28 May 2024: Original Paper
Effect of Dexmedetomidine Combined with Remifentanil on Emergence Agitation During Awakening from Sevoflurane Anesthesia for Pediatric Liver Surgery
Yanting Wang1AE, Cuicui Liu1AE, Pei Wang1AE, Linlin Li1AE, Wei Feng1AE*DOI: 10.12659/AOT.943281
Ann Transplant 2024; 29:e943281
Abstract
BACKGROUND: We aimed to assess the effect of dexmedetomidine (Dex) combined with remifentanil on emergence agitation (EA) during awakening from sevoflurane anesthesia for pediatric liver surgery.
MATERIAL AND METHODS: Sixty children who underwent liver surgery in our hospital were prospectively selected and randomly allocated into group A (placebo+remifentanil+sevoflurane) or group B (Dex+remifentanil+sevoflurane). Mean arterial pressure (MAP) and heart rate (HR) at different time points, agitation score during awakening, behavioral status, pain level, and the incidence of postoperative adverse effects were compared in both groups.
RESULTS: Children in group B had lower HR and MAP levels immediately after tracheal extubation and 5 min after tracheal extubation than those in group A. The Aono’s scores, PAED agitation scores, and CHIPP scores at 15 min and 30 min of admission to the PACU were lower in group B than in group A. The incidence of agitation during postoperative anesthesia awakening was lower in group B in contrast to group A. There was no significant difference in postoperative adverse reactions between group A and group B.
CONCLUSIONS: In pediatric liver surgery, the use of Dex+remifentanil+sevoflurane anesthesia can reduce the incidence of EA during the awakening period, stabilize hemodynamic levels, and relieve postoperative pain, and has fewer postoperative adverse effects, which warrants clinical application.
Keywords: Dexmedetomidine, remifentanil, Humans, sevoflurane, Female, Male, Anesthetics, Inhalation, Child, Preschool, Emergence Delirium, Prospective Studies, Hypnotics and Sedatives, Infant, Child, Psychomotor Agitation, Liver, Anesthesia Recovery Period, Piperidines, Double-Blind Method, Drug Therapy, Combination, Methyl Ethers, Analgesics, Opioid
Introduction
Inhalational anesthesia induction is a method commonly used by pediatric anesthesiologists. This is a pivotal technique in such cases due to the lack of venous access and possible airway difficulties [1]. Sevoflurane is broadly applied in pediatric anesthesia for its quick action, hemodynamic stability, low hepatotoxicity, relative lack of airway irritation, and rapid emergence from anesthesia. However, sevoflurane anesthesia in children correlates with a higher prevalence of emergence agitation (EA) [2]. EA is a self-limited condition of psychomotor excitement in the process of awakening from general anesthesia, which is limited to the emergence period when consciousness recovery sharply differs from other psychiatric states after surgery [3]. The chief clinical manifestations of EA in children include moaning, wiggling, crying, restlessness, incoherent speech, and disorientation, as well as inability to be comforted and to recognize people or objects [4,5]. Therefore, it is imperative to select suitable and targeted medicines according to the personal condition of the patient to better prevent the occurrence of EA.
Several types of drugs are applied for preventing EA from sevoflurane anesthesia in children, such as the use of opioids, benzodiazepines, α2 agonists, intravenous anesthetics, non-opioid analgesics, and short-acting midazolam [6]. Dexmedetomidine (Dex) is defined as a highly selective α2-adrenoceptor agonist, which uniquely retains respiratory drive, generates arousable sedation, and decreases the incidence and duration of delirium after surgery [7,8]. Dex produces both sympatholytic and dose-dependent analgesic and sedative effects, and weakens neuroendocrine and hemodynamic responses associated with surgery and anesthesia [9]. Evidence has shown that Dex significantly decreases the incidence of EA of pediatric patients after ophthalmologic surgery under sevoflurane anesthesia [10]. Remifentanil is a potent opioid with strong, ultra-short action and fast action rate, which is used by anesthesiologists as an adjunct to total intravenous anesthesia because of its ease of dose adjustment, rapid offset, and synergistic effects with propofol [11]. Additionally, remifentanil has been utilized as an adjuvant to general anesthesia because of its analgesic influence diminishing intraoperative analgesia, its intraoperative antihypertensive effect, and subsequent reduction of intraoperative blood loss [12,13]. Moreover, remifentanil used as an adjuvant to sevoflurane does not cause acute opioid tolerance or hyperalgesia [14]. In recent years, with the advances in surgical techniques and technology, and better recognition of the underlying physiology, anesthesia for hepatic resection has changed considerably [15]. The clinical use of these drugs has recently been revealed to reduce EA following sevoflurane anesthesia, but it has adverse effects of postoperative vomiting, respiratory depression, and opioid-evoked hyperalgesia [16]. In the present study, we assessed the effect of Dex combined with remifentanil on EA during awakening from sevoflurane anesthesia for pediatric liver surgery.
Material and methods
ETHICS STATEMENT:
The study was approved by the Medical Ethics Committee of our hospital (approval number: 20201029), and patients and their families were informed of the study and signed consent forms.
GENERAL DATA:
Sixty children who underwent liver surgery in our hospital were recruited in this prospective study and were randomly allocated into group A and group B using a computer-generated random number list (n=30 cases). The general data on age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) grading, and type of liver mass between the 2 groups of children showed no significant differences between groups (P>0.05; Table 1) and were comparable.
Inclusion criteria were age 1–6 years, ASA grade I or II, need to perform surgical removal of liver masses, and met relevant surgical indications.
Exclusion criteria were upper respiratory tract infection within the past 3 weeks; cognitive developmental abnormalities or dysplasia, contraindications to the medications used, respiratory diseases, associated disorders that cause agitation such as epileptic seizures and schizophrenia, cardiac or renal dysfunction, and incomplete clinical data.
PREPARATION FOR ANESTHESIA:
Children in both groups were routinely fasted for 6 h and liquid-fasted for 2 h before the operation. All children had a venous access established before admission to the operating room and were continuously dripped with glucose solution (Hengrui Pharmaceuticals, Jiangsu, China). Heart rate (HR), blood oxygen saturation (SpO2), electrocardiogram (ECG), and electroencephalographic bispectral index (BIS) were routinely monitored.
INDUCTION OF ANESTHESIA:
At the end of monitoring, the child inhaled 8% sevoflurane (Baxter Healthcare Corporation, Guayama, Puerto Rico, USA) through a mask, and was injected intravenously with 2 μg/kg fentanyl citrate (Yichang Humanwell Pharmaceutical, Hubei, China) and 0.1 mg/kg cisatracurium (Hengrui Pharmaceuticals). After loss of consciousness, the child was ventilated with oxygen denaturation through a mask with pressurized ventilation, and endotracheal intubation was performed. The anesthesia machine was connected, mechanical ventilation was used, and respiratory parameters were adjusted to oxygen flow (2 L/min), tidal volume (Vt) (6–8 mL/kg), respiratory ratio (1: 1.5), respiratory rate (15–20 breaths/min), and partial pressure of end-tidal carbon dioxide (PETCO2: 35–40 mmHg).
INTRAOPERATIVE MAINTENANCE:
After correctly connecting all the monitoring equipment, children in group B were first injected intravenously with a 1 μg/kg loading dose of dexmedetomidine hydrochloride (Sichuan Guorui Pharmaceutical Co., Ltd., Sichuan, China), and then dexmedetomidine was continuously pumped at a rate of 0.2–0.4 μg/kg·h. In group A, saline was continuously pumped intravenously in an equal volume. Then, the child was maintained under anesthesia with intraoperative inhalation of 2% sevoflurane and intravenous infusion of 0.2 μg/kg/mim remifentanil hydrochloride (Yichang Humanwell Pharmaceutical). The child’s hemodynamic stability was maintained, blood pressure and HR were maintained within the basal values of ±20%, and BIS values were maintained at 40–60. An intraoperative warming blanket was used, and the body temperature was maintained at 36.0–37.5°C.
POSTOPERATIVE AWAKENING:
At the end of the operation, drug administration was stopped and the children in both groups were admitted to the post-anesthesia care unit (PACU). When the child resumed spontaneous and regular respiration, tidal volume was >6 ml/kg, and SpO2 was ≥95% and lasted for >1 min, the endotracheal tube was removed after suctioning the oral and airway secretions.
OBSERVATIONAL INDICATORS:
MAP and HR were recorded before induction of anesthesia (T0), immediately after tracheal intubation (T1), at the end of surgery (T2), immediately after tracheal extubation (T3), and 5 min after tracheal extubation (T4) in the 2 groups of children.
Sedation, agitation, and pain level were assessed using Aono’s score [17], Craner’s Pediatric Anesthesia Emergence Delirium (PAED) [18], and Children’s and Infants’ Postoperative Pain Scale (CHIPPS) [19] for children in both groups at 0 min, 15 min, and 30 min of admission to the PACU, respectively. (1) Aono’s score was employed to evaluate the child’s behavioral state: 1 for quiet; 2 for not quiet, but comforting; 3 for not quiet, moderately agitated, or disturbed; and 4 for belligerent, excitable, or disoriented. (2) The PAED scored the degree of agitation on a 5-item scale that included obeying commands and being communicative, behavior being purposeful, attention to changes in the surroundings, inability to be soothed by crying, and inability to be comforted. Each item was divided into 5 grades, of which the first 3 indicators were scored once: none (4 points), poor (3 points), moderate (2 points), good (1 point), and very good (0 point). The latter 2 indicators were scored in the following order: none (0 point), mild (1 point), moderate (2 points), severe (3 points), and very severe (4 points). The sum of the scores was taken as the PAED score, with higher scores representing more severe agitation during the awakening period. (3) The CHIPPS score examined the pain level and was based on 5 dimensions: crying, facial expression, trunk posture, lower limb posture, and restlessness, with a range of 0–2 points for each item and a total score of 10 points. Higher scores represented higher pain levels.
The general surgical conditions of the children in both groups were recorded, including the surgery time, extubation time, awakening time, and PACU retention time.
The incidence of awakening agitation (Aono’s score ≥4 and total score >12 on the PAED scale) and adverse effects (respiratory depression, nausea, and vomiting) were recorded in both groups.
STATISTICAL METHODS:
Data analysis was conducted with SPSS 26.0 software (IBM SPSS Statistics, Chicago, IL, USA). The homogeneity of variance and normal distribution were confirmed by Levene and Kolmogorov-Smirnov (K-S) tests. Measurement data were represented by mean±standard deviation and compared by
Results
GENERAL SURGICAL CONDITIONS OF THE CHILDREN:
No significant difference was noted in the surgery time, extubation time, awakening time, and PACU retention time of children in the 2 groups (P>0.05, Table 2).
MAP AND HR AT DIFFERENT TIME POINTS:
At T0, there was no significant difference in HR and MAP levels between the 2 groups (P>0.05); HR was higher than T0 in both groups from T1 to T4 time points; MAP levels were higher than T0 time point in both groups at T1 and T3 time points; and children in group B had significantly lower HR and MAP levels at T3 and T4 time points than those in group A (P<0.05, Table 3).
AONO’S SCORE AND PAED AGITATION SCORE AT DIFFERENT TIME POINTS:
Aono’s scores and PAED agitation scores were lower at 15 min into the PACU, at 30 min into the PACU compared to entry into the PACU in both groups; the Aono’s scores and PAED agitation scores at 15 min and 30 min of admission to the PACU were lower in children in group B than in group A (P<0.05) (Table 4).
CHIPP SCORES AT DIFFERENT TIME POINTS:
Compared with the time of admission to the PACU, the CHIPP scores were reduced in both groups at 15 min and 30 min of admission to the PACU, and the CHIPP scores were lower in group B than in group A at 15 min and 30 min of admission to the PACU (P<0.05, Table 5).
INCIDENCE OF AGITATION AND ADVERSE REACTIONS DURING AWAKENING:
The incidence of agitation during postoperative anesthesia awakening was lower in children in group B in contrast to group A (P<0.05). There was no significant difference in postoperative adverse reactions between group A and group B (P>0.05) (Table 6).
Discussion
EA is an undesirable postoperative complication that can occur in patients at any age, but most commonly in children [20]. Effective EA preventive approaches consist of elimination of correctable risk factors, identification of risk factors, and application of pharmacological and non-pharmacological therapies [21]. Emergence delirium (ED) is a severe subtype of EA, which is a state of consciousness. Usually, EA and ED are used interchangeably [22]. Anesthetic adjuvants such as ketamine, remifentanil, propofol, fentanyl, and other drugs have been applied for preventing EA, but these drugs can increase sedation after anesthesia, causing slow awakening and some adverse effects [23].
In our study, 60 children who underwent liver surgery were allocated into group A (treated with placebo+remifentanil+sevoflurane) or group B (treated with Dex+remifentanil+sevoflurane). MAP and HR were recorded, sedation, agitation, and CHIPPS scores were assessed, the general surgical conditions were recorded, and the incidence of awakening agitation and adverse effects were recorded in both groups. The PAED scale is widely applied to assess ED in children after surgery or diagnostic procedures [24]. Postoperative pain must be considered when assessing ED. Dex administration can be accompanied by a reduction in HR and blood pressure [25]. We found that Dex combined with remifentanil significantly relieves EA during awakening from sevoflurane anesthesia for pediatric liver surgery, improves hemodynamic levels and postoperative pain, and has fewer adverse effects.
Dex has been reported to diminish the risk of ED and EA in pediatric patients [26]. The clinical effect of titration of Dex doses is particularly crucial after liver transplantation in children because the weight-based administration may be poorly correlated with plasma concentrations [27]. Intraoperative low-dose Dex administration is associated with reduced incidence of moderate-to-severe hepatic ischemia-reperfusion injury in pediatric deceased liver transplantation [28]. Specifically, Dex can restrain the sympathetic nerve activity, thereby decreasing blood pressure and HR, maintaining hemodynamic stability during anesthesia, and possessing the functions of analgesia, sedation, anti-anxiety, and suppression of sympathetic nerve excitement [23,29]. A meta-analysis found that Dex reduces the incidence of ED in children under sevoflurane anesthesia, and its use reduces postoperative pain, thus diminishing the adverse effects of opioid administration [30]. Shi et al reported that Dex administration can decrease the ED incidence post-tonsillectomy under sevoflurane anesthesia and can be utilized to safely avoid postoperative behavior changes [31]. These articles validate the positive functions of Dex in mitigating EA. Remifentanil has multiple additional clinical benefits, such as rapid onset, lack of accumulation, and very short context-sensitive half-time [32,33]. Continuous intraoperative infusion of remifentanil effectively decreases EA incidence without clinically marked hemodynamic deterioration or delayed recovery time in children receiving ophthalmic surgery under sevoflurane anesthesia [34]. Choi et al have also supported that remifentanil reduces EA in children aged 3–9 years undergoing strabismus surgery under sevoflurane anesthesia [34]. These articles verify the positive functions of remifentanil in mitigating EA.
Moreover, our study highlighted the combined effect of Dex and remifentanil in EA during awakening from sevoflurane anesthesia for pediatric liver surgery. As previously reported, target-controlled infusion of remifentanil and a single dose of Dex can reduce hemodynamic stimulation and airway response during recovery from anesthesia [35]. For gastric sleeve bariatric surgery patients, the intraoperative use of Dex or remifentanil as adjuvant to general anesthesia is a suitable way to achieve hemodynamic control and improve perioperative outcomes [36]. Furthermore, combined administration of remifentanil with Dex under sevoflurane anesthesia has acceptable oxygenation, cardiovascular effects, and acid-base balance, and does not impede renal function [37]. These studies all agree with our results.
Conclusions
In conclusion, we found that, in pediatric liver surgery, the use of Dex+remifentanil+sevoflurane anesthesia can reduce the incidence of EA during the awakening period, stabilize hemodynamic levels, and relieve postoperative pain, and has fewer postoperative adverse effects, and it warrants clinical application. This study provides some guidelines for the clinical mitigation of EA during awakening from sevoflurane anesthesia for pediatric liver surgery. However, we did not calculate the sample size, which is a limitation of this study, and studies with larger samples are needed to verify our findings.
Tables
Table 1. Comparison of general data in the 2 group.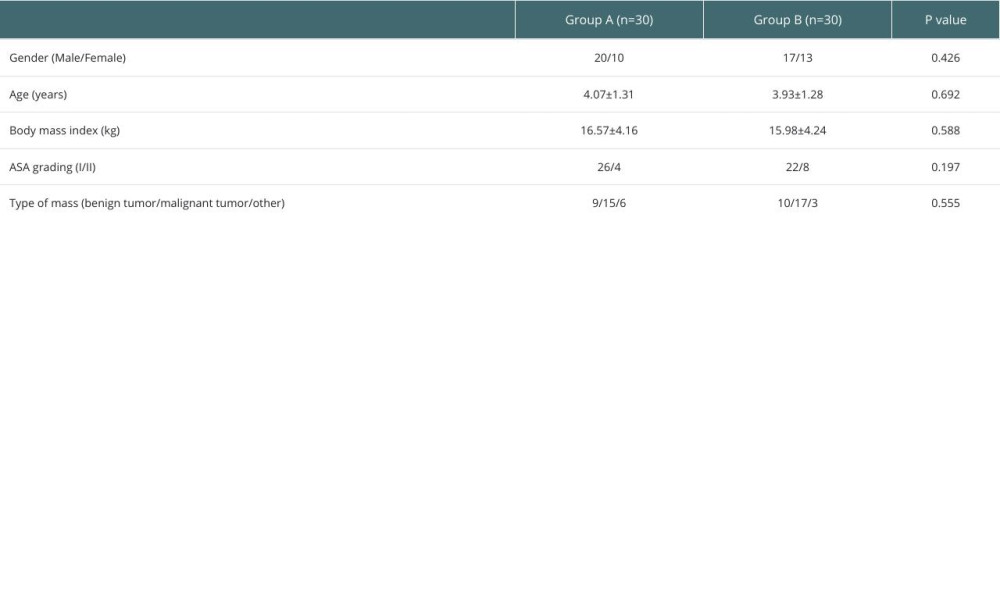 Table 2. Comparison of the general conditions of children in the 2 groups.
Table 2. Comparison of the general conditions of children in the 2 groups.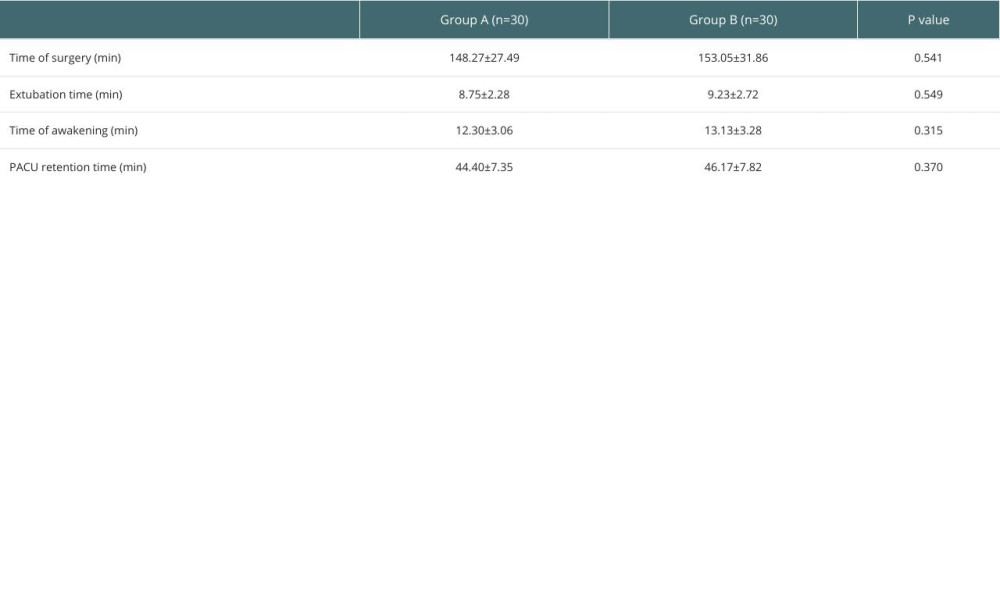 Table 3. Comparison of MAP and HR at different time points in the 2 groups.
Table 3. Comparison of MAP and HR at different time points in the 2 groups.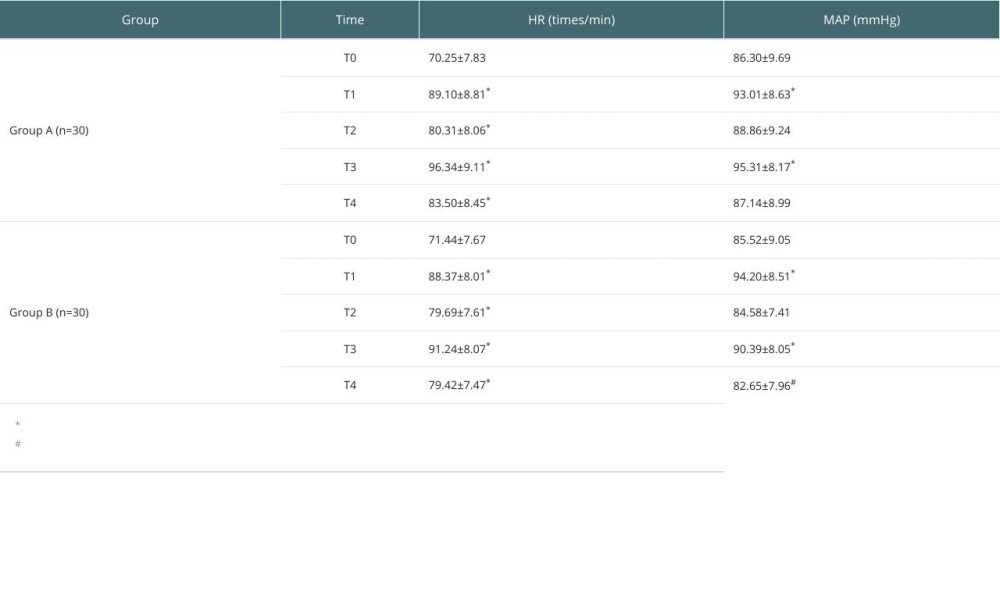 Table 4. Comparison of Aono’s score and PAED agitation score at different time points between the 2 groups of children.
Table 4. Comparison of Aono’s score and PAED agitation score at different time points between the 2 groups of children.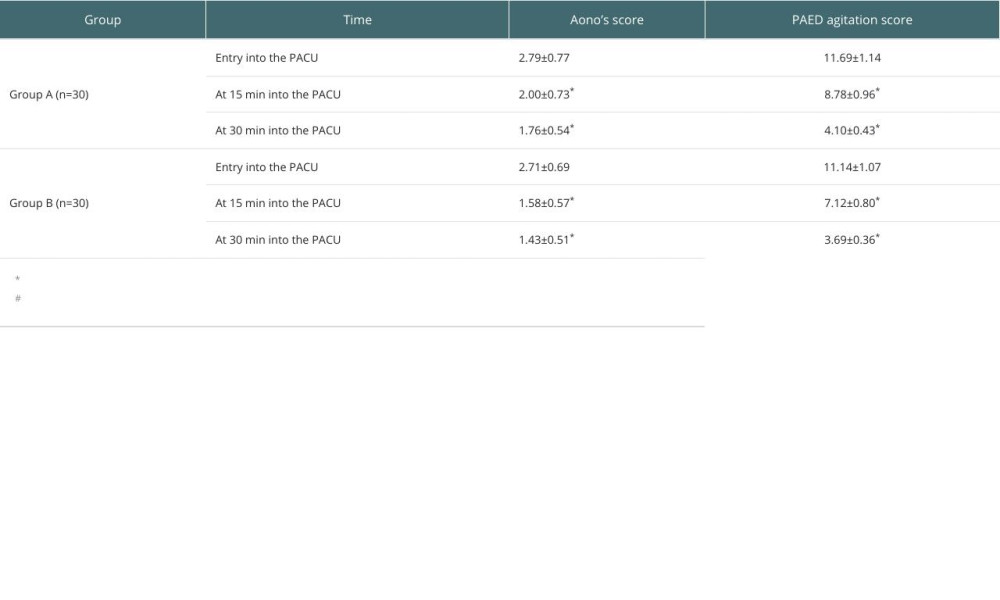 Table 5. Comparison of postoperative pain levels between the 2 groups of children.
Table 5. Comparison of postoperative pain levels between the 2 groups of children.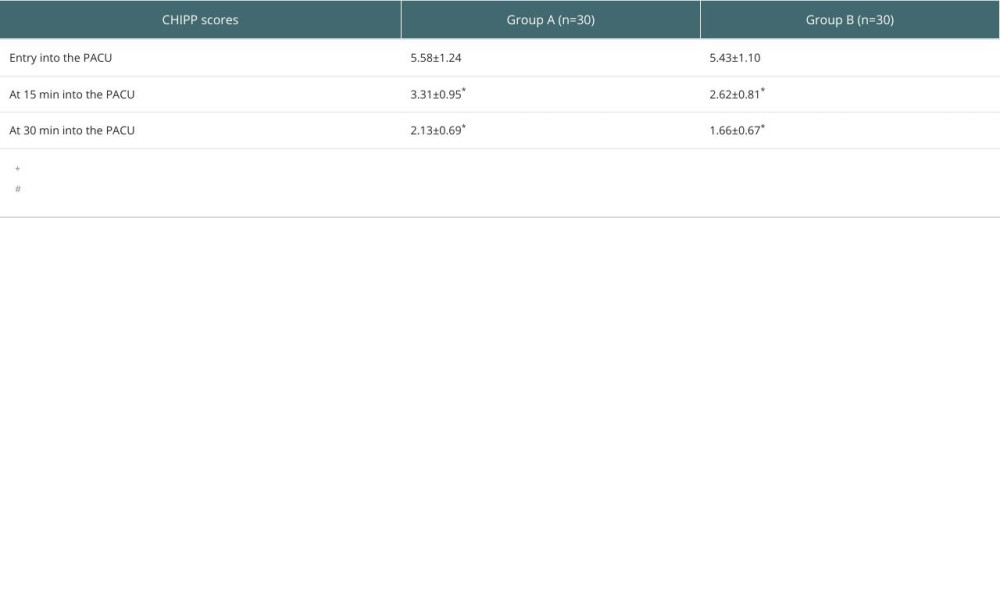 Table 6. Comparison of the incidence of agitation and adverse reactions during awakening in 2 groups of children.
Table 6. Comparison of the incidence of agitation and adverse reactions during awakening in 2 groups of children.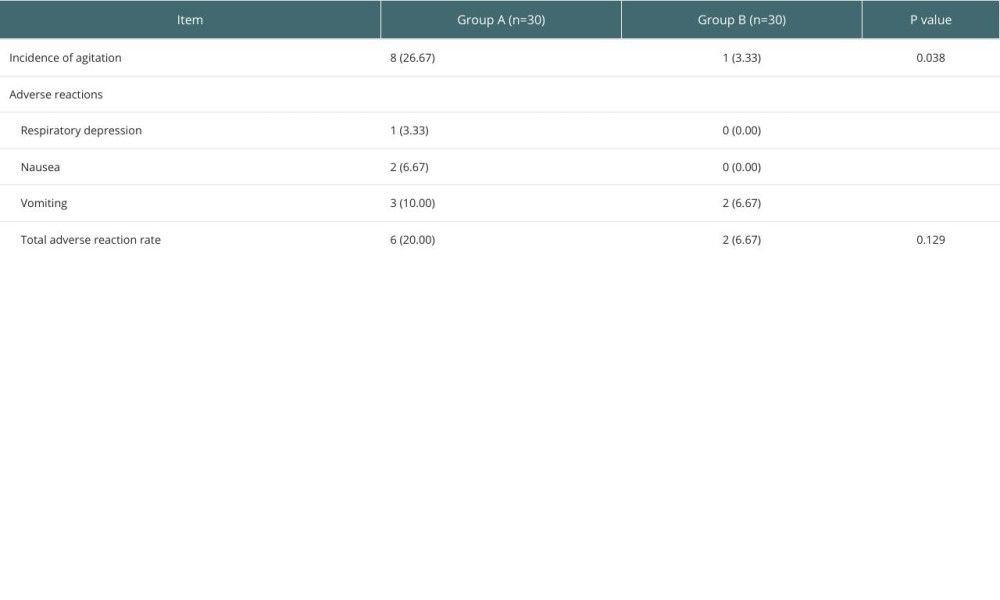
References
1. Sultana SP, Saikia D, Dey S, Fentanyl versus dexmedetomidine for the prevention of emergence agitation in children after sevoflurane anaesthesia: A comparative clinical study: Cureus, 2022; 14(8); e28587
2. Tang Y, Song Y, Tian W, A systematic review and meta-analysis on the efficacy and safety of dexmedetomidine combined with sevoflurane anesthesia on emergence agitation in children: Transl Pediatr, 2022; 11(7); 1156-70
3. Tolly B, Waly A, Peterson G, Adult emergence agitation: A veteran-focused narrative review: Anesth Analg, 2021; 132(2); 353-64
4. Ali MA, Abdellatif AA, Prevention of sevoflurane related emergence agitation in children undergoing adenotonsillectomy: A comparison of dexmedetomidine and propofol: Saudi J Anaesth, 2013; 7(3); 296-300
5. Xu J, Deng XM, Wei LX, Effects of two intranasal dexmedetomidine doses as premedication on sevoflurane EC(50) for successful laryngeal mask airway placement in children: Zhongguo Yi Xue Ke Xue Yuan Xue Bao, 2016; 38(6); 627-31
6. Kawai M, Kurata S, Sanuki T, The effect of midazolam administration for the prevention of emergence agitation in pediatric patients with extreme fear and non-cooperation undergoing dental treatment under sevoflurane anesthesia, a double-blind, randomized study: Drug Des Devel Ther, 2019; 13; 1729-37
7. Colin PJ, Hannivoort LN, Eleveld DJ, Dexmedetomidine pharmacokinetic-pharmacodynamic modelling in healthy volunteers: 1. Influence of arousal on bispectral index and sedation: Br J Anaesth, 2017; 119(2); 200-10
8. Djaiani G, Silverton N, Fedorko L, Dexmedetomidine versus propofol sedation reduces delirium after cardiac surgery: A randomized controlled trial: Anesthesiology, 2016; 124(2); 362-68
9. Tarikci Kilic E, Aydin G, Effects of dexmedetomidine infusion during spinal anesthesia on hemodynamics and sedation: Libyan J Med, 2018; 13(1); 1436845
10. Wu XL, Peng B, Shan CJ, The influence of dexmedetomidine on the emergence agitation of pediatric patients after the operations of sense organs under general anesthesia using sevoflurane: Minerva Pediatr (Torino), 2022; 74(2); 144-50
11. Oriby ME, Elrashidy A, Comparative effects of total intravenous anesthesia with propofol and remifentanil versus inhalational sevoflurane with dexmedetomidine on emergence delirium in children undergoing strabismus surgery: Anesth Pain Med, 2021; 11(1); e109048
12. Renaud-Roy E, Stockle PA, Maximos S, Correlation between incremental remifentanil doses and the Nociception Level (NOL) index response after intraoperative noxious stimuli: Can J Anaesth, 2019; 66(9); 1049-61
13. Kawano H, Manabe S, Matsumoto T, Comparison of intraoperative blood loss during spinal surgery using either remifentanil or fentanyl as an adjuvant to general anesthesia: BMC Anesthesiol, 2013; 13(1); 46
14. Yeom JH, Kim KH, Chon MS, Remifentanil used as adjuvant in general anesthesia for spinal fusion does not exhibit acute opioid tolerance: Korean J Anesthesiol, 2012; 63(2); 103-7
15. Jacob R, Anaesthesia for biliary atresia and hepatectomy in paediatrics: Indian J Anaesth, 2012; 56(5); 479-84
16. Baek J, Park SJ, Kim JO, The effects of remifentanil and fentanyl on emergence agitation in pediatric strabismus surgery: Children (Basel), 2022; 9(5); 606
17. Bajwa SA, Costi D, Cyna AM, A comparison of emergence delirium scales following general anesthesia in children: Paediatr Anaesth, 2010; 20(8); 704-11
18. Ringblom J, Wahlin I, Proczkowska M, A psychometric evaluation of the Pediatric Anesthesia Emergence Delirium scale: Paediatr Anaesth, 2018; 28(4); 332-37
19. Alves MM, Carvalho PR, Wagner MB, Cross-validation of the children’s and infants’ postoperative pain scale in Brazilian Children: Pain Pract, 2008; 8(3); 171-76
20. Wei B, Feng Y, Chen W, Risk factors for emergence agitation in adults after general anesthesia: A systematic review and meta-analysis: Acta Anaesthesiol Scand, 2021; 65(6); 719-29
21. Lee SJ, Sung TY, Emergence agitation: Current knowledge and unresolved questions: Korean J Anesthesiol, 2020; 73(6); 471-85
22. Mason KP, Paediatric emergence delirium: A comprehensive review and interpretation of the literature: Br J Anaesth, 2017; 118(3); 335-43
23. Tan D, Xia H, Sun S, Effect of ancillary drugs on sevoflurane related emergence agitation in children undergoing ophthalmic surgery: A Bayesian network meta-analysis: BMC Anesthesiol, 2019; 19(1); 138
24. Sakai EM, Connolly LA, Klauck JA, Inhalation anesthesiology and volatile liquid anesthetics: Focus on isoflurane, desflurane, and sevoflurane: Pharmacotherapy, 2005; 25(12); 1773-88
25. Aouad MT, Zeeni C, Al Nawwar R, Dexmedetomidine for improved quality of emergence from general anesthesia: A dose-finding study: Anesth Analg, 2019; 129(6); 1504-11
26. Urits I, Peck J, Giacomazzi S, Emergence delirium in perioperative pediatric care: A review of current evidence and new directions: Adv Ther, 2020; 37(5); 1897-909
27. Damian MA, Hammer GB, Elkomy MH, Pharmacokinetics of dexmedetomidine in infants and children after orthotopic liver transplantation: Anesth Analg, 2020; 130(1); 209-16
28. Zhang L, Li N, Cui LL, Intraoperative low-dose dexmedetomidine administration associated with reduced hepatic ischemia-reperfusion injury in pediatric deceased liver transplantation: A retrospective cohort study: Ann Transplant, 2021; 26; e933354
29. Sun M, Dong Y, Li M, Dexmedetomidine and clonidine attenuate sevoflurane-induced tau phosphorylation and cognitive impairment in young mice via alpha-2 adrenergic receptor: Anesth Analg, 2021; 132(3); 878-89
30. Zhu M, Wang H, Zhu A, Meta-analysis of dexmedetomidine on emergence agitation and recovery profiles in children after sevoflurane anesthesia: Different administration and different dosage: PLoS One, 2015; 10(4); e0123728
31. Shi M, Miao S, Gu T, Dexmedetomidine for the prevention of emergence delirium and postoperative behavioral changes in pediatric patients with sevoflurane anesthesia: A double-blind, randomized trial: Drug Des Devel Ther, 2019; 13; 897-905
32. Dong YX, Meng LX, Wang Y, The effect of remifentanil on the incidence of agitation on emergence from sevoflurane anaesthesia in children undergoing adenotonsillectomy: Anaesth Intensive Care, 2010; 38(4); 718-22
33. Davis PJ, Finkel JC, Orr RJ, A randomized, double-blinded study of remifentanil versus fentanyl for tonsillectomy and adenoidectomy surgery in pediatric ambulatory surgical patients: Anesth Analg, 2000; 90(4); 863-71
34. Choi YH, Kim KM, Lee SK, Effects of remifentanil and remifentanil-alfentanil administration on emergence agitation after brief ophthalmic surgery in children: BMC Anesthesiol, 2016; 16(1); 50
35. Park JS, Kim KJ, Lee JH, A randomized comparison of remifentanil target-controlled infusion versus dexmedetomidine single-dose administration: A better method for smooth recovery from general sevoflurane anesthesia: Am J Ther, 2016; 23(3); e690-96
36. Hamed JME, Refaat HSM, Al-Wadaani H, Dexmedetomidine compared to remifentanil infusion as adjuvant to sevoflurane anesthesia during laparoscopic sleeve gastrectomy: Anesth Essays Res, 2019; 13(4); 636-42
37. Akashi NY, Murahata M, Hosokawa , Cardiovascular and renal effects of constant rate infusions of remifentanil, dexmedetomidine and their combination in dogs anesthetized with sevoflurane: J Vet Med Sci, 2021; 83(2); 285-96
Tables
 Table 1. Comparison of general data in the 2 group.
Table 1. Comparison of general data in the 2 group. Table 2. Comparison of the general conditions of children in the 2 groups.
Table 2. Comparison of the general conditions of children in the 2 groups. Table 3. Comparison of MAP and HR at different time points in the 2 groups.
Table 3. Comparison of MAP and HR at different time points in the 2 groups. Table 4. Comparison of Aono’s score and PAED agitation score at different time points between the 2 groups of children.
Table 4. Comparison of Aono’s score and PAED agitation score at different time points between the 2 groups of children. Table 5. Comparison of postoperative pain levels between the 2 groups of children.
Table 5. Comparison of postoperative pain levels between the 2 groups of children. Table 6. Comparison of the incidence of agitation and adverse reactions during awakening in 2 groups of children.
Table 6. Comparison of the incidence of agitation and adverse reactions during awakening in 2 groups of children. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








