13 December 2022: Original Paper
Hepatic Ischemia/Reperfusion Injury After Liver Transplantation Is Not Associated with Early Impairment of Left Ventricular Ejection Fraction
Zachary P. Rokop1ABCDEF*, Kyle Frick2ABDE, Joseph Zenisek2ABDE, Elizabeth Kroepfl3ABDE, Plamen Mihaylov4ABDE, Kavish R. PatidarDOI: 10.12659/AOT.938105
Ann Transplant 2022; 27:e938105
Abstract
BACKGROUND: Early myocardial dysfunction is a known complication following liver transplant. Although hepatic ischemia/reperfusion injury (hIRI) has been shown to cause myocardial injury in rat and porcine models, the clinical association between hIRI and early myocardial dysfunction in humans has not yet been established. We sought to define this relationship through cardiac evaluation via transthoracic echocardiography (TTE) on postoperative day (POD) 1 in adult liver transplant recipients.
MATERIAL AND METHODS: TTE was performed on POD1 in all liver transplant patients transplanted between January 2020 and April 2021. Hepatic IRI was stratified by serum AST levels on POD1 (none: <200; mild: 200-2000; moderate: 2000-5000; severe: >5000). All patients had pre-transplant TTE as part of the transplant evaluation.
RESULTS: A total of 173 patients underwent liver transplant (LT) between 2020 and 2021 and had a TTE on POD 1 (median time to echo: 1 day). hIRI was present in 142 (82%) patients (69% mild, 8.6% moderate, 4% severe). Paired analysis between pre-LT and post-LT left ventricular ejection fraction (LVEF) of the entire study population demonstrated no significant decrease following LT (mean difference: -1.376%, P=0.08). There were no significant differences in post-LT LVEF when patients were stratified by severity of hIRI. Three patients (1.7%) had significant post-transplant impairment of LVEF (<35%). None of these patients had significant hIRI.
CONCLUSIONS: hIRI after liver transplantation is not associated with immediate reduction in LVEF. The pathophysiology of post-LT cardiomyopathy may be driven by extra-hepatic triggers.
Keywords: Heart Failure, Liver Transplantation, Adult, Humans, Rats, Swine, Animals, Stroke Volume, Ventricular Function, Left, Ischemia
Background
Hepatic ischemia/reperfusion injury (hIRI) is a complex process characterized by 2 distinct phases. During the ischemic period, hepatocytes are subjected to hypoxic stress, resulting in both necrotic and apoptotic cell death. This promotes the accumulation of cytokines and toxic metabolic byproducts within the ischemic liver. Upon restoration of oxygenated blood flow, a surge of reactive oxygen species mediates the second phase of hepatic injury [1]. Simultaneously, cytokines such as TNF-α and IL-1 are released, promoting neutrophil chemotaxis, activation, and additional injury [2].
In addition to producing significant local effects on the liver, hIRI has also been implicated in remote organ damage. Researchers have demonstrated myocardial necrosis following hepatic ischemia in rat and porcine models of hIRI [3–5]. Importantly, myocardial injury was detected in these experiments within 6 h of hepatic reperfusion, suggesting such damage was mediated by products of hIRI alone and not from any ensuing liver failure that may occur. Another study, performed in dogs, demonstrated that TNF-α can reversibly impair left ventricular systolic function [6]. As TNF-α is released by ischemic liver during reperfusion, this finding suggests that hIRI contributes to post-liver transplantation (LT) cardiac dysfunction.
Although the link between hIRI and myocardial injury has been made in animal models, clinical manifestations of this effect have yet to be defined. Previous efforts have identified biochemical evidence of myocardial injury (via elevated serum troponin levels) in up to 74% of all patients with acute liver failure [7], but failed to specify hIRI as a mechanism. Several other studies sought to characterize the phenomenon of acute postoperative systolic heart failure via echocardiography. These identified an incidence of acute systolic heart failure of 1–7%, most often secondary to stress-induced cardiomyopathy [8–12]. However, these studies did not distinguish between early- and late-onset of heart failure and did not address any possible association with hIRI. Furthermore, these studies were retrospective in nature, with echocardiography that was performed on a “for-cause” basis, potentially missing patients who developed myocardial dysfunction without overt clinical manifestation.
We hypothesized that hIRI induces suppression of left ventricular function in the immediate postoperative period. To test this hypothesis, we evaluated cardiac function on postoperative day (POD) 1 by transthoracic echocardiogram (TTE). In this report, we present the findings of this study on 173 consecutive liver transplant recipients.
Material and Methods
STUDY POPULATION:
All patients undergoing liver transplant between January 2020 and April 2021 underwent echocardiogram on POD 1 (n=173) and were subsequently included in this study. All patients had previous echocardiography performed within 6 months of transplant as part of their preoperative workup. Based on individual risk factors for coronary artery disease, and cardiologist assessment, a subset of patients were subjected to further non-invasive cardiac evaluation (treadmill or dobutamine stress test), and coronary CT angiography/cardiac catheterization.
CLINICAL DATA COLLECTION:
Donor and recipient demographics were extracted from the Indiana University liver transplant database, in accordance with Institutional Review Board approval. Recipient variables extracted included age, sex, race, MELD at transplant, indication for liver transplant, graft outcome, and patient outcome. Donor variables included age, donation after circulatory death (DCD), and warm and cold ischemia times. Serum chemistries, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine (Cr), and B-type natriuretic peptide (BNP), were collected on POD 1. Patients were stratified by severity of hIRI as defined by AST levels: none: AST<200, mild injury: AST 200–2000, moderate injury: AST 2000–5000, severe injury: AST >5000. All liver grafts were biopsied at the time of transplant. Pathologic grading of steatosis was performed on these samples in accordance with the guidelines created by Kleiner et al [13].
ECHOCARDIOGRAPHY:
Transthoracic echocardiography was performed on POD1, or as close to POD1 as logistically feasible, in all patients included in this study. Image review and interpretation was performed by 2 independent cardiologists with experience in cardiac imaging of patients with liver disease, who were blinded to patient history and clinical outcomes. Parameters evaluated included left ventricular ejection fraction (LVEF) and the presence of wall motion abnormalities.
STATISTICAL ANALYSIS:
Categorical data are reported as frequencies. Continuous data are reported as means with standard deviation, or medians with range depending on their distribution. Statistical analysis was performed using IBM SPSS Statistics for Mac, version 28 (IBM Corp., Armonk, N.Y., USA). Differences in frequencies were assessed using Fisher’s exact test, while differences in means were assessed using the
Results
DEMOGRAPHICS AND PREOPERATIVE AND INTRAOPERATIVE VARIABLES:
A total of 173 patients were identified who underwent LT between 2020 and 2021 and had an echocardiogram performed on POD 1. Recipient demographics are summarized in Table 1. Patients were predominately white (88%) and male (61%). Liver failure secondary to alcoholic cirrhosis was the most common indication for transplant. There was a 4.07% mortality rate within this cohort; no patients died from cardiac complications.
Preoperative and intraoperative variables, stratified by severity of hIRI, are displayed in Table 2. Overall, hIRI was present in 142 patients (82%). Of those with hIRI, 69% had mild IRI, 8.6% had moderate IRI, and 4% had severe IRI, as classified by POD1 AST levels. There was a significantly greater proportion of donation after cardiac death (DCD) livers transplanted in the hIRI groups as compared to the no hIRI group (P<0.05). A significantly lower mean MELD score was observed in the severe hIRI group as compared to the no hIRI group (P<0.05); this trend did not persist between the other study groups. Patients in the severe hIRI group required significantly greater intraoperative doses of epinephrine (P<0.001), norepinephrine (P=0.004), and vasopressin (P=0.03).
Table 3 summarizes post-LT outcomes stratified by severity of hIRI. There were no significant differences in pre- or post-LT LVEF, postoperative pressor use, or POD1 BNP between the 4 groups. Rates of graft loss and patient mortality were similar as well.
ECHOCARDIOGRAPHIC FINDINGS AND HIRI:
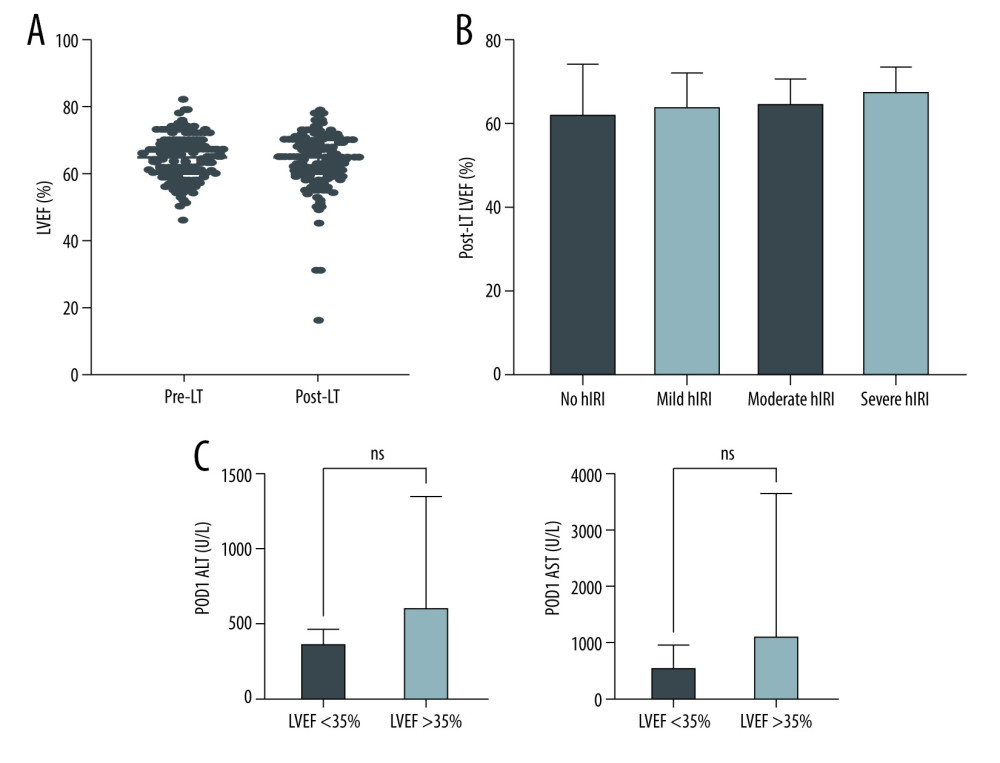
The median time to echocardiogram following liver transplant was 1 day. There was no significant difference between pre-LT and post-LT ejection fractions on pairwise analysis of the entire study population (mean difference: −0.74%, P=0.16) (Figure 1A).
To define the relationship between hIRI and immediate postoperative LVEF suppression, we stratified patients by severity of IRI. There was no significant difference in post-LT LVEF between patients without hIRI, or with mild, moderate, or severe hIRI (Figure 1B). We additionally found there to be no significant difference in POD1 ALT or AST between patients who had an LVEF of <35% after LT as compared to the rest of the study population. (ALT: P=0.57, AST: P=0.68) (Figure 1C).
Discussion
Our data do not support a clinical association between hIRI and reduction in LVEF following liver transplant. There was no significant decline in post-LT LVEF, even in patients with severe hIRI. Furthermore, although a small proportion of this study (1.7%) population developed severe impairment of LVEF following LT (LVEF <35%), these patients all had mild hIRI at a level that was below average for the overall study population. Taken together, these findings suggest that hIRI does not cause a clinically detectable reduction in LVEF in the immediate postoperative period. Previous experiments in porcine models have demonstrated biochemical and histological evidence of myocardial injury without notable impairment of cardiac function, supporting our findings [4]. Troponin levels were not routinely checked postoperatively in our cohort. Therefore, we were unable to compare the incidence of myocardial injury, as detected biochemically, to echocardiographic findings.
Although rare, post-LT cardiac dysfunction is a major clinical problem and is poorly characterized from a mechanistic point of view. The negative finding of our study could be one of the pieces of evidence needed to unravel the pathophysiology of cardiomyopathy in this setting. Although our results do not support a direct correlation between hIRI and suppression of LVEF, it is possible that hIRI can indirectly contribute to post-LT cardiomyopathy through other systemic effects. For example, hIRI, via release of proinflammatory cytokines and vasoactive molecules, can induce profound systemic hypotension requiring administration of vasopressor agents to correct [14]. In our study population, we observed that patients with severe hIRI required a significantly greater cumulative dose of intraoperative inotropic and vasopressor medications. Pressor use is an established risk factor for development of stress-induced cardiomyopathy, which is a known post-LT cardiac complication that has significant impact on recipient mortality [10,15–20]. It is also hypothesized that cirrhotic cardiomyopathy can be responsible for post-LT cardiac dysfunction [10,21]. Cirrhotic cardiomyopathy is known to develop due to systemic changes in chronic liver failure and is characterized by the constellation of low systemic vascular resistance, high cardiac output at rest, and impaired cardiac response to stress [22]. As a result, patients with this condition can develop left ventricular systolic dysfunction when subjected to the physiologic stress of the liver transplant operation. Although all patients in our study had preserved cardiac function prior to transplantation, it should be noted that cirrhotic cardiomyopathy is not easily detected with standard resting or dobutamine stress echocardiography [23,24]. Therefore, the contribution of this process to post-LT cardiac dysfunction may be underappreciated.
There are some limitations to our study. We chose to assess cardiac function primarily through LVEF as measured by TTE, as this value correlates well with systolic cardiac function and has standardized measurement criteria [25]. Furthermore, impairment in cardiac systolic function has been associated with poor outcomes following LT, making this a relevant parameter with which to examine [8,10,26,27]. However, it is possible that cardiac dysfunction following hIRI is not captured through this measurement. Future efforts could include evaluation of global left ventricular strain immediately postoperatively. Additionally, we observed an overall low incidence of abnormal echocardiogram findings in our study population, which may suggest our study was underpowered. Nonetheless, this study examined the association of hIRI with immediate post-transplant cardiac function in a systematic fashion that included all consecutive patients. This methodology avoided any selection bias which could develop should patients have been selected for echocardiogram on a “for-cause” basis, as patients with clinical concern for cardiac dysfunction are more likely to have abnormal echocardiographic findings. Therefore, we believe our results accurately reflect trends in left ventricular function immediately following LT.
Conclusions
Our study demonstrated a lack of association between hIRI and early myocardial suppression in liver transplant recipients. Other extra-hepatic factors may contribute to post-LT cardiomyopathy.
Tables
Table 1. Recipient demographics.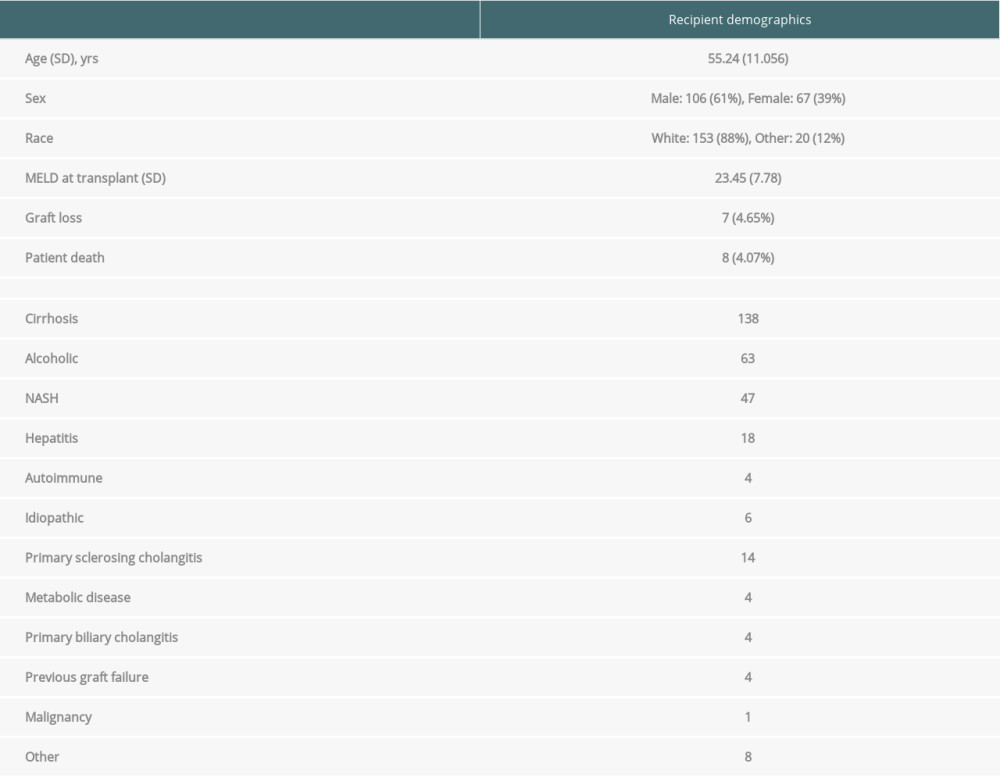 Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group.
Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group.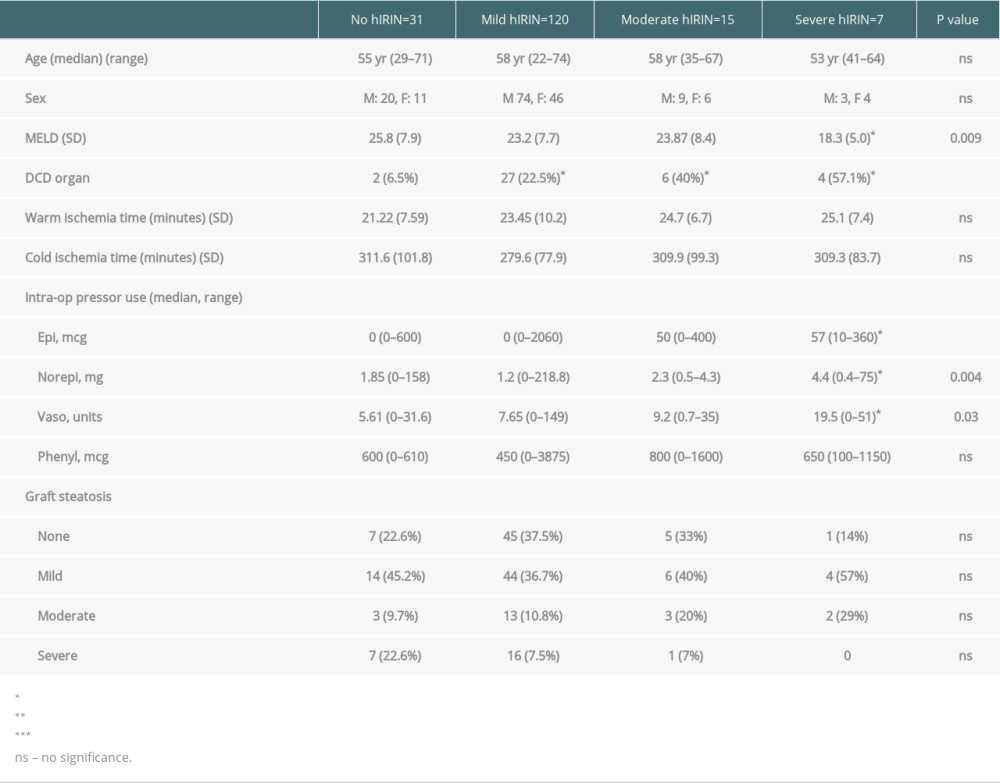 Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified.
Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified.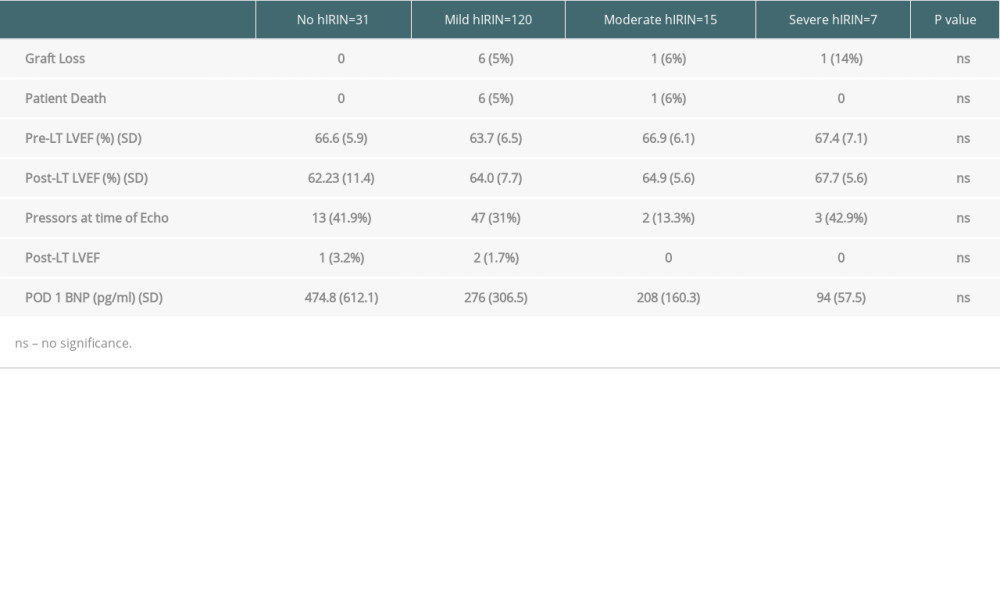
References
1. Peralta C, Jiménez-Castro MB, Gracia-Sancho J, Hepatic ischemia and reperfusion injury: Effects on the liver sinusoidal milieu: J Hepatol, 2013; 59(5); 1094-106
2. Suzuki S, Toledo-Pereyra LH, Interleukin 1 and tumor necrosis factor production as the initial stimulants of liver ischemia and reperfusion injury: J Surg Res, 1994; 57(2); 253-58
3. Chen CF, Wang D, Lin HI, Ischemia/reperfusion of the liver induces heart injury in rats: Transplant Proc, 2007; 39(4); 855-57
4. Papoutsidakis N, Arkadopoulos N, Smyrniotis V, Early myocardial injury is an integral component of experimental acute liver failure – a study in two porcine models: Arch Med Sci, 2011; 7(2); 217-23
5. Chen Y, Liu Z, Xie X, Hydrogen sulphide attenuates renal and cardiac injury after total hepatic ischemia and reperfusion: J Surg Res, 2010; 164(2); e305-13
6. Pagani FD, Baker LS, Hsi C, Left ventricular systolic and diastolic dysfunction after infusion of tumor necrosis factor-alpha in conscious dogs: J Clin Invest, 1992; 90(2); 389-98
7. Parekh NK, Hynan LS, De Lemos J, Lee WMAcute Liver Failure Study Group, Elevated troponin I levels in acute liver failure: is myocardial injury an integral part of acute liver failure?: Hepatology, 2007; 45(6); 1489-95
8. Sampathkumar P, Lerman A, Kim BY, Post-liver transplantation myocardial dysfunction: Liver Transpl Surg, 1998; 4(5); 399-403
9. Stewart KS, Rhim CH, Bahrain ML, Nonischemic cardiomyopathy after orthotopic liver transplantation: A report of three cases and a review of the literature: Liver Transpl, 2005; 11(5); 573-78
10. Schnell F, Donal E, Lorho R, Severe left-sided heart failure early after liver transplantation: Liver Transpl, 2009; 15(10); 1296-305
11. Sonny A, Govindarajan SR, Jaber WA, Cywinski JB, Systolic heart failure after liver transplantation: Incidence, predictors, and outcome: Clin Transplant, 2018; 32(3); e13199
12. Sakr AE, Fraser GE, Doctorian TP, Predictors of systolic heart failure and mortality following orthotopic liver transplantation: A single-center cohort: Transplant Proc, 2019; 51(6); 1950-55
13. Kleiner DE, Brunt EM, Van Natta MNonalcoholic Steatohepatitis Clinical Research Network, Design and validation of a histological scoring system for nonalcoholic fatty liver disease: Hepatology, 2005; 41(6); 1313-21
14. Manning MW, Kumar PA, Maheshwari K, Arora H, Post-reperfusion syndrome in liver transplantation – an overview: J Cardiothorac Vasc Anesth, 2020; 34(2); 501-11
15. Guglin M, Nazif K, New onset nonischemic cardiomyopathy post liver transplantation: Heart Fail Rev, 2022; 27(5); 1829-36
16. Tiwari AK, D’Attellis N, Intraoperative left ventricular apical ballooning: Transient Takotsubo cardiomyopathy during orthotopic liver transplantation: J Cardiothorac Vasc Anesth, 2008; 22(3); 442-45
17. Yataco ML, Difato T, Bargehr J, Reversible non-ischaemic cardiomyopathy and left ventricular dysfunction after liver transplantation: A single-centre experience: Liver Int, 2014; 34(6); e105-10
18. Lyon AR, Rees PS, Prasad S, Stress (Takotsubo) cardiomyopathy – a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning: Nat Clin Pract Cardiovasc Med, 2008; 5(1); 22-29
19. Paur H, Wright PT, Sikkel MB, High levels of circulating epinephrine trigger apical cardiodepression in a β2-adrenergic receptor/Gi-dependent manner: A new model of Takotsubo cardiomyopathy: Circulation, 2012; 126(6); 697-706
20. Avegliano G, Huguet M, Costabel JP, Morphologic pattern of late gadolinium enhancement in Takotsubo cardiomyopathy detected by early cardiovascular magnetic resonance: Clin Cardiol, 2011; 34(3); 178-82
21. Sakr AE, Fraser GE, Doctorian TP, Predictors of systolic heart failure and mortality following orthotopic liver transplantation: A single-center cohort: Transplant Proc, 2019; 51(6); 1950-55
22. Ruiz-del-Árbol L, Serradilla R, Cirrhotic cardiomyopathy: World J Gastroenterol, 2015; 21(41); 11502-21
23. Kazankov K, Holland-Fischer P, Andersen NH, Resting myocardial dysfunction in cirrhosis quantified by tissue Doppler imaging: Liver Int, 2011; 31(4); 534-40
24. Dahl EK, Møller S, Kjær A, Diastolic and autonomic dysfunction in early cirrhosis: A dobutamine stress study: Scand J Gastroenterol, 2014; 49(3); 362-72
25. Klaeboe LG, Edvardsen T, Echocardiographic assessment of left ventricular systolic function: J Echocardiogr, 2019; 17(1); 10-16
26. Mandell MS, Seres T, Lindenfeld J, Risk factors associated with acute heart failure during liver transplant surgery: A case control study: Transplantation, 2015; 99(4); 873-78
27. Qureshi W, Mittal C, Ahmad U, Clinical predictors of post-liver transplant new-onset heart failure: Liver Transpl, 2013; 19(7); 701-10
Tables
 Table 1. Recipient demographics.
Table 1. Recipient demographics. Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group.
Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group. Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified.
Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. Table 1. Recipient demographics.
Table 1. Recipient demographics. Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group.
Table 2. Preoperative and intraoperative recipient variables, stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. P values represent significance of pairwise comparison in relation to no hIRI group. Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified.
Table 3. Post-liver transplant outcomes stratified by severity of hepatic ischemia reperfusion injury (hIRI) as defined by: no hIRI: AST <200; mild hIRI: AST <2000, moderate hIRI: 2000 <AST <5000, severe hIRI: AST >5000. Values are reported as means and standard deviations unless otherwise specified. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588