, Alharbi B: Can Urol Assoc J, 2018; 12(11); E440-46
30. Yang A, Barman N, Chin E: J Robot Surg, 2018; 12(2); 343-50
31. Bargman V, Sundaram CP, Bernie J, Randomized trial of laparoscopic donor nephrectomy with and without hand assistance: J Endourol, 2006; 20(10); 717-22
32. Branco AW, Kondo W, Branco Filho AJ, A comparison of hand-assisted and pure laparoscopic techniques in live donor nephrectomy: Clinics (Sao Paulo), 2008; 63(6); 795-800
33. Buell JF, Abreu SC, Hanaway MJ, Right donor nephrectomy: A comparison of hand-assisted transperitoneal and retroperitoneal laparoscopic approaches: Transplantation, 2004; 77(4); 521-25
34. Choi SW, Kim KS, Kim S, Hand-assisted and pure laparoscopic living donor nephrectomy: A matched-cohort comparison over 10 yr at a single institute: Clin Transplant, 2014; 28(11); 1287-93
35. Dols LF, Kok NF, d’Ancona FC, Randomized controlled trial comparing hand-assisted retroperitoneoscopic versus standard laparoscopic donor nephrectomy: Transplantation, 2014; 97(2); 161-67
36. El-Galley R, Hood N, Young CJ, Donor nephrectomy: A comparison of techniques and results of open, hand assisted and full laparoscopic nephrectomy: J Urol, 2004; 171(1); 40-43
37. Gershbein AB, Fuchs GJ, Hand-assisted and conventional laparoscopic live donor nephrectomy: A comparison of two contemporary techniques: J Endourol, 2002; 16(7); 509-13
38. Gjertsen H, Sandberg AK, Wadström J, Introduction of hand-assisted retroperitoneoscopic living donor nephrectomy at Karolinska University Hospital Huddinge: Transplant Proc, 2006; 38(8); 2644-45
39. Greco F, Hamza A, Wagner S, Hand-assisted laparoscopic living-donor nephrectomy versus open surgery: Evaluation of surgical trauma and late graft function in 82 patients: Transplant Proc, 2009; 41(10); 4039-43
40. Hirose T, Hotta K, Iwami D, Safety and efficacy of retroperitoneoscopic living donor nephrectomy: Comparison of early complication, donor and recipient outcome with hand-assisted laparoscopic living donor nephrectomy: J Endourol, 2018; 32(12); 1120-24
41. Hofker HS, Nijboer WN, Niesing J, A randomized clinical trial of living donor nephrectomy: A plea for a differentiated appraisal of mini-open muscle splitting incision and hand-assisted laparoscopic donor nephrectomy: Transpl Int, 2012; 25(9); 976-86
42. Klop KW, Kok NF, Dols LF, Can right-sided hand-assisted retroperitoneoscopic donor nephrectomy be advocated above standard laparoscopic donor nephrectomy: A randomized pilot study: Transpl Int, 2014; 27(2); 162-69
43. Kocak B, Baker TB, Koffron AJ, Laparoscopic living donor nephrectomy: A single-center sequential experience comparing hand-assisted versus standard technique: Urology, 2007; 70(6); 1060-63
44. Lai IR, Yang CY, Yeh CC, Hand-assisted versus total laparoscopic live donor nephrectomy: Comparison and technique evolution at a single center in Taiwan: Clin Transplant, 2010; 24(5); E182-87
45. Lucas SM, Liaw A, Mhapsekar R, Comparison of donor, and early and late recipient outcomes following hand assisted and laparoscopic donor nephrectomy: J Urol, 2013; 189(2); 618-22
46. Mateo RB, Sher L, Jabbour N, Comparison of outcomes in noncomplicated and in higher-risk donors after standard versus hand-assisted laparoscopic nephrectomy: Am Surg, 2003; 69(9); 771-78
47. Minnee RC, Bemelman F, Kox C, Comparison of hand-assisted laparoscopic and open donor nephrectomy in living donors: Int J Urol, 2008; 15(3); 206-9
48. Mjøen G, Holdaas H, Pfeffer P, Minimally invasive living donor nephrectomy -introduction of hand-assistance: Transpl Int, 2010; 23(10); 1008-14
49. Percegona LS, Bignelli AT, Adamy A, Hand-assisted laparoscopic donor nephrectomy: Comparison to pure laparoscopic donor nephrectomy: Transplant Proc, 2008; 40(3); 687-88
50. Ruiz-Deya G, Cheng S, Palmer E, Open donor, laparoscopic donor and hand assisted laparoscopic donor nephrectomy: A comparison of outcomes: J Urol, 2001; 166(4); 1270-73
51. Ruszat R, Sulser T, Dickenmann M, Retroperitoneoscopic donor nephrectomy: Donor outcome and complication rate in comparison with three different techniques: World J Urol, 2006; 24(1); 113-17
52. Salazar A, Pelletier R, Yilmaz S, Use of a minimally invasive donor nephrectomy program to select technique for live donor nephrectomy: Am J Surg, 2005; 189(5); 558-62
53. Stifelman MD, Hull D, Sosa RE, Hand assisted laparoscopic donor nephrectomy: A comparison with the open approach: J Urol, 2001; 166(2); 444-48
54. Sundqvist P, Feuk U, Häggman M, Hand-assisted retroperitoneoscopic live donor nephrectomy in comparison to open and laparoscopic procedures: A prospective study on donor morbidity and kidney function: Transplantation, 2004; 78(1); 147-53
55. Ungbhakorn P, Kongchareonsombat W, Leenanupan C, Comparative outcomes of open nephrectomy, hand-assisted laparoscopic nephrectomy, and full laparoscopic nephrectomy for living donors: Transplant Proc, 2012; 44(1); 22-25
56. Velidedeoglu E, Williams N, Brayman KL, Comparison of open, laparoscopic, and hand-assisted approaches to live-donor nephrectomy: Transplantation, 2002; 74(2); 169-72
57. Wadström J, Lindström P, Engström BM, Hand-assisted retroperitoneoscopic living donor nephrectomy superior to laparoscopic nephrectomy: Transplant Proc, 2003; 35(2); 782-83
58. Elmaraezy A, Abushouk AI, Kamel M, Should hand-assisted retroperitoneoscopic nephrectomy replace the standard laparoscopic technique for living donor nephrectomy? A meta-analysis: Int J Surg, 2017; 40; 83-90
59. Rodríguez Faba O, Boissier R, Budde K, European Association of Urology guidelines on renal transplantation: Update 2018: Eur Urol Focus, 2018; 4(2); 208-15
60. Dols LF, Kok NF, Terkivatan T, Optimizing left-sided live kidney donation: hand-assisted retroperitoneoscopic as alternative to standard laparoscopic donor nephrectomy: Transpl Int, 2010; 23(4); 358-63
61. Broe MP, Galvin R, Keenan LG, Laparoscopic and hand-assisted laparoscopic donor nephrectomy: A systematic review and meta-analysis: Arab J Urol, 2018; 16(3); 322-34
62. Horgan S, Benedetti E, Moser F, Robotically assisted donor nephrectomy for kidney transplantation: Am J Surg, 2004; 188(4A Suppl); 45S-51S
Figures
 Figure 1. Flow diagram of study selection for inclusion.
Figure 1. Flow diagram of study selection for inclusion. Figure 2. Network plots for different outcomes of interest of different surgical approaches. The area of the nodes represents the cumulative number of enrolled patients for each intervention and the width of the lines represents the number of trials comparing each pair of treatments. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. (1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival.
Figure 2. Network plots for different outcomes of interest of different surgical approaches. The area of the nodes represents the cumulative number of enrolled patients for each intervention and the width of the lines represents the number of trials comparing each pair of treatments. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. (1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival. Figure 3. Assessment of study quality using the Cochrane Collaboration’s risk of bias tool.
Figure 3. Assessment of study quality using the Cochrane Collaboration’s risk of bias tool. Figure 4. The results of consistency analysis by node-splitting approach and the heat plots between the direct and indirect evidence comparisons among all outcomes of different surgical approaches. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. 1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival.
Figure 4. The results of consistency analysis by node-splitting approach and the heat plots between the direct and indirect evidence comparisons among all outcomes of different surgical approaches. OLDN, open living donor nephrectomy; LLDN, laparoscopic living donor nephrectomy; HALLDN, hand-assisted laparoscopic living donor nephrectomy; RLDN, robot-assisted laparoscopic living donor nephrectomy. 1) Right nephrectomy; (2) left nephrectomy; (3) body mass index (BMI); (4) warm ischemia time; (5) estimated blood loss; (6) operation time; (7) donor intraoperative complications; (8) donor postoperative complications; (9) visual analogue scale (VAS) on day 1; (10) Morphine intake on day 1; (11) donor length of hospital stay; (12) delayed graft function; (13) acute rejection; and (14) 1-year graft survival. Tables
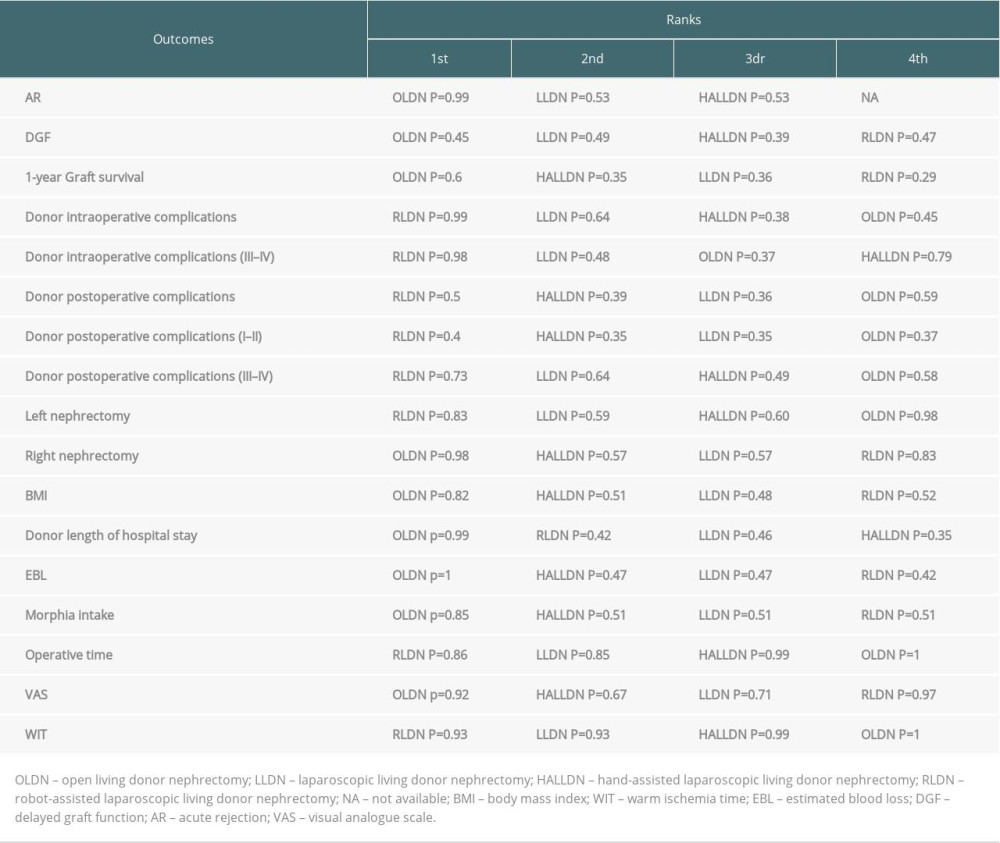 Table 1. The results of the ranking probabilities for all interventions.
Table 1. The results of the ranking probabilities for all interventions.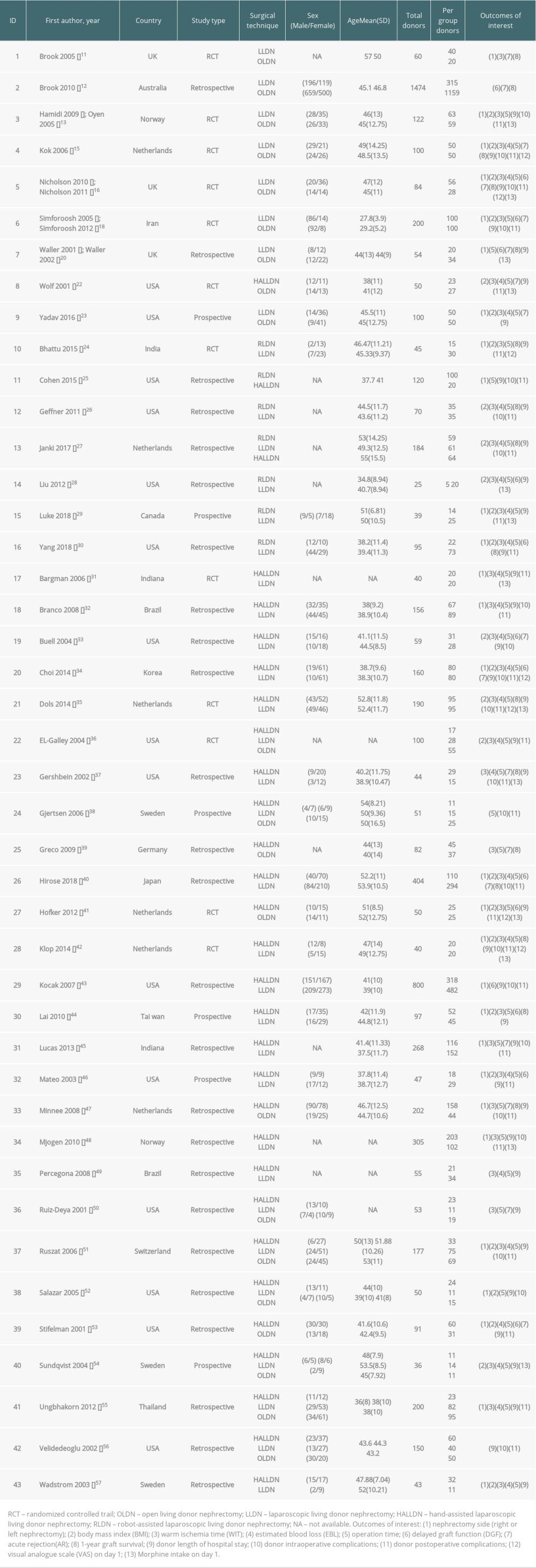 Table 2. Characteristics of studies included in the meta-analysis.
Table 2. Characteristics of studies included in the meta-analysis.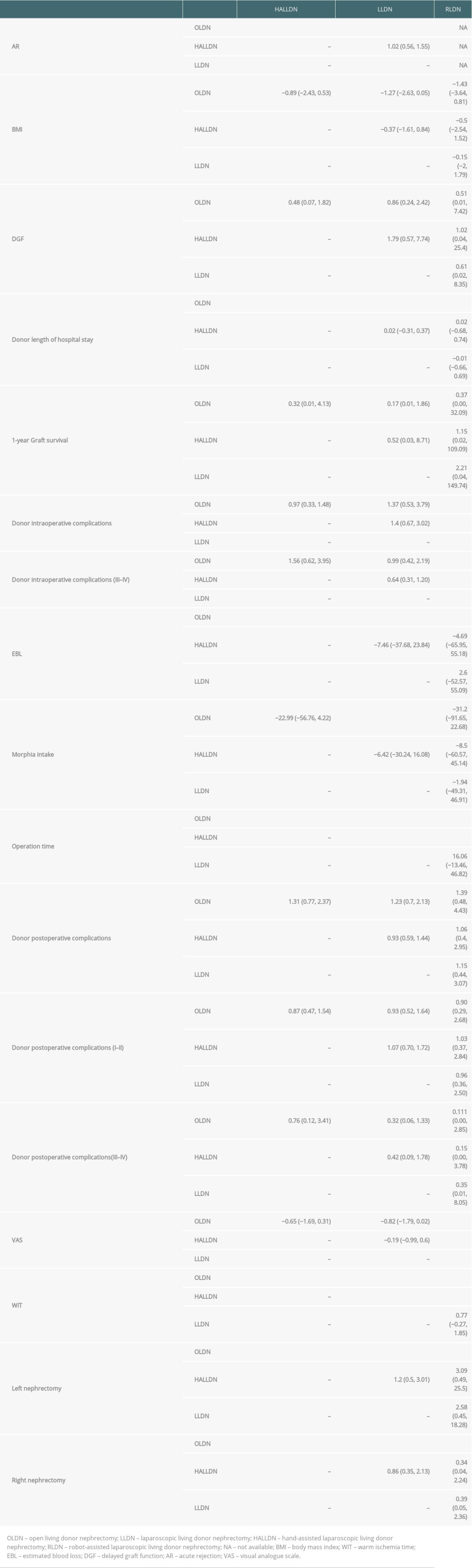 Table 3. The results of all pairwise comparisons of the different surgical techniques for the outcomes of interest.
Table 3. The results of all pairwise comparisons of the different surgical techniques for the outcomes of interest. Table 1. The results of the ranking probabilities for all interventions.
Table 1. The results of the ranking probabilities for all interventions. Table 2. Characteristics of studies included in the meta-analysis.
Table 2. Characteristics of studies included in the meta-analysis. Table 3. The results of all pairwise comparisons of the different surgical techniques for the outcomes of interest.
Table 3. The results of all pairwise comparisons of the different surgical techniques for the outcomes of interest. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








