16 August 2022: Original Paper
Evaluation of Deceased Donor Kidney Transplantation in the Eurotransplant Senior Program in Comparison to Standard Allocation
Nelly Delali Eklou1BCDE, Bernd M. Jänigen2E, Przemyslaw Pisarski2E, Gerd Walz1E, Johanna Schneider1ABCDEF*DOI: 10.12659/AOT.936514
Ann Transplant 2022; 27:e936514
Abstract
BACKGROUND: The organ shortage and long waiting times have dramatically increased the age of potential kidney transplant recipients. The Eurotransplant Senior Program (ESP) was initiated to allocate kidneys from deceased donors aged ≥65 years to recipients with a comparable age independent of pre-transplant human leucocyte antigen (HLA) matching; however, parameters affecting the long-term benefits of this strategy remain poorly defined.
MATERIAL AND METHODS: We retrospectively evaluated outcome and risk factors for mortality in kidney recipients aged ≥65 years that were transplanted according to the ESP protocol relative to patients aged >50 years transplanted according to the Eurotransplant kidney allocation system (ETKAS) criteria at the University Freiburg Medical Center, Germany, between 2008 and 2018.
RESULTS: Graft survival, graft function, the maintenance immunosuppressive therapy, and the incidence of rejections and infections did not differ between groups. Infectious diseases were the main cause of death in both groups; however, infection-associated mortality was more than double in the ESP group, and 5-year patient survival was 61.4% in the ESP group compared to 83.2% in the ETKAS group. Multivariate analysis identified age, the number of HLA mismatches, and the CMV serostatus with a seropositive donor and negative recipient as the main risk factors for mortality.
CONCLUSIONS: A comparable immunosuppressive regimen used in ESP and ETKAS patients was associated with similar rejection rates and infectious disease complications, and infections were the most common cause of death in both groups. CMV-negative patients receiving an organ from a CMV-positive donor and patients with a high number of HLA mismatches require close follow-up to reduce mortality.
Keywords: cold ischemia, Graft Survival, Kidney Transplantation, Mortality, Cytomegalovirus Infections, Graft Rejection, Humans, Immunosuppressive Agents, Kidney, Retrospective Studies, Tissue Donors
Background
Due to demographic changes, the age of dialysis patients constantly increases. Meanwhile, more than 50% of patients with end-stage kidney disease (ESKD) are ≥65 years of age [1], and the percentage of patients >50 years awaiting kidney transplantation continuously increases. As a result of the organ shortage, waiting time increases dramatically and only a subgroup of patients can receive a kidney transplant. Since life expectancy of recipients decreases with age, a significant number of elderly patients die with a functioning graft [2]. It is therefore critical to identify ESKD patients that might benefit from organ transplantation in an aging high-risk population, and to implement allocation strategies to match the life expectancy of organs and recipients [3]. Therefore, the Eurotransplant kidney allocation program for patients and donors ≥65 years of age, the Eurotransplant Senior Program (ESP), was implemented in 1999. This program offers patients aged ≥65 years to either be listed in the Eurotransplant kidney allocation system (ETKAS), the regular kidney allocation program for adults ≥18 years in participating Eurotransplant countries, or in the ESP. One objective of the ESP is the efficient usage of organs from elderly donors. The median age of deceased kidney donors is constantly increasing [4], and high donor age is associated with reduced graft survival [5–7]. Therefore, organs from elderly donors have been rejected more frequently for younger recipients. Since prolonged cold ischemia times increase the risk of graft loss [8], the ESP allocates organs locally based on blood group compatibility and waiting time without considering the donor human leucocyte antigen (HLA) typing as a mandatory criterion. In case donor HLA typing is available prior to ESP matching, unacceptable antigens are considered during the allocation process. Presensitized patients are also included in the ESP. The ESP reduced cold ischemia time compared to standard allocation in several studies [8,9], which was accompanied by a reduced incidence of delayed graft function (DGF) [8]. The incidence of acute rejections is greater in ESP patients [8,9], with more patients developing donor-specific antibodies and a higher incidence of T cell-mediated rejection [10].
As expected, long-term survival of transplanted patients ≥65 years of age is shorter relative to younger patients after kidney transplantation [11,12]. However, elderly patients still benefit from kidney transplantation, with improved life expectancy and quality of life compared to dialysis patients [13–17]. Survival of patients with transplanted kidneys from donors aged ≥80 years remains better compared to patients awaiting kidney transplantation [18]. However, outcome parameters and risk factors are currently incompletely understood, which we aimed to identify in ESP patients ≥65 years of age after kidney transplantation compared to younger patients aged ≥50 years that were transplanted according to the ETKAS criteria.
Material and Methods
STUDY DESIGN:
A retrospective analysis of all patients transplanted in the ESP between February 2008 and July 2018 at the University Freiburg Medical Center, Germany, was performed. A similar number of patients transplanted in the ETKAS in the same time period with a minimum age of 50 years served as a control group. Between January 2013 and January 2014, a study was conducted in which 29 patients of our cohort in the ESP were transplanted considering HLA-DR compatibility. This study was called the Eurotransplant Senior DR-compatible program (ESDP). Patients from the ESP and the ESDP were combined in the present study to simplify presentation. A total of 227 patients were analyzed: 116 patients in the ESP and 111 patients in the ETKAS. ETKAS patients transplanted in the same time period at <50 years of age were excluded. Patients with a combined pancreas-kidney transplantation were also excluded. The observation period was from February 2008 to August 2019. Baseline characteristics were age, sex, weight, body mass index, coexisting disorders, smoking history, immunosuppressive therapy and chemotherapy prior to transplantation, cause of ESKD, time on dialysis, the number of previous transplantations, cytomegalovirus (CMV) serostatus, cold and warm ischemia time, and number of panel-reactive antibodies (PRA) and HLA mismatches. The primary endpoint was death. The time point of death after transplantation, cause of death, and death with a functioning graft were analyzed. Secondary outcome parameters were graft function as assessed by serum creatinine levels, infections, cardiovascular events, cancer, bleeding events, new-onset diabetes after transplantation (NODAT), development of transplant renal artery stenosis, and hospitalization within the first year. Additional parameters analyzed for follow-up were immunosuppressive therapy, incidence of delayed graft function, rejections, and graft loss. In case of more than 1 infection, cardiovascular, or bleeding event, the number of patients were analyzed. The study was approved by the Ethics Committee of the University Freiburg Medical Center, Germany (protocol number 505/18).
DATA COLLECTION AND DEFINITIONS:
Clinical data and follow-up data were collected from historical records and data transmitted to our center. DGF was defined as the need for at least 1 dialysis treatment during the first postoperative week. Primary nonfunction (PNF) was defined as the dependence on dialysis for 3 months after transplantation. Acute rejections were confirmed by biopsies and classified according to the 2017 Banff Criteria [19]. Rejections were divided into antibody-mediated rejection, T cell-mediated rejection, the combination of both, and borderline rejections. Renal function was assessed by serum creatinine levels.
STATISTICAL ANALYSIS:
SPSS Statistics 26® software (IBM Corp., Armonk, NY) was used for statistical analysis. Continuous variables are expressed as mean±standard error of the mean (SEM). Group comparison was performed with the
Results
PATIENT CHARACTERISTICS:
Recipient patients in the ESP group were older at the time of transplantation (69.5 years vs 58.1 years, P<0.001, Table 1). Sex, weight, and body mass index were evenly distributed. Patients in the ESP group had more coexisting disorders, mainly due to an increased incidence of diabetes (33.9% vs 18.9%, P=0.016, Table 1) and cardiac diseases (78.4% vs 41.4%, P<0.001, Table 1). The main causes of ESKD were autoimmune disorders and chronic glomerulonephritis in both groups. The number of patients with ESKD resulting from diabetes and genetic disorders was evenly distributed. More patients in the ESP group had ESKD due to hypertensive disorders (10.3% vs 0.9%, P=0.003, Table 1). Patients in the ETKAS group had a significantly longer time on dialysis prior to transplantation (93.2 vs 66.3 mean months, P<0.001, Table 1). Cold ischemia time was significantly shorter in the ESP group (9.3 vs 11.8 mean hours, P<0.001, Table 1). Patients in the ETKAS group had more previous transplantations (second transplantation: 15.3% vs 6.0% in ESP, P=0.030, Table 1). Significantly more patients developed ESKD due to kidney transplant failure or the use of a calcineurin inhibitor due to organ transplantation (ETKAS: 16.2%, ESP: 6.9%, P=0.036, Table 1). The number of both, the current and highest PRA, was significantly higher in the ETKAS group. Patients in the ESP group had a mean HLA mismatch of 4.0 relative to 2.1 in the ETKAS group (P<0.001, Table 1).
IMMUNOSUPPRESSION AND GRAFT SURVIVAL:
Since patients in the ETKAS group had a higher degree of immunization, more patients received antithymocyte globulin as induction therapy (19.8% vs 9.5%, p=0.037, Table 2) and the fraction of patients with basiliximab was lower (64.9% vs 81.0%, p=0.007, Table 2). Most patients were treated with tacrolimus, prednisone, and mycophenolate for maintenance therapy, with no difference between groups. Triple therapy was used in 96.3% of ESP patients and 96.2% of ETKAS patients (P=1.000, Table 2). The total number of rejections and the type of rejection were similar between groups (Table 2 and Figure 1A), with T cell-mediated rejection as the main cause. The Kaplan-Meier plot illustrating survival free from rejection showed no difference between groups (P=0.897, Figure 1B).
Survival free from graft loss was not different between groups (P=0.171, Figure 2A). After 1 year, ETKAS patients had better kidney graft function as determined by serum creatinine levels, but not directly after transplantation and after 2 and 5 years after transplantation (Figure 2B). The number of patients with DGF was similar between groups (ESP 15.5% vs ETKAS 15.3%, P=1.000, Table 2). No difference existed for the number of patients with graft loss within the first year (ESP 9.5%, ETKAS 7.2%, P=0.635, Table 2), with a similar distribution of causes.
MORTALITY AND CAUSE OF DEATH:
The overall mortality was higher in the ESP group (44.8% vs 13.5%, P<0.001, Table 3 and Figure 3A) during the median follow-up period of 43 months. In the ESP group, patient survival after 1 year was 86.4% and 61.2% after 5 years compared to ETKAS group with 92.6% after 1 and 82.4% after 5 years.
The main cause of death was infectious diseases in both groups (Figure 3B). Death due to infectious diseases occurred more than twice as often in the ESP group than in the ETKAS group (17.2% vs 8.1%, P=0.047, Table 3 and Figure 3B). Although more patients died from infectious diseases in the ESP group, the number of patients with infectious diseases was evenly distributed (ESP 83.6% vs ETKAS 77.5%, P=0.314, Table 4). Kaplan-Meier analysis revealed that infections trended to occur earlier in the ESP group (P=0.127, Figure 4A). The pathogens causing bacterial, viral, and fungal infections were evenly distributed (Figure 4B). The incidence of urinary tract infections, pneumonia, and infections due to cytomegalovirus was higher in the ESP group (Figure 4C, 4D).
Patients in the ESP group had significantly more cardiovascular events during the follow-up period (20.7% vs 9.9%, P=0.028, Table 4), mainly due to a higher incidence of acute coronary syndromes (6.9% vs 0.9%, P=0.036, Table 4). Significantly more patients in the ESP group were hospitalized within the first year after transplantation (1 hospitalization: ESP 20.7% vs ETKAS 9.0%, P=0.016, Table 4). We observed a trend toward more bleeding events in the ESP group (12.1% vs 5.4%, P=0.101, Table 4). The number of patients with transplant artery stenosis, NODAT, or cancer did not differ between groups (Table 4). Skin tumors were the main cause of tumor diseases.
RISK FACTORS ASSOCIATED WITH MORTALITY:
Risk factors for mortality in both groups were age (odds ratio 1.11, P<0.001, Table 5), ESKD due to diabetic nephropathy (odds ratio 2.51, P=0.027), cardiac disease (odds ratio 3.43, P<0.001), the number of HLA mismatches (odds ratio 1.44, P<0.001), the number of previous diseases (odds ratio 1.41, P=0.003, Table 5), and the CMV high-risk serostatus with a CMV-positive donor and a CMV-negative recipient (D+/R−, odds ratio 3.27, P<0.001). A CMV serostatus with a seropositive donor and seropositive recipient (D+/R+) tended to be a protective factor, but this was not statistically significant (odds ratio 0.55, P=0.070, Table 5). Although not statistically significant, male sex (odds ratio 1.82, P=0.077, Table 5) and diabetes (odds ratio 1.78, P=0.071), trended to increase mortality. Multivariate analysis revealed age, number of HLA mismatches, and CMV high-risk serostatus with a CMV-positive donor and a negative recipient as the most robust risk factors for mortality (Table 5).
Discussion
The age of patients requiring renal replacement therapy and the age at the time point of kidney transplantation are steadily increasing. The present study was conducted to identify risk factors for mortality of elderly patients following kidney transplantation.
The median patient survival after 5 years was 61.2% in the ESP group (Figure 3A), which is consistent with data noted by Frei and colleagues, reporting a 5-year survival rate of 60% in ESP patients [8]. We observed increased mortality in the ESP group relative to the ETKAS group, which is similar to previous studies [11,12]; 44.8% of patients in the ESP group died during the observation period of 43 months in comparison to 13.5% in the ETKAS group. Almost half of the ESP patients who died during the observation period had a functioning graft at the time of death (n=24, n=52 patients total; Table 3). ESP patients were significantly older than patients in the ETKAS group (Table 1). Age at transplantation is a well-identified risk factor for mortality, as indicated by the present study (Table 5) and previous studies [20]. Diabetes is an additional risk factor for mortality [8,21]. Though the incidence of diabetes was evenly distributed between groups in the present study (Table 1), more patients in the ESP group had ESKD due to diabetic nephropathy, likely due to the increased patient age. Our univariate analysis identified ESKD resulting from diabetic nephropathy as a risk factor for mortality (odds ratio 2.51, Table 5). The multivariate analysis indicated age, number of HLA mismatches, and CMV serostatus with a seropositive donor and seronegative recipient as risk factors for mortality. Cardiac diseases were significantly more prevalent in ESP patients at the time of transplantation (Table 1) and were identified as an additional risk factor for mortality in the univariate analysis performed (odds ratio 3.43, Table 5), which is supported by a previous study [21].
Infections and cardiovascular disease are the predominant causes of death in elderly transplant recipients [22–24], with infections as the leading cause of death [25,26]. Similarly, our study indicated infectious diseases as the main cause of death in both the ESP and the ETKAS groups (Figure 3B). In the present study, the number of patients with infectious diseases in the ESP group was similar to the ETKAS group, but the number of patients dying from infectious diseases was more than double in the ESP group. The severity of infections increases with age [27]. Our data suggest that elderly patients have more severe infections, which increases mortality.
Increased donor age is associated with reduced graft survival [5,6,28,29]. Donor age was therefore included in the kidney donor risk index, which stratifies factors that are associated with a worse outcome [30]. Additional risk factors for graft loss are DGF, number of HLA mismatches, and cold ischemia time [8,31]. In our study, the incidence of DGF was similar between groups (Table 2), which might have contributed to a comparable incidence of graft loss within the first year (Table 2) and a similar long-term kidney graft function after 5 years (Figure 2B). Death-censored survival free from graft loss revealed a significant difference between the ESP and ETKAS group, which resulted from higher mortality in the ESP group. Due to regional organ allocation, the cold ischemia time was shorter in the ESP cohort and the number of HLA mismatches was higher (Table 1).
In our study, the maintenance immunosuppressive therapy did not differ between groups (Table 2), which resulted in a similar incidence and severity of rejections in both groups (Table 2 and Figure 1). We observed acute rejections in only 24.1% of ESP and 23.4% of ETKAS patients (Table 2). Importantly, this was in the presence of poorly matched organs, with a higher number of HLA mismatches in the ESP group. Previous studies reported an increased rate of acute rejections and greater prevalence of donor-specific antibodies after transplantation in ESP patients [8,10,32]. Frei and colleagues reported acute rejections in 29.1% of ESP patients, which made acute rejections the main cause of graft loss [8]. In their cohort, 79.9% of patients received triple immunosuppressive therapy at the time of initiation and only 59.1% of patients after 12 months, which contrasts to 96.3% in our ESP cohort (Table 2). Of note, the reported 5-year patient survival was similar to our ESP cohort, which was subjected to an intensified immunosuppressive regimen. Fritsche and colleagues reported an even higher rejection rate for ESP patients, with 43.2% within the first year [32]. In their cohort, 77% of ESP patients were treated with mycophenolate and 52% with tacrolimus. In contrast, 93.1% of patients in our study were treated with mycophenolate and 98.3% of patients were treated with tacrolimus, which might have contributed to the lower incidence of rejections observed in our study (Table 2). Changes in pharmacokinetics and pharmacodynamics in elderly patients are a subject of discussion concerning adjustments in the standard immunosuppressive therapy [33]. However, triple therapy with prednisone, tacrolimus, and mycophenolate should be considered in older patients since reported regimens with less immunosuppressive therapy increased the rate of rejections in the context of similar mortality [8,32].
Additional risk factors for mortality identified in our patient cohort are the number of HLA mismatches according to both, the univariate and multivariate analysis, and previous diseases in the univariate analysis (Table 5). HLA mismatching is a critical prognostic factor for graft and recipient survival [34]. The CMV serostatus with a CMV-negative recipient receiving an organ from a CMV-positive donor was identified as a risk factor for mortality in our study (Table 5). This serostatus is also associated with an increased risk for leukopenia, which increases mortality following kidney transplantation [35]. In contrast, the CMV serostatus with a seropositive donor and recipient trended to be protective, although this result was not statistically significant (Table 5). The time on dialysis was a protective factor according to the univariate analysis (odds ratio 0.99,
Despite numerous risk factors for increased mortality following kidney transplantation, it is important to emphasize that kidney transplantation in ESP patients results in longer life expectancy and improved quality of life compared to dialysis patients in this high-risk patient cohort [13,15–17]. The ESP aims for a shorter cold ischemia time, which was also observed in our study. Of note, this resulted in a similar incidence of delayed graft function and a similar graft function in the long term despite significantly older donors. Major limitations of our study are its retrospective observational design, small study population, and the restriction to a single transplant center. Based on these limitations, the clinical significance of this analysis should be interpreted with caution.
Conclusions
In summary, our study showed increased mortality in the ESP cohort. Our identified risk factors in the multivariate analysis include older age, number of HLA mismatches, and CMV serostatus with a CMV-positive donor and -negative recipient. According to the univariate analysis, additional risk factors are ESKD due to diabetic nephropathy, cardiac disease, and number of previous diseases. It is critical to identify these patients, who warrant special attention. Despite increased graft and recipient age, kidney allocation according to the ESP is associated with a graft survival comparable to patients allocated in the ETKAS. A similar immunosuppressive regimen in ESP compared to ETKAS patients results in a comparable rejection rate, but also increases the risk of infectious diseases, which is the most prevalent cause of death in elderly kidney transplant recipients. Randomized controlled studies with larger numbers of patients are needed to elucidate the optimal immunosuppressive regimen in this high-risk patient population.
Figures
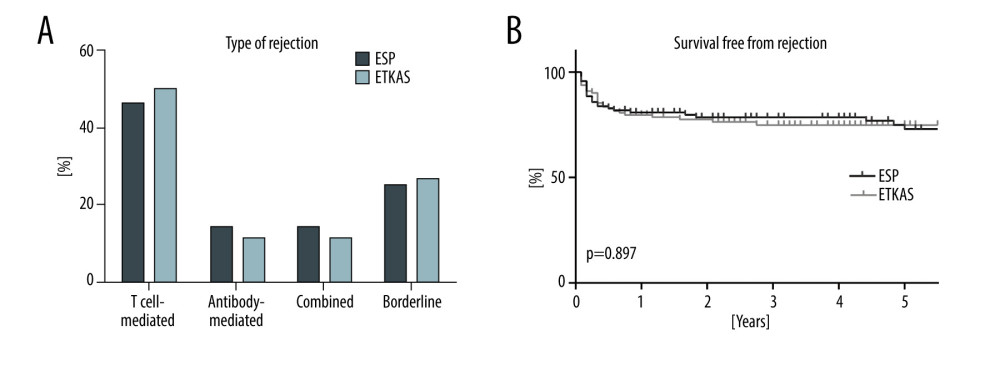 Figure 1. Acute rejection after kidney transplantation. (A) Proportion of different type of rejections following transplantation. The percentage of rejections refers to the total number of rejections. (B) Kaplan-Meier plot illustrating survival free from rejection by organ allocation method. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 1. Acute rejection after kidney transplantation. (A) Proportion of different type of rejections following transplantation. The percentage of rejections refers to the total number of rejections. (B) Kaplan-Meier plot illustrating survival free from rejection by organ allocation method. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). 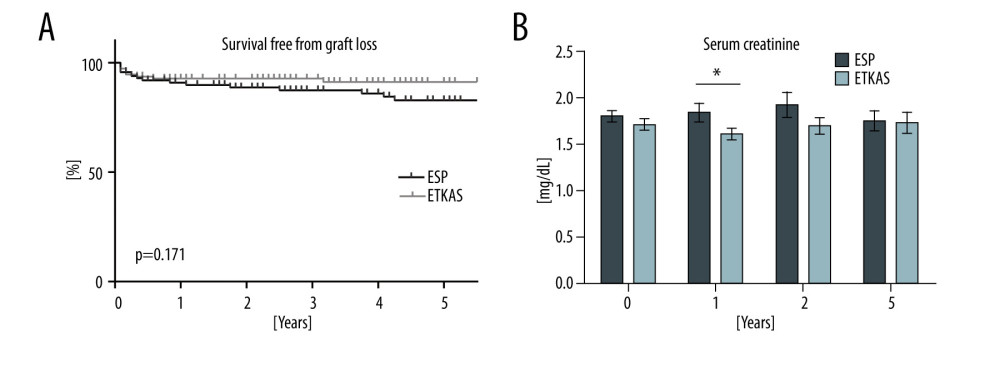 Figure 2. Graft loss and graft function. (A) Kaplan-Meier plot illustrating survival free from graft loss by organ allocation method. (B) Graft function following kidney transplantation as assessed by serum creatinine levels. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 2. Graft loss and graft function. (A) Kaplan-Meier plot illustrating survival free from graft loss by organ allocation method. (B) Graft function following kidney transplantation as assessed by serum creatinine levels. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). 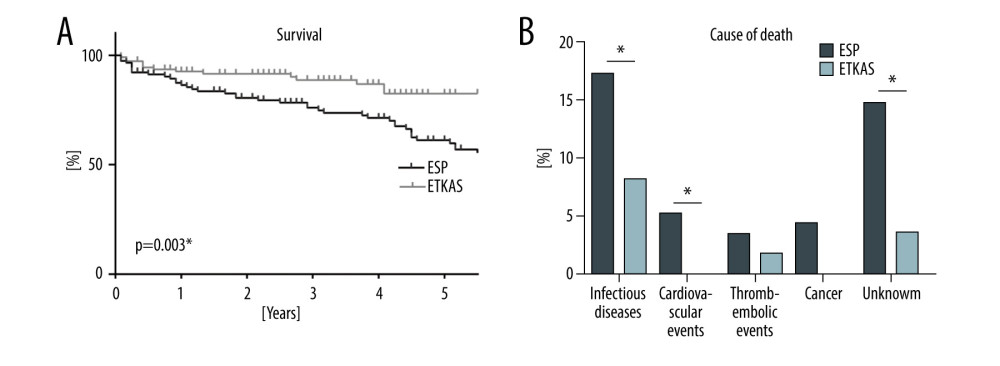 Figure 3. Mortality and cause of death. (A) Kaplan-Meier plot illustrating survival by organ allocation method and (B) Cause of death by organ allocation method. The percentage of cause of death refers to the total number of patients. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 3. Mortality and cause of death. (A) Kaplan-Meier plot illustrating survival by organ allocation method and (B) Cause of death by organ allocation method. The percentage of cause of death refers to the total number of patients. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). 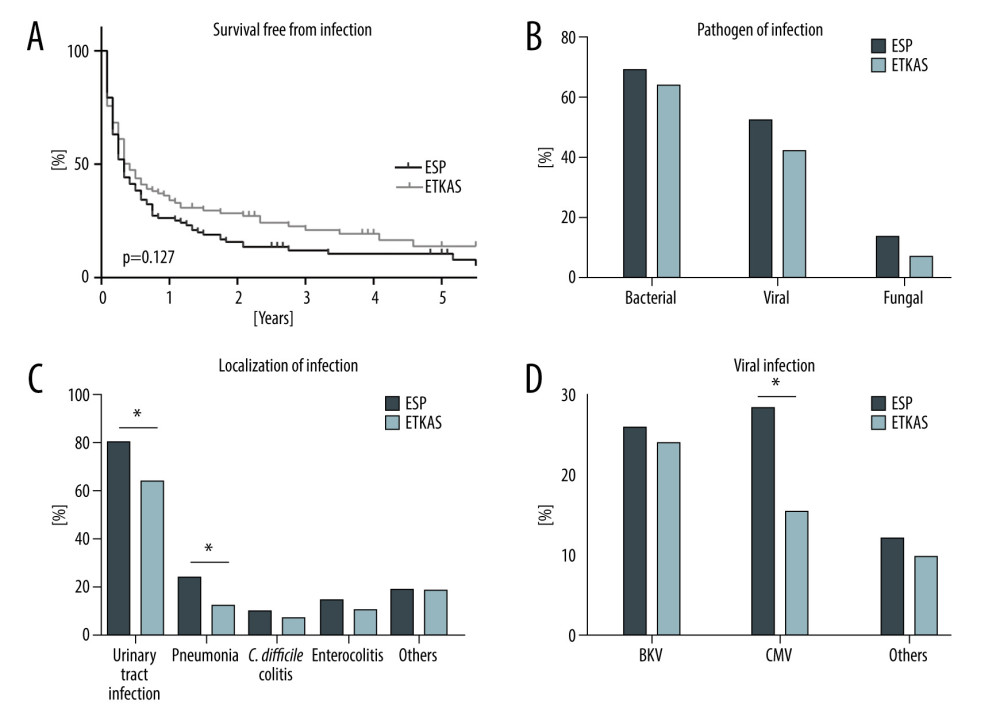 Figure 4. Infectious diseases. (A) Kaplan-Meier plot illustrating survival free from infections by organ allocation method. (B) Incidence of infections according to different pathogens. The percentage of pathogens refers to the total number of patients. (C) Incidence of infections according to different localizations. (D) Incidence of different viral infections. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 4. Infectious diseases. (A) Kaplan-Meier plot illustrating survival free from infections by organ allocation method. (B) Incidence of infections according to different pathogens. The percentage of pathogens refers to the total number of patients. (C) Incidence of infections according to different localizations. (D) Incidence of different viral infections. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). References
1. www.usrds.org/qtr/default.aspx
2. Yu MY, Kim YC, Lee JP, Death with graft function after kidney transplantation: A single-center experience: Clin Exp Nephrol, 2018; 22(3); 710-18
3. Meier-Kriesche HU, Schold JD, Gaston RS, Kidneys from deceased donors: Maximizing the value of a scarce resource: Am J Transplant, 2005; 5(7); 1725-30
4. : Eurotransplant, 2019 https://www.eurotransplant.org/wp-content/uploads/2020/06/Annual-Report-2019.pdf
5. Darmady EM, Transplantation and the ageing kidney: Lancet, 1974; 2(7888); 1046-47
6. Alexander JW, Bennett LE, Breen TJ, Effect of donor age on outcome of kidney transplantation. A two-year analysis of transplants reported to the United Network for Organ Sharing Registry: Transplantation, 1994; 57(6); 871-76
7. Dayoub JC, Cortese F, Anzic A, The effects of donor age on organ transplants: A review and implications for aging research: Exp Gerontol, 2018; 110; 230-40
8. Frei U, Noeldeke J, Machold-Fabrizii V, Prospective age-matching in elderly kidney transplant recipients – a 5-year analysis of the Eurotransplant Senior Program: Am J Transplant, 2008; 8(1); 50-57
9. Giessing M, Conrad S, Schonberger BKidney donors and kidney transplantation in the elderly: Der Urologe Ausg A, 2004; 43(8); 947-54 [in German]
10. Halleck F, Khadzhynov D, Liefeldt L, Immunologic outcome in elderly kidney transplant recipients: Is it time for HLA-DR matching?: Nephrol Dial Transplant, 2016; 31(12); 2143-49
11. Tullius SG, Tran H, Guleria I, The combination of donor and recipient age is critical in determining host immunoresponsiveness and renal transplant outcome: Ann Surg, 2010; 252(4); 662-74
12. McAdams-DeMarco MA, James N, Trends in kidney transplant outcomes in older adults: J Am Geriatr Soc, 2014; 62(12); 2235-42
13. Rao PS, Merion RM, Ashby VB, Renal transplantation in elderly patients older than 70 years of age: Results from the Scientific Registry of Transplant Recipients: Transplantation, 2007; 83(8); 1069-74
14. Wolfe RA, Ashby VB, Milford EL, Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant: N Engl J Med, 1999; 341(23); 1725-30
15. Hunsicker LG, A survival advantage for renal transplantation: N Engl J Med, 1999; 341(23); 1762-63
16. Johnson DW, Herzig K, Purdie D, A comparison of the effects of dialysis and renal transplantation on the survival of older uremic patients: Transplantation, 2000; 69(5); 794-99
17. Schachtner T, Otto NM, Reinke P, Two decades of the Eurotransplant Senior Program: The gender gap in mortality impacts patient survival after kidney transplantation: Clin Kidney J, 2020; 13(6); 1091-100
18. Arcos E, Perez-Saez MJ, Comas J, Assessing the limits in kidney transplantation: Use of extremely elderly donors and outcomes in elderly recipients: Transplantation, 2020; 104(1); 176-83
19. Haas M, Loupy A, Lefaucheur C, The Banff 2017 Kidney Meeting Report: Revised diagnostic criteria for chronic active T cell-mediated rejection, antibody-mediated rejection, and prospects for integrative endpoints for next-generation clinical trials: Am J Transplant, 2018; 18(2); 293-307
20. Ojo AO, Hanson JA, Wolfe RA, Long-term survival in renal transplant recipients with graft function: Kidney Int, 2000; 57(1); 307-13
21. Legeai C, Andrianasolo RM, Moranne O, Benefits of kidney transplantation for a national cohort of patients aged 70 years and older starting renal replacement therapy: Am J Transplant, 2018; 18(11); 2695-707
22. Martins PN, Pratschke J, Pascher A, Age and immune response in organ transplantation: Transplantation, 2005; 79(2); 127-32
23. Trouillhet I, Benito N, Cervera C, Influence of age in renal transplant infections: Cases and controls study: Transplantation, 2005; 80(7); 989-92
24. Kappes U, Schanz G, Gerhardt U, Influence of age on the prognosis of renal transplant recipients: Am J Nephrol, 2001; 21(4); 259-63
25. Jacobi J, Beckmann S, Heller K, Deceased donor kidney transplantation in the Eurotransplant Senior Program (ESP): A single-center experience from 2008 to 2013: Ann Transplant, 2016; 21; 94-104
26. Schamberger B, Lohmann D, Sollinger D, Association of kidney donor risk index with the outcome after kidney transplantation in the Eurotransplant Senior Program: Annals Transplant, 2018; 23; 775-81
27. Gavazzi G, Krause KH, Ageing and infection: Lancet Infect Dis, 2002; 2(11); 659-66
28. Englum BR, Schechter MA, Irish WD, Outcomes in kidney transplant recipients from older living donors: Transplantation, 2015; 99(2); 309-15
29. Susal C, Kumru G, Dohler B, Should kidney allografts from old donors be allocated only to old recipients?: Transplant Int, 2020; 33(8); 849-57
30. Rao PS, Schaubel DE, Guidinger MK, A comprehensive risk quantification score for deceased donor kidneys: The kidney donor risk index: Transplantation, 2009; 88(2); 231-36
31. Bahde R, Vowinkel T, Unser J, Prognostic factors for kidney allograft survival in the Eurotransplant Senior Program: Ann Transplant, 2014; 19; 201-9
32. Fritsche L, Horstrup J, Budde K, Old-for-old kidney allocation allows successful expansion of the donor and recipient pool: Am J Transplant, 2003; 3(11); 1434-39
33. Peeters LEJ, Andrews LM, Hesselink DA, Personalized immunosuppression in elderly renal transplant recipients: Pharmacol Res, 2018; 130; 303-7
34. Shi X, Lv J, Han W, What is the impact of human leukocyte antigen mismatching on graft survival and mortality in renal transplantation? A meta-analysis of 23 cohort studies involving 486,608 recipients: BMC Nephrol, 2018; 19(1); 116
35. Henningsen M, Jaenigen B, Zschiedrich S, Risk factors and management of leukopenia after kidney transplantation: A single-center experience: Transplant Proc, 2021; 53(5); 1589-98
Figures
 Figure 1. Acute rejection after kidney transplantation. (A) Proportion of different type of rejections following transplantation. The percentage of rejections refers to the total number of rejections. (B) Kaplan-Meier plot illustrating survival free from rejection by organ allocation method. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 1. Acute rejection after kidney transplantation. (A) Proportion of different type of rejections following transplantation. The percentage of rejections refers to the total number of rejections. (B) Kaplan-Meier plot illustrating survival free from rejection by organ allocation method. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). Figure 2. Graft loss and graft function. (A) Kaplan-Meier plot illustrating survival free from graft loss by organ allocation method. (B) Graft function following kidney transplantation as assessed by serum creatinine levels. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 2. Graft loss and graft function. (A) Kaplan-Meier plot illustrating survival free from graft loss by organ allocation method. (B) Graft function following kidney transplantation as assessed by serum creatinine levels. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). Figure 3. Mortality and cause of death. (A) Kaplan-Meier plot illustrating survival by organ allocation method and (B) Cause of death by organ allocation method. The percentage of cause of death refers to the total number of patients. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 3. Mortality and cause of death. (A) Kaplan-Meier plot illustrating survival by organ allocation method and (B) Cause of death by organ allocation method. The percentage of cause of death refers to the total number of patients. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). Figure 4. Infectious diseases. (A) Kaplan-Meier plot illustrating survival free from infections by organ allocation method. (B) Incidence of infections according to different pathogens. The percentage of pathogens refers to the total number of patients. (C) Incidence of infections according to different localizations. (D) Incidence of different viral infections. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA).
Figure 4. Infectious diseases. (A) Kaplan-Meier plot illustrating survival free from infections by organ allocation method. (B) Incidence of infections according to different pathogens. The percentage of pathogens refers to the total number of patients. (C) Incidence of infections according to different localizations. (D) Incidence of different viral infections. ESP – Eurotransplant Senior Program; ETKAS – Eurotransplant Kidney Allocation System. * P<0.05. (GraphPad Prism version 8.0, GraphPad Software, Inc., San Diego, CA, USA). Tables
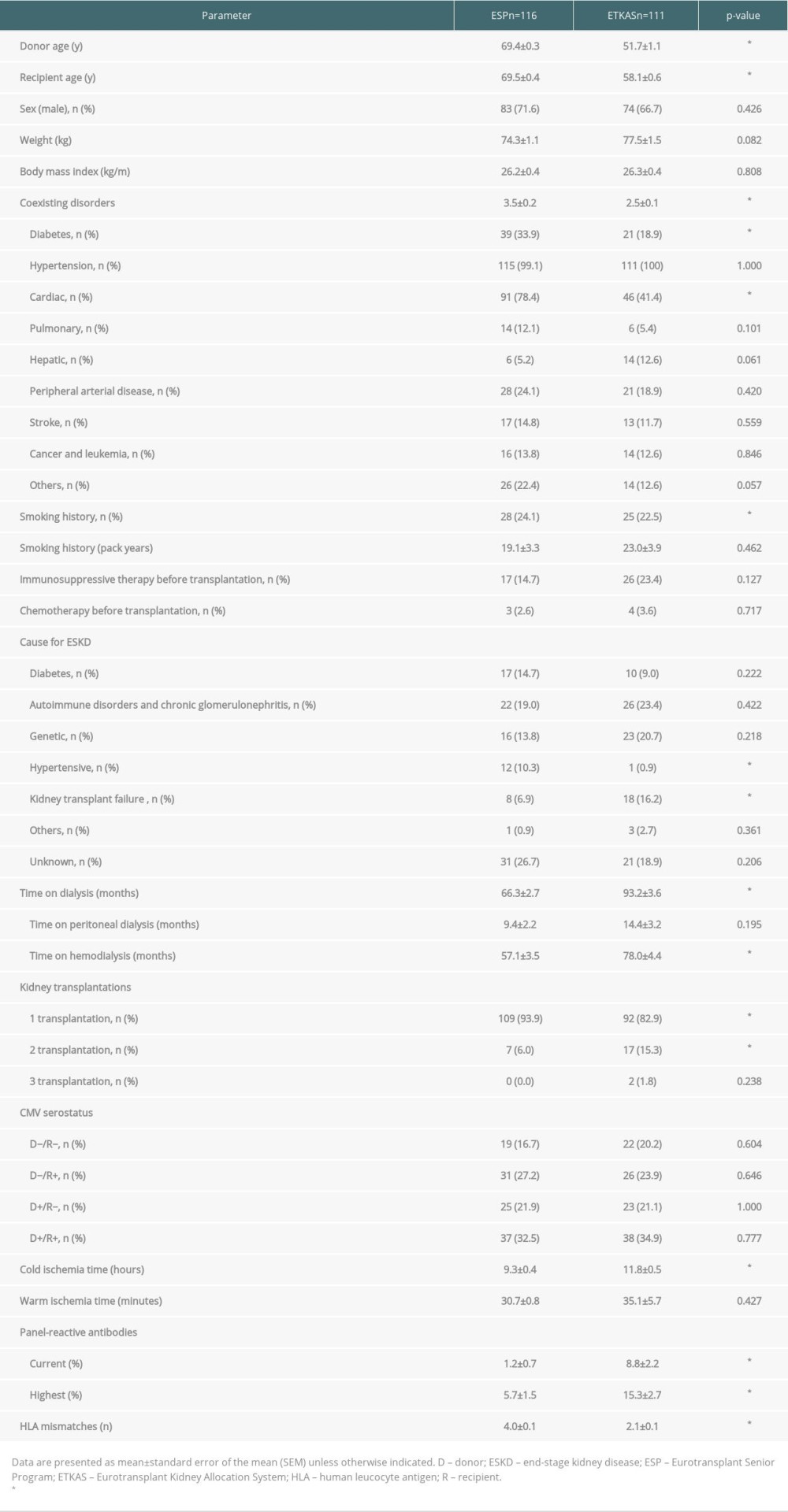 Table 1. Baseline characteristics.
Table 1. Baseline characteristics.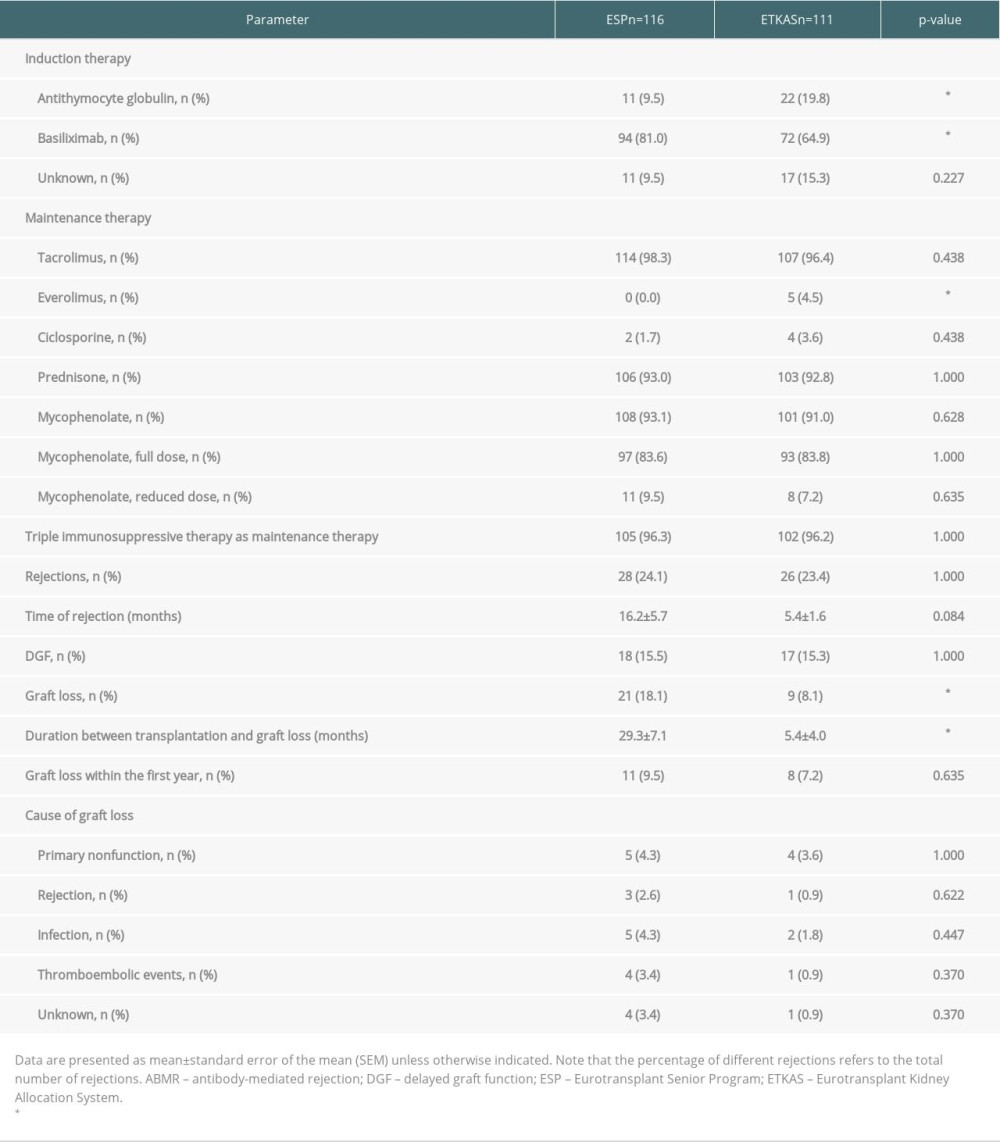 Table 2. Immunosuppressive therapy, acute rejections, and graft function.
Table 2. Immunosuppressive therapy, acute rejections, and graft function.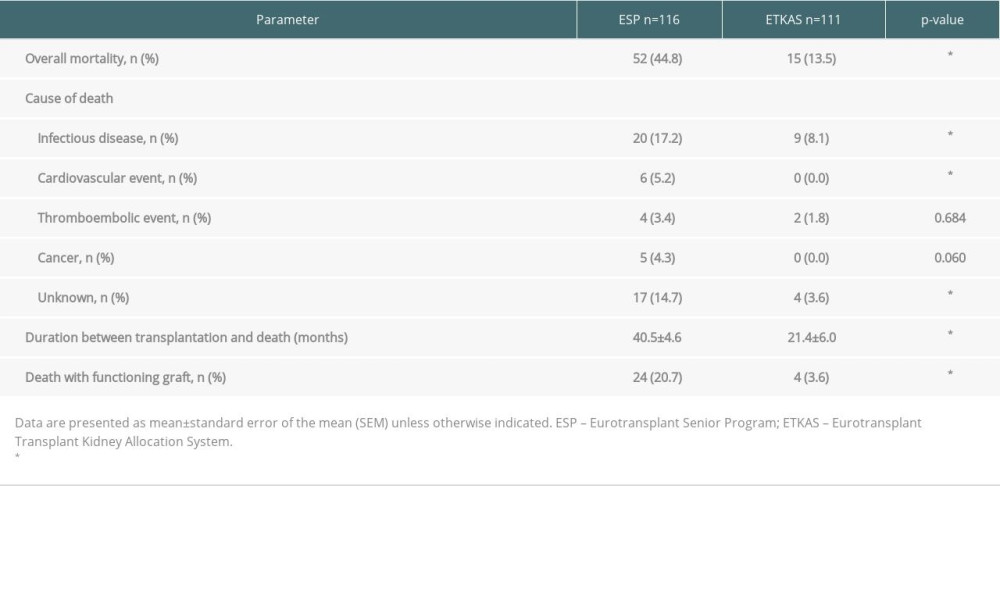 Table 3. Graft function and mortality.
Table 3. Graft function and mortality.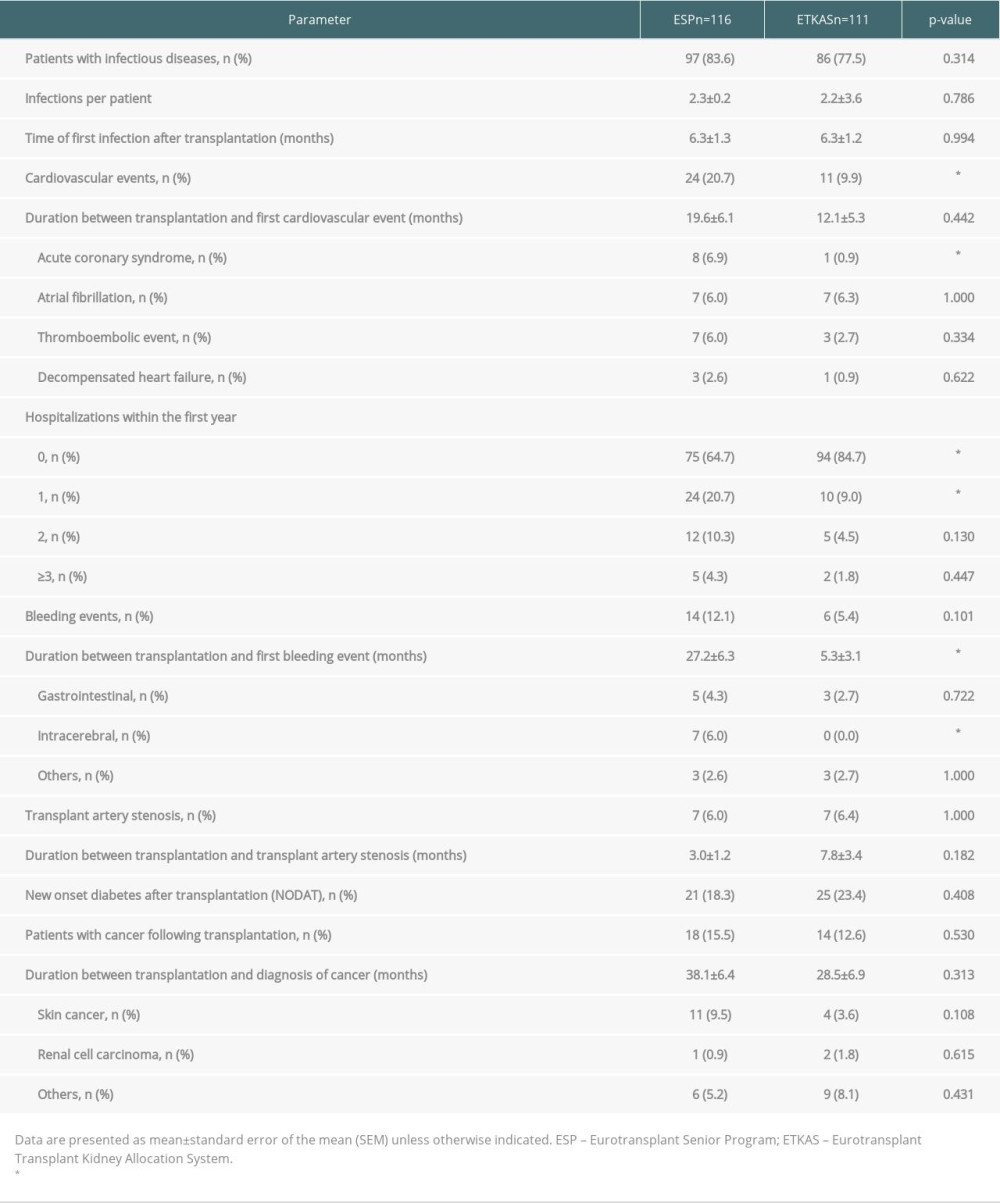 Table 4. Outcome parameters.
Table 4. Outcome parameters.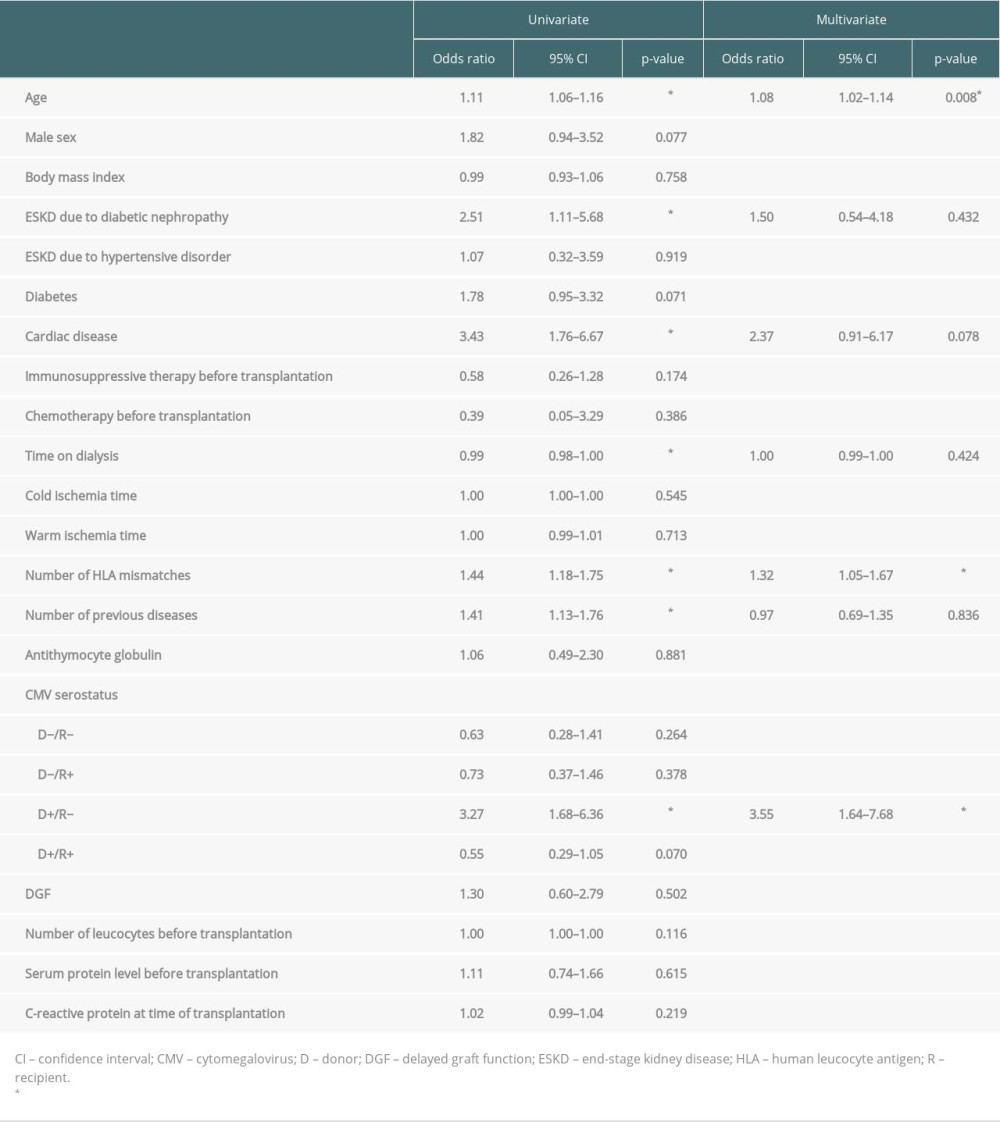 Table 5. Risk factors for mortality.
Table 5. Risk factors for mortality. Table 1. Baseline characteristics.
Table 1. Baseline characteristics. Table 2. Immunosuppressive therapy, acute rejections, and graft function.
Table 2. Immunosuppressive therapy, acute rejections, and graft function. Table 3. Graft function and mortality.
Table 3. Graft function and mortality. Table 4. Outcome parameters.
Table 4. Outcome parameters. Table 5. Risk factors for mortality.
Table 5. Risk factors for mortality. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








