24 May 2022: Review Paper
A Systematic Review of the Literature on Chronic Kidney Disease Following Liver Transplantation
Hitomi Miyata1ADE*, Yoshiaki Morita2ADE, Anil Kumar3DEDOI: 10.12659/AOT.935170
Ann Transplant 2022; 27:e935170
Abstract
ABSTRACT: Chronic kidney disease (CKD) is a serious comorbidity affecting liver transplant recipients (LTRs). Calcineurin inhibitor dosing minimization protocols and everolimus use purportedly increased from 2010, potentially impacting CKD development. This systematic literature review was designed to identify CKD incidence in adult LTRs, focusing on studies published from 2010 onwards. PubMed, Embase, and the Cochrane Database of Systematic Reviews were searched for papers reporting renal function (glomerular filtration rate [GFR]; estimated GFR [eGFR] or Chronic Kidney Disease Epidemiology Collaboration) for adult LTRs ≥6 months after transplantation. Primary outcome: renal function ≥6 months after transplantation, with CKD stage. Bias was assessed using the Cochrane Collaboration bias tool and by reviewing disclosures/industry funding. Of 3960 records identified, 14 publications were included. In at least 1 study arm, mean GFR/eGFR remained stable/improved temporally in 4 and decreased in 8 publications. Where GFR/eGFR decreased, mean eGFR was 71.4-119.6 mL/min/1.73 m² (CKD stage 2-stage 1) across studies at baseline, and was 77.2 and 79.1 mL/min/1.73 m² (stage 2) at 12 months. The proportion of patients with CKD increased between baseline and follow-up; 23.2-36.8% of patients had CKD stage 3a or higher at 12 months (2 studies). Rates ranged from 85.7-100% (6 months) for patient survival, 81.0% (12 months) to 100.0% (17 months) for graft survival, and 0-40% (12 months) for acute rejection. Most studies carried risk of bias. Evidence of temporal renal function decline highlights the need for continuous renal monitoring of LTRs, particularly regarding potential CKD development/progression.
Keywords: Liver Transplantation, Renal Insufficiency, Chronic, review, Glomerular Filtration Rate, Adult, Glomerular Filtration Rate, Graft Survival, Humans
Background
Chronic kidney disease (CKD) is one of the main comorbidities affecting liver transplant recipients [1]. Kidney disease is diagnosed in 20–25% of patients with liver disease [1] and can be due to pathological conditions affecting both the kidney and the liver (eg, sarcoidosis or diabetes), or a complication of certain liver diseases, such as hepatitis B, hepatitis C, or alcoholic cirrhosis [1]. The liver allocation Model for End-Stage Liver Disease (MELD) score was introduced in 2002, and includes serum creatinine levels that reflect the risk of end-stage renal disease in the post-liver transplant period [2]. Additionally, liver transplant recipients can develop de novo CKD after transplantation, and this is associated with poor long-term survival and increased healthcare costs compared with liver recipients without CKD and the general population [3–5].
The Organ Procurement Transplant Network database shows that, between 1997 and 2008, 1090 (0.7%) of 157 086 kidney transplantations performed in the United States were carried out in patients with a previous liver transplant [6]. It is, therefore, important to identify the risk factors for kidney damage after liver transplantation and the incidence of de novo kidney injury after liver transplantation, and its impact on long-term patient survival. For example, a retrospective cohort study of liver transplant recipients identified female sex and renal risk index score as risk factors for the development of post-transplantation stage 4–5 de novo CKD [7]. The widespread use of calcineurin inhibitors (CNIs) in post-transplantation immunosuppression regimens has also been associated with the development of end-stage renal disease after liver transplantation [8]. However, it is unclear whether this remains the case following the introduction of CNI dosing minimization strategies [7,9,10]. Indeed, from approximately 2010 onwards, the target blood concentration of CNIs may have decreased [9,10], and the use of once-daily tacrolimus (Advagraf) may have increased following its approval for use after liver transplantation in Europe (2007) and in the United States (2013). Furthermore, use of post-transplantation immunosuppression with mycophenolate mofetil (MMF) plus everolimus purportedly increased following approval of everolimus for the prevention of graft rejection in liver transplantation in Europe (2012) and in the United States (2013) [11]. All of these factors might reduce the development of CKD.
To the best of our knowledge, there are no recent systematic literature reviews that assess the risk of developing de novo CKD after liver transplantation in the context of modern immunosuppressive regimens and seek to determine its relationship with transplant outcomes. This systematic literature review focusing on studies published from 2010 onwards was developed to identify the incidence rate of CKD in adult liver transplant recipients, based on renal function at least 6 months after transplantation and irrespective of immunosuppressive regimen used. Progression of CKD after liver transplantation, and acute rejection rate, patient survival, and graft survival were also assessed.
Protocol and Registration
This study was conducted according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) [12,13]. The protocol can be accessed on the National Institute of Health Research International prospective register of systematic reviews (PROSPERO; CRD42020210457; https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42020210457).
Eligibility Criteria and Study Selection Process
The selection criteria for the systematic literature review were based on the Population, Exposure, Comparator, Outcome and Study type (PECOS) framework (Table 1). Randomized controlled clinical trials, quasi- and non-randomized clinical studies, observational studies (cohort [including registry], case-control, or cross-sectional studies), and systematic literature reviews in liver transplantation were eligible for inclusion if they reported renal function data (glomerular filtration rate [GFR] by iohexol clearance; estimated GFR [eGFR] by Modified Diet in Renal Disease [MDRD] or Chronic Kidney Disease Epidemiology Collaboration [CKD-EPI]) for adult (aged ≥18 years) liver transplant recipients (de novo or retransplantation) ≥6 months after liver transplantation. Studies were included if patients received a previous solid organ transplant. However, patients receiving combined liver and kidney transplantation were excluded. Other exclusion criteria were receipt of maintenance dialysis prior to the current transplant (CKD stage 5d). Where a subset of patients in a study were receiving maintenance dialysis prior to the current transplant, the whole study was excluded unless it was possible to exclude data for patients who received prior dialysis. However, studies were included when it was not stated if patients were receiving maintenance dialysis prior to the current transplant, or if it was not specified when dialysis occurred. Studies reporting adult and pediatric data were excluded unless adult data alone could be extracted from the paper.
Retrospective studies were excluded if all transplantations were performed before 2010. When transplantations were conducted both before and after 2010, the study was included if data for only transplantations performed after 2010 could be extracted. Systematic literature reviews were excluded when all studies in the review were published before 2010.
Information Sources and Search Strategy
A systematic literature search of PubMed, Embase, and the Cochrane Database of Systematic Reviews was performed. The search terms and conditions used are presented in Table 2. To minimize the risk of excluding relevant citations, the bibliography of each included study was also manually searched by 2 people. Database searches were performed on December 2, 2020, and were limited to full papers published on or after January 1, 2010. This cut-off was applied as, from around this date: 1) the target blood concentration of CNIs may have decreased following introduction of dosing minimization strategies [9,10]; 2) use of once-daily tacrolimus may have increased; and 3) the combined use of MMF and everolimus as post-transplant immunosuppression purportedly increased [11], all of which might impact the development of CKD. Only full publications in the English language were included.
Retrieved articles were individually reviewed for eligibility by 2 people; where there was disagreement, the article was included for author review. All articles were confirmed by all authors. Discrepancies were addressed and solved by consensus among authors following review of the articles.
Data Collection Process and Summary Measures
Structured data collection spreadsheets were used to collate the key information from each study. One person extracted the data into the spreadsheets, which were then checked by a second person. All data are presented descriptively in summary tables.
Data Items and Outcomes
The primary outcome was renal function (GFR by iohexol clearance; eGFR by MDRD or CKD-EPI) ≥6 months after liver transplantation, presented with CKD stage. CKD was defined according to the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF-K/DOQI) and the Kidney Disease Improving Global Outcomes (KDIGO) CKD guidelines [14,15]. The classification of CKD was based on GFR (iohexol clearance) or eGFR (abbreviated MDRD formula or CKD-EPI equation) stage in CKD: stage 1: normal or high GFR/eGFR (≥90 mL/min/1.73 m2); stage 2: mild reduction in GFR/eGFR (60–89 mL/min/1.73 m2); stage 3a: mild to moderate reduction in GFR/eGFR (45–59 mL/min/1.73 m2); stage 3b: moderate to severe reduction in GFR/eGFR (30–44 mL/min/1.73 m2); stage 4: severe reduction in GFR/eGFR (15–29 mL/min/1.73 m2); stage 5: kidney failure (GFR/eGFR <15 mL/min/1.73 m2 or dialysis). CKD herein was defined as GFR/eGFR <60 mL/min/1.73 m2 (stage 3a or higher). In the absence of evidence of kidney damage by urine abnormality, diagnostic imaging, blood, and/or pathology, neither GFR/eGFR stage 1 nor 2 fulfilled the criteria for CKD [14,15]. The progression of CKD was defined as a change from stages 1, 2, 3a, or 3b to stage 4, and from any prior stage to stage 5.
Secondary outcomes were patient survival rate, graft survival rate, and acute rejection rate.
Risk of Bias in Individual Studies
Risk of bias was assessed by 2 people at the study level, reviewing disclosures and/or industry funding. Bias of randomized controlled trials was assessed using the Cochrane Collaboration bias tool, which examines bias based on random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias [16].
Grading of Included Studies
The quality of evidence in the included studies was assessed using the Oxford Centre for Evidence-based Medicine – Levels of Evidence [17].
Study Selection
Overall, 3960 records were identified through database searching, of which 1854 had the title and abstract screened and 362 subsequently had the full text assessed for eligibility (Figure 1). Of these 362 studies, 348 articles were excluded, primarily because transplantation was conducted before 2010 (n=235) and GFR/eGFR beyond 6 months after transplantation was not reported (n=72). Fourteen studies met eligibility criteria and were included in the systematic literature review (Table 3) [7,18–30]. Of these, 11 were single-center non-randomized studies (level of evidence 3 or 4), 2 were multicenter randomized trials (level of evidence 2), and 1 was a multicenter non-randomized study (level of evidence 4). No additional papers were eligible based on searches of the citation lists in the included studies.
Duration of follow-up after liver transplantation ranged across studies from 6 months to 24 months, and Sharma et al reported last follow-up at a median of 4 years after transplantation [7] (Table 3). All studies except Yoon et al and Chauhan et al assessed treatment regimens that included tacrolimus (Table 3). Patients in the study by Yoon et al generally received CNI-based regimens, while treatment regimens were not reported by Chauhan et al [20,21].
Baseline Characteristics
Patient demographics and baseline characteristics are presented in Table 4. With few exceptions, most patients across study arms were male (up to 92.9% and 100% of participants in Chen et al and Yoon et al, respectively [21,23]). Where mean patient age was reported, this was between 48.5 and 61.5 years across studies and study arms. When information was available, ≥78.1% of patients were White; the exception was Chauhan et al, in which 36.3–43.3% of patients across study arms were White [20]. The primary diseases at baseline varied between studies and study arms (Table 4), but included alcoholism or ethanol abuse (23.1–71.4% across 3 studies [23,24,29]) hepatitis C virus infection (0–41.8% in 11 studies [7,19,20,22–26,28–30]), and hepatocellular carcinoma (2.3–63.6% in 10 studies [7,19–22,24,27–30]).
Seven studies reported baseline creatinine level. In 5 studies reporting mean±standard deviation (SD) values, these ranged from 0.8±0.3 mg/dL in Hong et al [27] to 1.45±1.6 mg/dL in the telbivudine arm of Lin et al [18]. Jochmans et al reported baseline median (range) creatinine levels of 0.77 (0.62–0.98) mg/dL and 0.86 (0.74–1.04) mg/dL in patients with and without post-transplantation acute kidney injury (AKI), respectively [28]. Sharma et al reported baseline median (interquartile range [IQR]) creatinine levels of 0.8 (0.6–1.0) mg/dL [7].
Immunosuppressive Treatment
Available data for immunosuppressant dose and trough levels are presented in Supplementary Table 1, but are not reported here because data were limited, preventing any conclusions from being drawn.
Renal Function
Renal function at baseline and during follow-up varied markedly between studies and treatment regimens (Table 5). From baseline values, mean GFR/eGFR remained stable or improved over time in at least 1 study arm from 4 publications [18,22,23,29]. Mean±SD eGFR increased numerically from baseline in patients receiving tacrolimus+MMF+steroid+telbivudine (from 77.3 to 88.9 mL/min at 12 months [means within CKD stage 2] [18]) and in non-betel nut chewers receiving once-daily tacrolimus (from 72.2±21.9 to 77.8 mL/min/1.73 m2 at 17 months [means within CKD stage 2] [23]). Dopazo et al also reported a numerical increase in mean±SD eGFR between baseline and 12 months in patients with renal dysfunction at liver transplant who received anti-human T-lymphocyte globulin (ATG) induction+tacrolimus+steroids (49±9 vs 58±16 mL/min/1.73 m2, respectively; means within CKD stage 3a) [29]. In patients receiving tacrolimus+enteric-coated mycophenolate sodium (EC-MPS) followed by everolimus, mean±SD eGFR was 91.4±36.6 and 95.8±27.7 mL/min/1.73 m2 (means within CKD stage 1) at baseline and 6 months, respectively, which equated to a mean change of 0.1±32.6 mL/min/1.73 m2 [22].
Mean GFR/eGFR decreased numerically over time in at least 1 study arm from 6 publications where data were reported according to study arm [19,22–24,26,27]. Where mean data were available, eGFR ranged from 71.4 to 119.6 mL/min/1.73 m2 (means within CKD stage 2 to stage 1) across studies at baseline, from 76.0 to 85.2 mL/min/1.73 m2 (CKD stage 2) at 6 months [22,26,27], and was 77.2 and 79.1 mL/min/1.73 m2 (CKD stage 2) in 2 studies at 12 months [24,26]. Median data from Pascher et al at 6 months were consistent with this, irrespective of study arm (Table 5) [30]. At 24 months, Gojowy et al reported mean±SD eGFR of 76.9±21.3 mL/min/1.73 m2 (CKD stage 2) [24]. In Sharma et al, eGFR decreased from median (IQR) 96.4 (74–123) to 61 (47–74) mL/min/1.73 m2 between baseline and last follow-up (median 4 years) [7]. This equated to median renal function decreasing from CKD stage 1 to stage 2.
Jochmans et al, Levitsky et al, and Yoon et al split data according to the presence or absence of renal dysfunction (Table 5) [21,25,28]. In patients with >10% decline in eGFR from 3 months to year 1 after transplantation, mean±SD 12-month eGFR was 62.5±16.7 mL/min/1.73 m2, compared with 84.6±16.0 mL/min/1.73 m2 in patients with preserved eGFR [25]. Patients with post-transplantation AKI had numerically lower median (IQR) eGFR at 12 months than patients with normal postoperative renal function (68 [54–85] vs 77 [47–110] mL/min/1.73 m2, respectively) [28].
CKD Incidence/Prevalence and Stage
Five studies reported the prevalence of CKD (it was unclear if these were new cases after transplantation), and Lim et al reported the incidence of new-onset CKD [7,19,22,24,26,28] (Table 6). Of the 2 studies reporting the prevalence of CKD at baseline and follow-up, there was an increase in the proportion of patients with CKD [7]. Gojowy et al reported that 23/130 patients (17.7%) had CKD at the time of qualifying for liver transplantation, which increased to 39 (30.0%) and 48 (36.9%) patients with CKD by 12 and 24 months, respectively [24]. Sharma et al reported that while no patients had CKD stage 4–5 at baseline, 7.5% had stage 4–5 CKD after a median of 4 years [7].
Defining CKD as stage 3a or higher (GFR/eGFR <60 mL/min/1.73 m2), 5.4–32.6% of patients had CKD at 6 months across 3 study arms in 2 studies (Table 6) [19,22]. Across 3 study arms in 2 studies, 23.2–36.8% of patients had CKD at 12 months, with the highest proportion reported in patients with post-transplantation AKI (Table 6) [26,28].
As data were reported for entire study populations, it was not possible to ascertain progression of CKD. Furthermore, none of the eligible studies reported causes of or risk factors for CKD.
Patient and Graft Survival
Ten studies reported data for patient deaths (Table 7) [7,19,20,22,23,26–30]. The incidence of patient deaths at 6 months was generally between 0% and 3.8%, with the exception of the sotrastaurin arms of Pascher et al, in which death rates of up to 14.3% were cited (Table 7) [30]. Patient death rate at 12 months ranged between 1.7% and 22.4%, with the highest rates reported in patients with pre-transplantation CKD stage 3 or AKI [20].
Five studies reported data for graft survival (Table 8) [22,23,28–30]. Across study arms and studies, graft survival ranged between 93.5% and 100.0% at 6 months [22,30], and between 81.0% and 95.0% at 12 months, including in patients with renal dysfunction at liver transplantation [28,29]. At 17 months – the longest duration of follow-up in studies reporting graft survival – no patients receiving prolonged-release tacrolimus had graft loss in the study by Chen et al [23].
Graft Rejection
Seven studies reported biopsy-confirmed acute rejection (BCAR) rates (Table 9) [7,19,22,26,27,29,30]. BCAR rate varied across studies and study arms, ranging from 0–29.5% at 6 months [19,22,27,30], and 0–40.0% at 12 months [7,26,29]. The highest BCAR rate (40.0%, 8 patients) at 12 months was in patients who had renal dysfunction at liver transplantation [29].
Diabetes
Ten studies reported data for diabetes (Supplementary Table 2) [7,19,20,22,24–26,28–30]. At baseline, 19.0–50.0% of patients across publications and study arms had diabetes [7,19,20,22,24–26,28,29]. At 6 months, new-onset diabetes mellitus (NODM) was reported in 5 (9.4%) patients by Lim et al [19], and in 38.1–42.5% of patients across study arms by Pascher et al [30]. Lladó et al reported NODM in 19 patients (27.5%) at 12 months [26].
Risk of Bias Across Studies
Based on author disclosures and industry funding, 4 studies were assessed as being at low risk of bias in both domains, 7 as being at low risk of bias in at least 1 domain, and 5 as being at high risk of bias in at least 1 domain (Figure 2A). Using the Cochrane Collaboration bias tool for randomized controlled trials, Pascher et al and Saliba et al, which were the randomized controlled trials included in the systematic literature review, were at low risk of bias in 4 domains, and Saliba et al was at high risk of bias regarding blinding of participants and personnel and blinding of outcome assessment (Figure 2B).
Discussion
This systematic literature review included the most current evidence from 14 studies that met the inclusion criteria, evaluating the incidence of CKD in more than 1600 adults after liver transplantation over a period of 6–48 months. Renal function during follow-up varied markedly between studies and treatment regimens and, where data were available, the incidence of CKD increased between baseline and follow-up. Patient and graft survival was generally high across all studies, while the BCAR rate noticeably varied.
GFR/eGFR values remained stable or improved in at least 1 study arm in 4 studies in patients with baseline GFR/eGFR ≥60 mL/min/1.73 m2 (CKD stage 2 or lower), and improved in patients with baseline renal dysfunction and who received ATG induction+tacrolimus+steroids in 1 study [18,22,23,29]. Detailed immunosuppression regimens were reported in 2 of these publications, and both included delayed initiation of tacrolimus therapy with subsequent reduced tacrolimus dosing, which is consistent with modern renal-sparing immunosuppression regimens that have been utilized in the post-2010 era. In Saliba et al, patients received an initial regimen of basiliximab induction followed by delayed initiation of tacrolimus (initiated 3–5 days after transplantation; target trough concentration 6–10 ng/mL) and EC-MPS 720 mg twice daily before converting at week 4 after transplantation to everolimus 1 mg twice daily with reduced tacrolimus dose (tacrolimus discontinued during week 12) [22]. In Dopazo et al, patients were administered ATG 1 mg/kg induction with low-dose (0.05 mg/kg twice daily) tacrolimus, which was delayed until at least day 2 after transplantation, targeting trough levels 5–8 ng/mL during the first 3 months and <5 ng/mL thereafter. MMF was introduced on day 7 if tacrolimus levels were inadequate [29]. Graft and patient survival rates were high with both regimens, which suggests that delaying and/or reducing the dose of tacrolimus treatment may be a successful renal-sparing strategy.
Eight studies in total reported deterioration in renal function from baseline in at least 1 study arm [7,19,22–24,26,27,30]. Some of these studies reported reduced or delayed tacrolimus initiation, while others reported standard tacrolimus administration immediately postoperatively. For example, in Sharma et al, tacrolimus was initiated on postoperative day 0–1 (target trough level 6–10 ng/mL for the first 3 months, 4–8 ng/mL thereafter) with MMF 1000 mg and corticosteroids [7]. By contrast, in Saliba et al, patients received basiliximab induction with tacrolimus initiation delayed until day 3–5 after transplantation (target trough concentration 6–10 ng/mL) and EC-MPS 720 mg twice daily with or without steroids [22]. However, although 8 studies reported deterioration in renal function, mean or median GFR/eGFR values generally remained ≥60 mL/min/1.73 m2 across timepoints in all 14 studies, suggesting that the immunosuppressive regimens used supported long-term renal function. In the 3 publications that cited mean or median GFR/eGFR ≤60 mL/min/1.73 m2 (CKD stage 3a or higher) [20,21,29], the lowest GFR/eGFR was median 37.7 mL/min/1.73 m2 (CKD stage 3b). This was reported at 24 months after transplantation, and might be expected as the patients had CKD stage 3 prior to transplantation [20].
Tacrolimus minimization protocols reported in the post-2010 era have been associated with renal-sparing effects in liver transplant recipients, without compromising BCAR, graft survival and patient survival rates. For example, in the DIAMOND study, initial low-dose prolonged-release tacrolimus (0.15–0.175 mg/kg/day) plus MMF and basiliximab (without maintenance corticosteroids), with subsequent lower tacrolimus exposure over the first month, was associated with better eGFR (76.4 mL/min/1.73 m2; by MDRD-4) at week 24 versus higher-dose prolonged-release tacrolimus (67.4 mL/min/1.73 m2, p=0.001) and higher-dose, delayed prolonged-release tacrolimus (73.3 mL/min/1.73 m2, p=0.047) [10]. BCAR was also improved with initial low-dose prolonged-release tacrolimus versus other study arms, while graft and patient survival rates were comparable. The renal-sparing effects of short-term induction therapy with delayed and/or reduced introduction of CNIs continue to be examined in liver transplantation recipients. Other regimens, such as those containing belatacept or sirolimus, have been attempted with limited success [31–33]. As such, practice-based guidelines suggest that CNI dosing (typically in combination with an adjunctive non-nephrotoxic immunosuppressive agent) can be reduced to improve renal function, but that a CNI is typically required to prevent rejection within the first postoperative year [34]. Indeed, the predominant immunosuppressive regimen in adult liver recipients in the US remains the combination of tacrolimus, MMF, and steroids (62.8%), followed by tacrolimus and mycophenolate (18.6%) [35]. Our findings also support the use of tacrolimus-based regimens and suggest that tacrolimus dosing minimization strategies may be considered in liver transplant recipients.
In the papers reporting baseline data, there was evidence for a temporal increase in the incidence of CKD and higher CKD stage after liver transplantation, although it was unclear whether post-transplant CKD was newly established [7,24]. This suggests a decline in renal function over time, highlighting the need for close monitoring. At 12 months, the highest proportion of patients with CKD (stage 3a or higher) was reported in those with post-transplant AKI. This might be expected, as the development of AKI after liver transplantation has been associated with an increased risk of CKD [36,37].
Whether the incidence of CKD has decreased over time in response to modern renal-sparing immunosuppressive regimens is unclear without further evidence from large-scale studies. The liver allocation MELD score was introduced in 2002 and, therefore, the incidence of CKD increased in the post-MELD era. However, from around 2010, the use of once-daily tacrolimus may have increased, target blood concentration of CNIs may have decreased following introduction of dosing minimization strategies [9,10], and the combined use of MMF and everolimus as post-transplant immunosuppression purportedly increased [11], all of which might be expected to reduce the development of CKD compared with before 2010. Comparing the incidence of CKD before versus after 2010 is challenging due to the different definitions for CKD applied and follow-up times reported. While no directly comparable data are available from our study, in a review of CKD after liver transplantation, Fabrizi et al cited frequencies of CKD ranging between 17.6% (at 12 months) and 62.3% (at 6 years) from studies published between 2004 and 2010 [38]. In a pooled analysis of 32 830 patients from studies published between 2003 and 2020, Lim et al reported that the cumulative incidence of post-liver transplantation CKD 1 year, 3 years, and 5 years after transplantation was 31.4%, 36.7%, and 43.5%, respectively [39]. In a cohort of 221 patients undergoing liver transplantation between February 2002 and February 2007 and receiving standard tacrolimus-based triple immunosuppression, Sharma et al reported that 31/207 (15%) patients not on renal replacement therapy (RRT) at transplantation developed CKD after a median of 2.6 years (defined as eGFR <30 mL/min/1.73 m2 [stage 4] for ≥3 months, need for RRT, or being placed on the kidney transplantation list) [40]. By contrast, the proportion of patients with CKD stage 4–5 reported in our study (0–7.5% across studies, up to a median of 4 years after transplantation) was lower, although our definition did not include need for RRT or being placed on the kidney transplantation list. This potential reduction in CKD incidence after 2010 cannot be specifically attributed to changes in immunosuppressive regimens given the paucity of available data, although modern immunosuppression protocols are likely to play a role. Nevertheless, these data are encouraging for clinicians, given that both pre- and post-transplant CKD is independently associated with mortality, making the prevention and management of renal injury a priority in the liver transplant setting [3,41]. Indeed, 5-year graft failure rates among adult deceased donor liver transplant recipients in the US have decreased from approximately 35% in 2005 to <25% in 2013 [35], and mortality rates have decreased in parallel. In a prospective cohort study in the US, 5-year patient mortality rates decreased from 37% from 2003 and through 2009, to 23% from 2010 and through 2016 in recipients of liver grafts from older donors [42].
In our analysis, patient survival rates were generally high across studies, ranging from 96.2% to 100% at 6 months; however, an exception was the sotrastaurin-containing study arms in Pascher et al, in which 6-month survival rates as low as 85.7% were reported [30]. The high rate of death in this study was partly responsible for the termination of the clinical development of sotrastaurin for transplantation [30]. Graft survival rates were also high across the 5 studies that reported data, ranging from 81.0% at 12 months in patients with post-transplant AKI [28] to 100% with prolonged-release tacrolimus-based therapy at the longest follow-up reported for graft survival (17 months) [23]. Among the 7 studies reporting BCAR, rates varied from 0% at 6 and 12 months to 40.0% at 12 months [26,27,29]. The latter was reported in patients with baseline renal dysfunction receiving ATG induction+tacrolimus+steroids; however, graft survival rates remained high in this population (95.0%) [29].
While this literature review comprised studies with lengthy follow-up periods and from broad geographical areas, there were limitations. Only 14 studies met the strict inclusion criteria, which limited the available body of evidence. There was also disparity between the included studies, such as patient populations, treatment regimens, and methods for reporting of outcomes, making it difficult to compare data between studies. Given that not all of the publications included herein reported detailed immunosuppression dosing, it is not possible to draw conclusions regarding association of specific drugs or regimens with reduced renal function. Limited or lack of data also precluded citing data for urinary protein, hemoglobin and albumin levels, operative time, infections, and information regarding renal ultrasound. Additionally, there was generally a low level of evidence (mostly level 3 or 4), with some studies including low patient numbers. Furthermore, most studies carried some risk of bias, which would affect the potential inferences that could be made from the data. Among these 14 studies, we were unable to collect data for all aspects of interest in this setting, such as identifying causes of, and risk factors for CKD. This indicates the need for future studies to help fill this knowledge gap.
Conclusions
Among the studies reported herein, GFR/eGFR tended to remain above 60 mL/min/1.73 m2 over time, and graft and patient survival rates were generally high. However, there was evidence of a decline in renal function over time in most of the included studies. This highlights the need for continued monitoring of renal function in liver transplant recipients and vigilance regarding the potential for CKD development and progression. Delaying and/or reducing the dose of tacrolimus treatment may be a successful renal-sparing strategy. However, a CNI is typically required to prevent rejection within the first postoperative year, and standard immunosuppressive regimens including tacrolimus may also support long-term renal function.
Figures
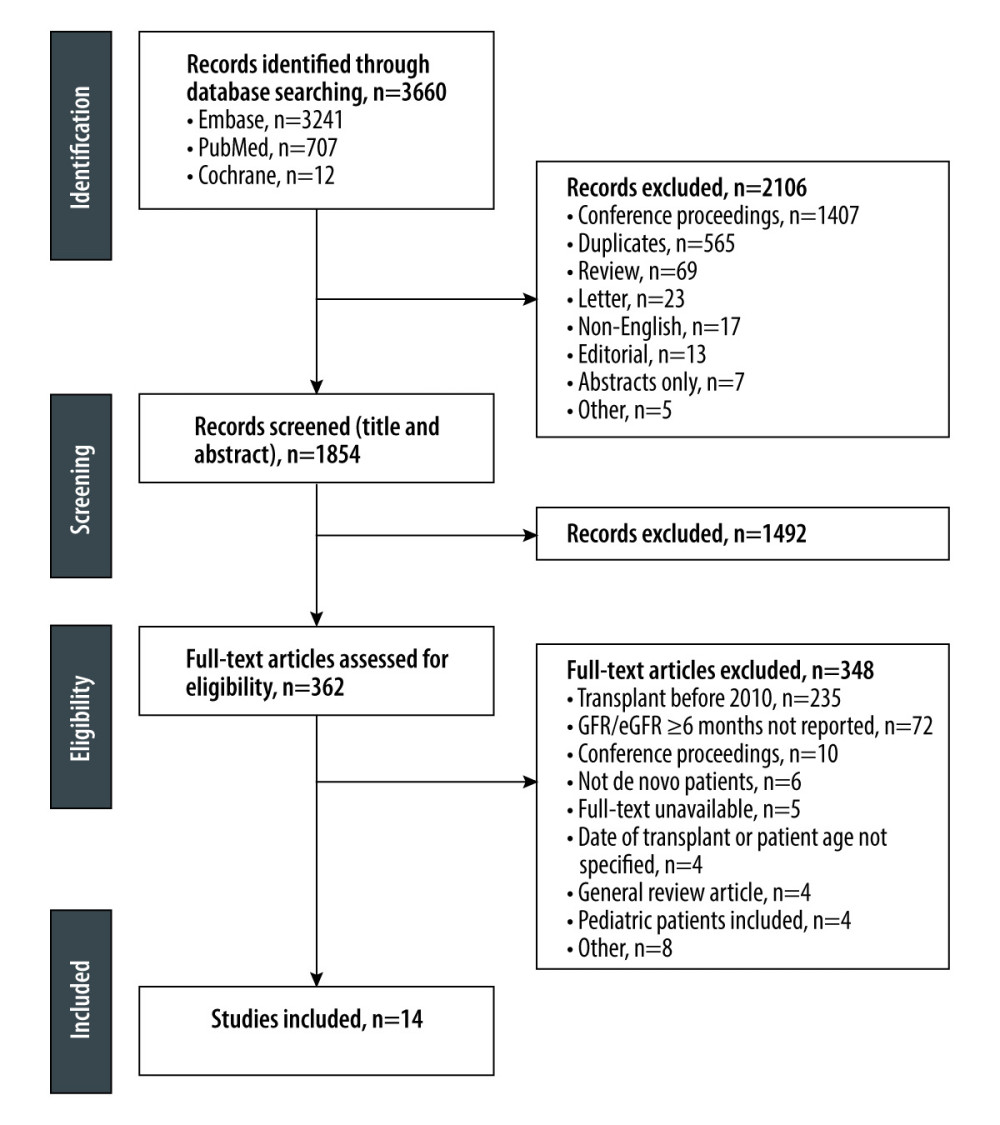 Figure 1. PRISMA diagram. eGFR – estimated glomerular filtration rate; Embase – Excerpta Medica Database; GFR – glomerular filtration rate; PRISMA – Preferred Reporting Items for Systematic reviews and Meta-Analyses.
Figure 1. PRISMA diagram. eGFR – estimated glomerular filtration rate; Embase – Excerpta Medica Database; GFR – glomerular filtration rate; PRISMA – Preferred Reporting Items for Systematic reviews and Meta-Analyses. 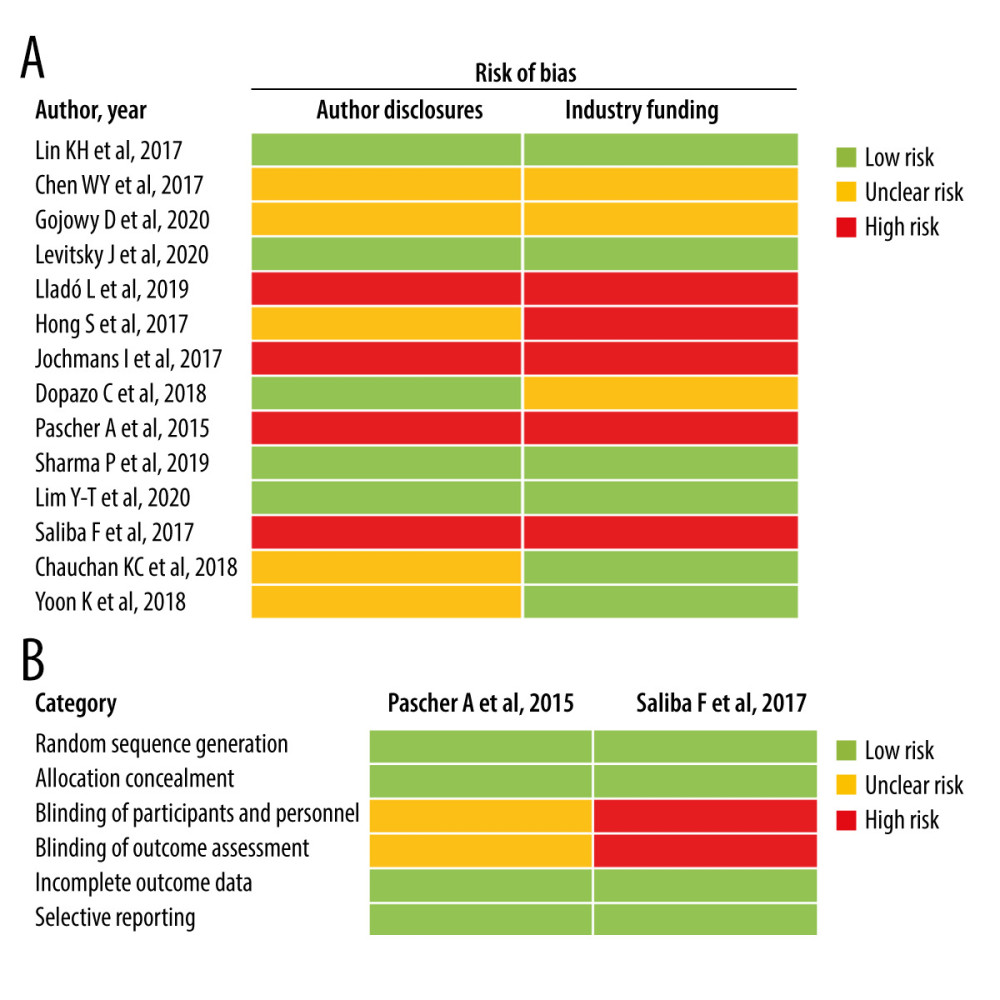 Figure 2. Risk of bias assessments for the included studies based on (A) author disclosures and industry funding and (B) using the Cochrane Collaboration bias tool for randomized controlled trials.
Figure 2. Risk of bias assessments for the included studies based on (A) author disclosures and industry funding and (B) using the Cochrane Collaboration bias tool for randomized controlled trials. Tables
Table 1. PECOS framework. Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews.
Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews.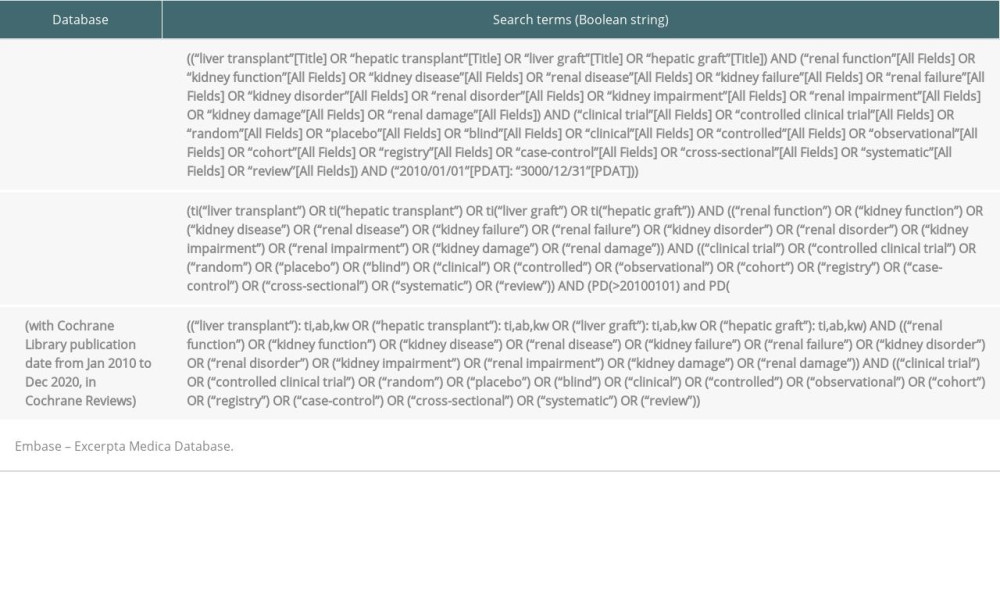 Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review.
Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review.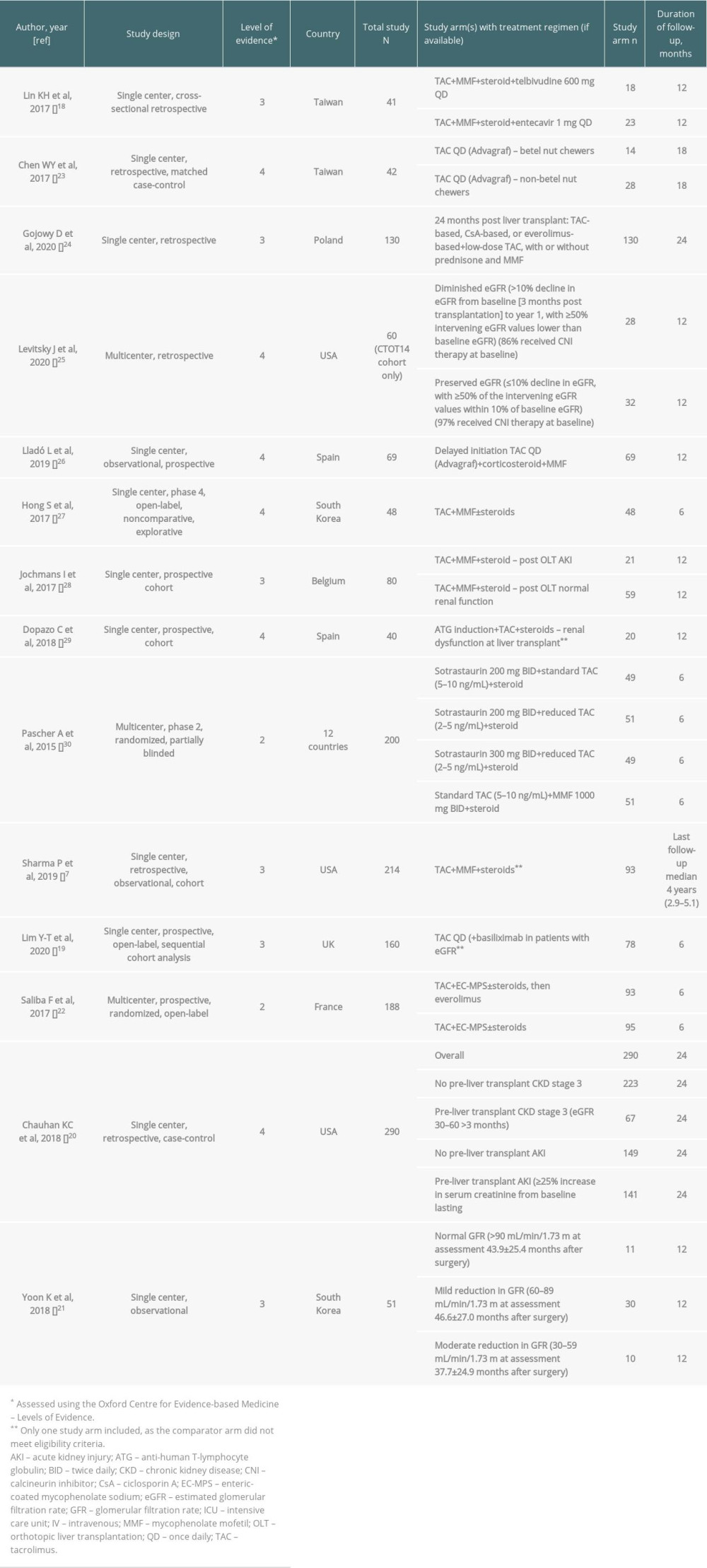 Table 4. Patient demographics and baseline characteristics for the included studies.
Table 4. Patient demographics and baseline characteristics for the included studies.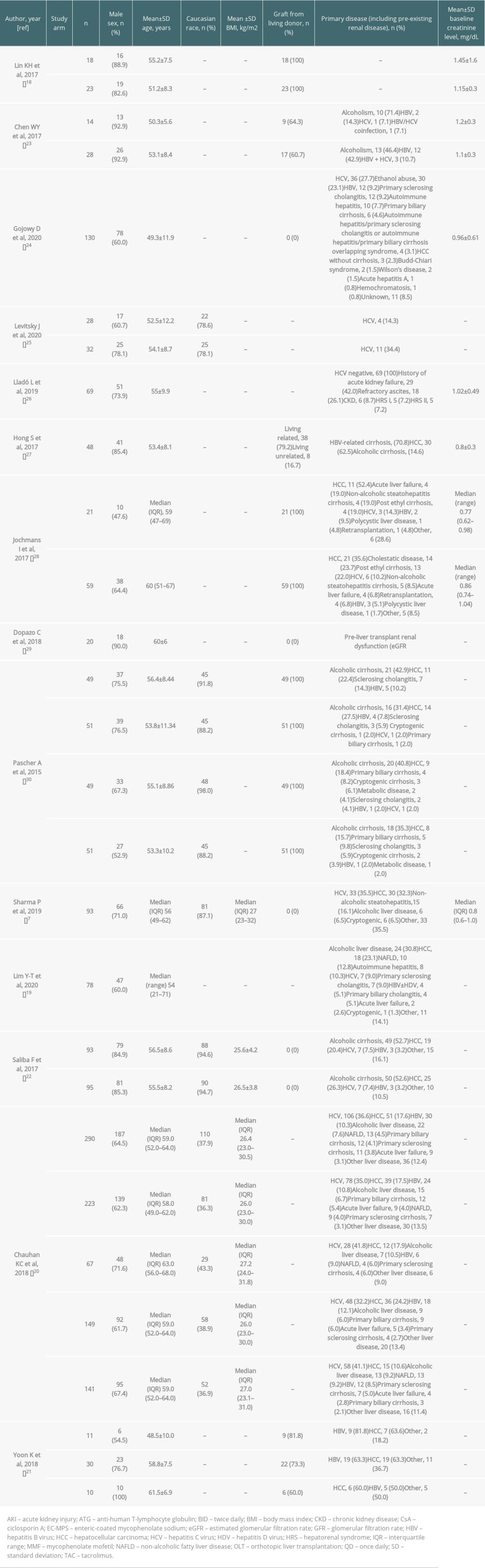 Table 5. GFR/eGFR and change from baseline for the included studies.
Table 5. GFR/eGFR and change from baseline for the included studies.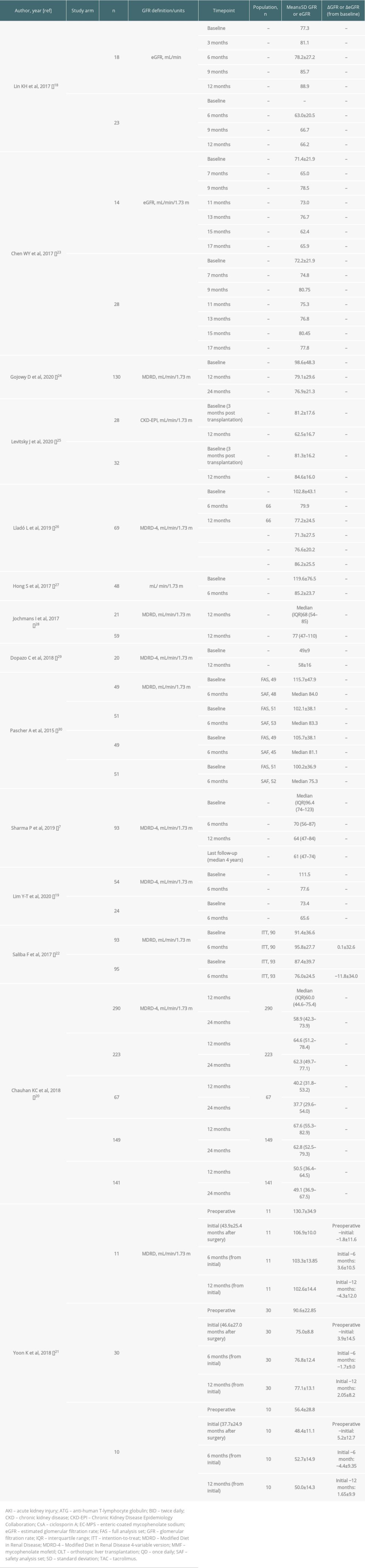 Table 6. CKD stage for the included studies.
Table 6. CKD stage for the included studies.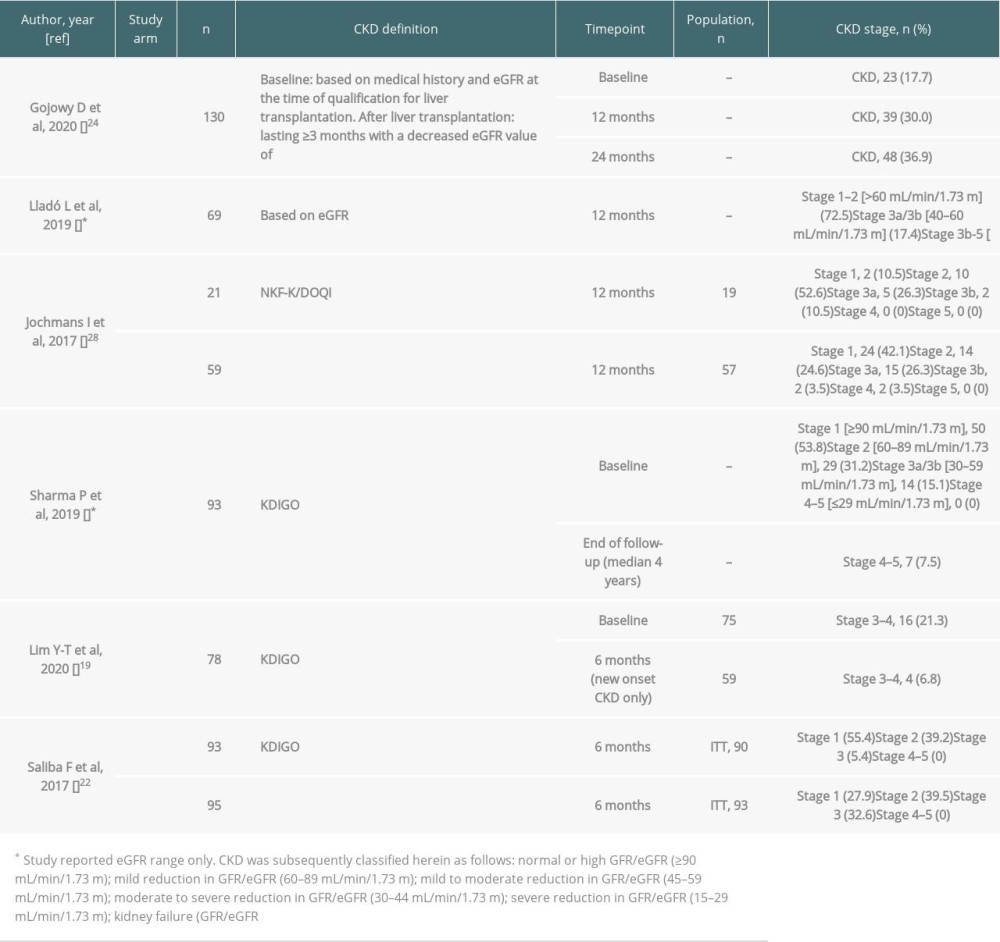 Table 7. Patient deaths for the included studies.
Table 7. Patient deaths for the included studies.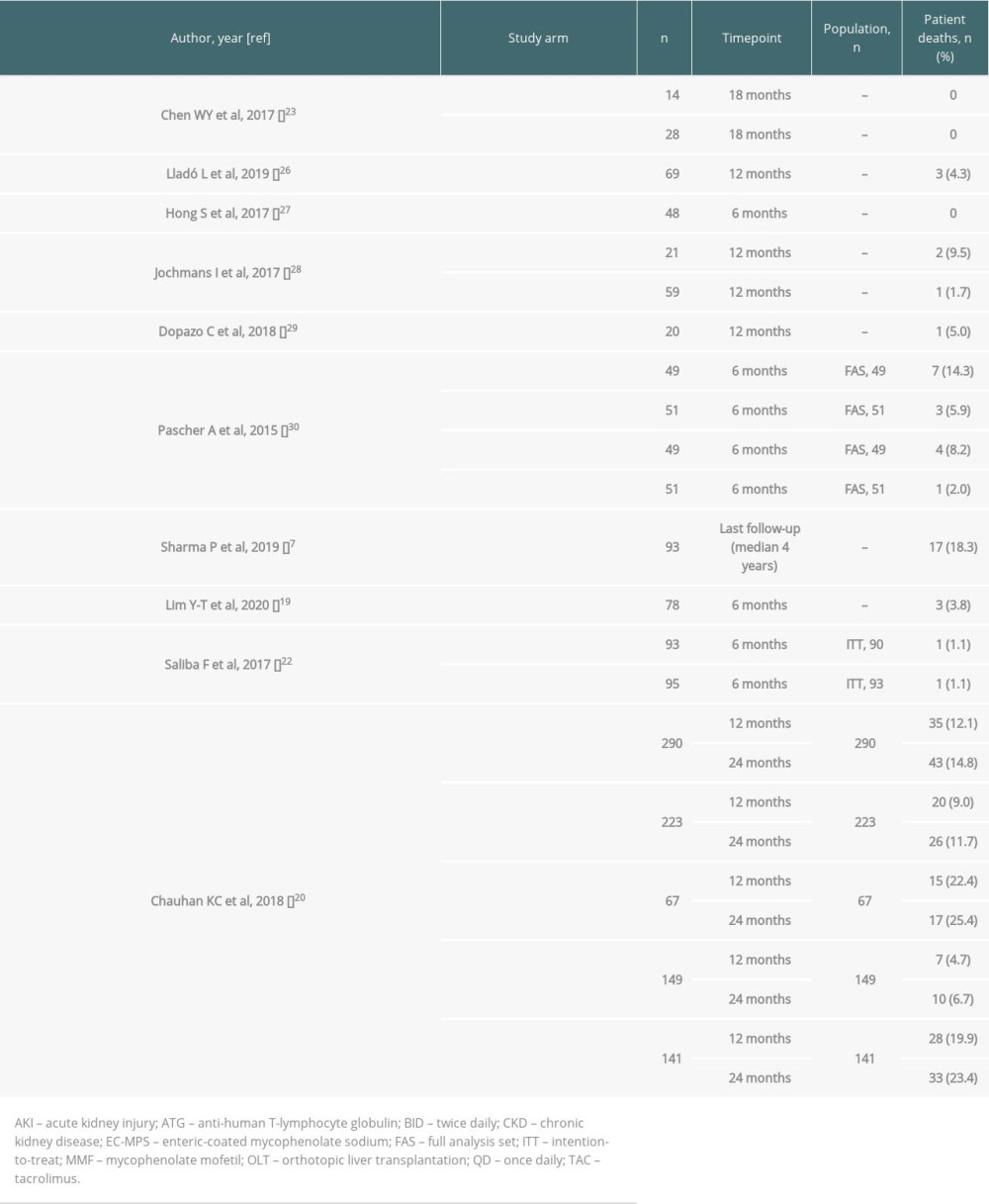 Table 8. Graft survival for the included studies.
Table 8. Graft survival for the included studies.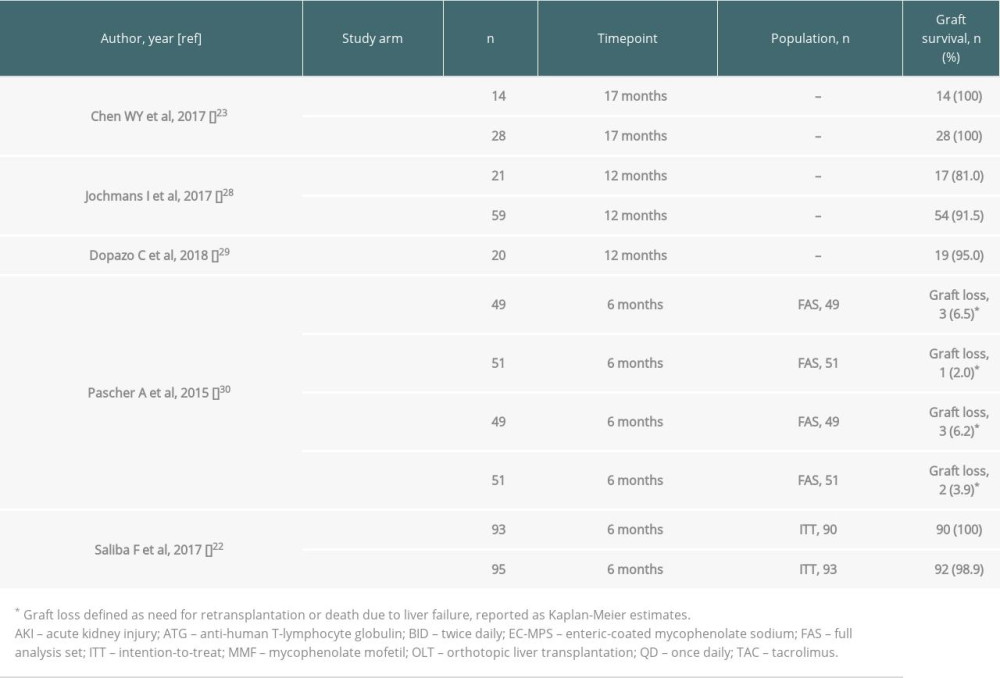 Table 9. Graft rejection for the included studies.
Table 9. Graft rejection for the included studies.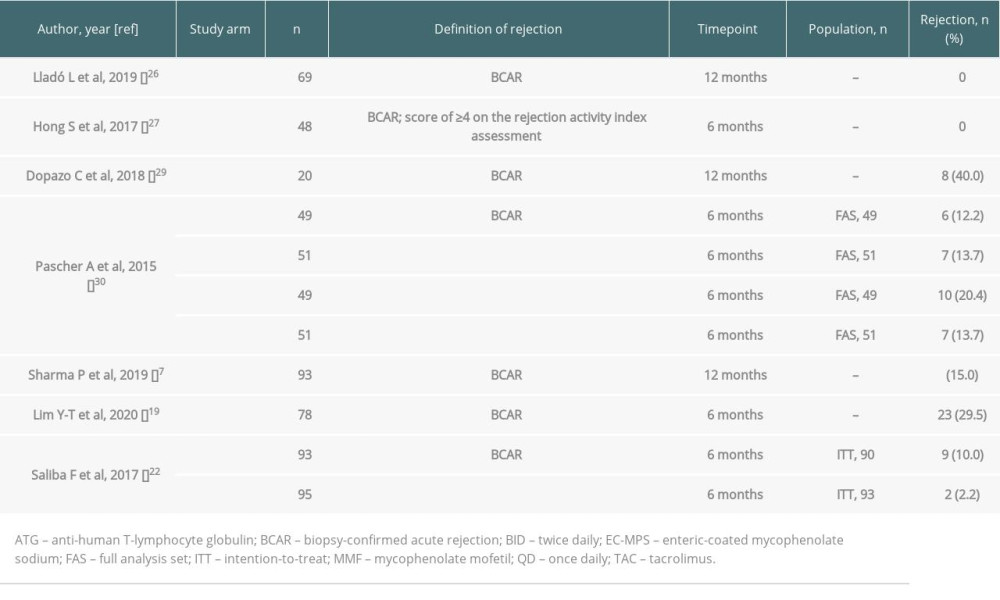 Supplementary Table 1. Immunosuppressive treatment dose and trough levels during follow-up for the included studies.
Supplementary Table 1. Immunosuppressive treatment dose and trough levels during follow-up for the included studies. Supplementary Table 2. Incidence or prevalence of diabetes for the included studies.
Supplementary Table 2. Incidence or prevalence of diabetes for the included studies.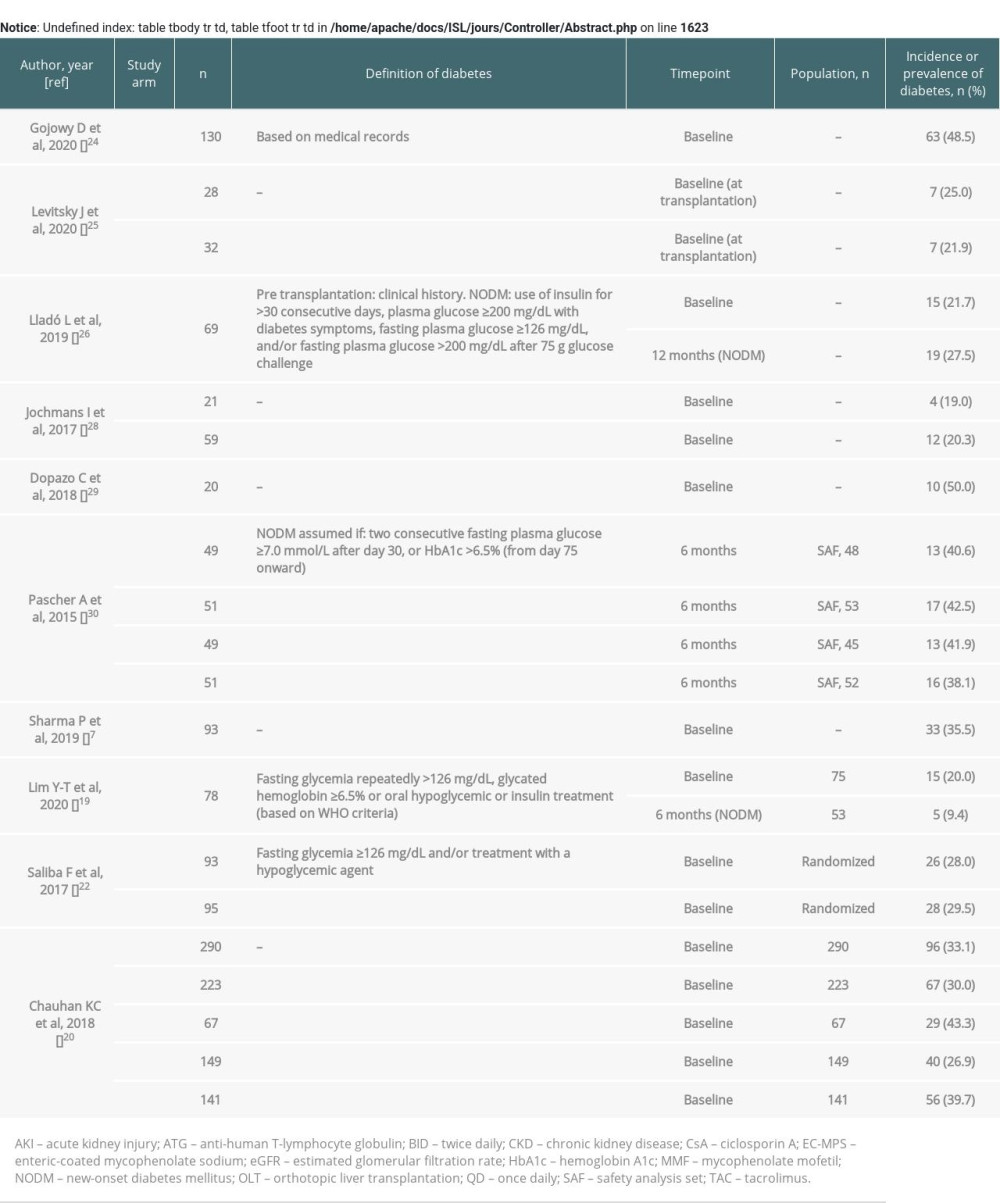
References
1. Gonwa TA, Wadei HM, Kidney disease in the setting of liver failure: Core curriculum 2013: Am J Kidney Dis, 2013; 62; 1198-212
2. Sharma P, Schaubel DE, Guidinger MK, Impact of MELD-based allocation on end-stage renal disease after liver transplantation: Am J Transplant, 2011; 11; 2372-78
3. Allen AM, Kim WR, Therneau TM, Chronic kidney disease and associated mortality after liver transplantation – a time-dependent analysis using measured glomerular filtration rate: J Hepatol, 2014; 61; 286-92
4. Ojo AO, Held PJ, Port FK, Chronic renal failure after transplantation of a nonrenal organ: N Engl J Med, 2003; 349; 931-40
5. Eriksson JK, Neovius M, Jacobson SH, Healthcare costs in chronic kidney disease and renal replacement therapy: A population-based cohort study in Sweden: BMJ Open, 2016; 6; e012062
6. Gonwa TA, McBride MA, Mai ML, Wadei HM, Kidney transplantation after previous liver transplantation: Analysis of the Organ Procurement Transplant Network database: Transplantation, 2011; 92; 31-35
7. Sharma P, Sun Y, Neal J, Renal outcomes of liver transplantation recipients receiving standard immunosuppression and early renal sparing immunosuppression: A retrospective single center study: Transplant Direct, 2019; 5; e480
8. Gonwa TA, Mai ML, Melton LB, End-stage renal disease (ESRD) after orthotopic liver transplantation (OLTX) using calcineurin-based immunotherapy: Risk of development and treatment: Transplantation, 2001; 72; 1934-39
9. Neuberger JM, Mamelok RD, Neuhaus P, Delayed introduction of reduced-dose tacrolimus, and renal function in liver transplantation: The “ReSpECT” study: Am J Transplant, 2009; 9; 327-36
10. Trunečka P, Klempnauer J, Bechstein WO, Renal function in de novo liver transplant recipients receiving different prolonged-release tacrolimus regimens: The DIAMOND study: Am J Transplant, 2015; 15; 1843-54
11. Yee M-L, Tan H-H, Use of everolimus in liver transplantation: World J Hepatol, 2017; 9; 990-1000
12. Page MJ, McKenzie JE, Bossuyt PM, The PRISMA 2020 statement: An updated guideline for reporting systematic reviews: BMJ, 2021; 372; n71
13. PRISMA: PRISMA 2020 checklist, 2020 http://www.prisma-statement.org/PRISMAStatement/Checklist
14. Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group, KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease: Kidney Int Suppl, 2013; 3; 1-150
15. Inker LA, Astor BC, Fox CH, KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD: Am J Kidney Dis, 2014; 63; 713-35
16. Higgins JPT, Altman DG, Gøtzsche PC, The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials: BMJ, 2011; 343; d5928
17. OCEBM Levels of Evidence Working Group: The Oxford Levels of Evidence 2, Oxford Centre for Evidence-Based Medicine Published Online First 2011https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence
18. Lin K-H, Chen Y-L, Lin P-Y, A follow-up study on the renal protective efficacy of telbivudine for hepatitis B virus-infected Taiwanese patients after living donor liver transplant: Exp Clin Transplant, 2017; 15; 65-68
19. Lim TY, McPhail MJ, Shah A, Sequential cohort analysis after liver transplantation shows de novo extended release tacrolimus is safe, efficacious, and minimizes renal dysfunction: Transplant Direct, 2020; 6; e528
20. Chauhan K, Azzi Y, Faddoul G, Pre-liver transplant renal dysfunction and association with post-transplant end-stage renal disease: A single-center examination of updated UNOS recommendations: Clin Transplant, 2018; 32; e13428
21. Yoon KC, Lee K-W, Oh SC, Urinary neutrophil gelatinase-associated lipocalin as a biomarker for renal injury in liver transplant recipients using calcineurin inhibitors: Transplant Proc, 2018; 50; 3667-72
22. Saliba F, Duvoux C, Gugenheim J, Efficacy and safety of everolimus and mycophenolic acid with early tacrolimus withdrawal after liver transplantation: A multicenter randomized trial: Am J Transplant, 2017; 17; 1843-52
23. Chen W-Y, Lee C-Y, Lin P-Y, Betel nut chewing is associated with reduced tacrolimus concentration in Taiwanese liver transplant recipients: Transplant Proc, 2017; 49; 326-29
24. Gojowy D, Kubis P, Gorecka M, Chronic kidney disease in patients after liver transplantation: A long-term retrospective analysis from 1 transplantation center: Transplant Proc, 2020; 52; 2492-96
25. Levitsky J, Asrani SK, Klintmalm G, Discovery and validation of a biomarker model (PRESERVE) predictive of renal outcomes after liver transplantation: Hepatology, 2020; 71; 1775-86
26. Lladó L, González-Castillo A, Fabregat J, Efficacy and safety of delayed prolonged-release tacrolimus initiation in de novo hepatitis C virus-negative orthotopic liver transplant recipients: A single-center, single-arm, prospective study: Ann Transplant, 2019; 24; 36-44
27. Hong SK, Lee K-W, Yoon KC, Efficacy and safety of generic mycophenolate mofetil (My-rept) 500-milligram tablets in primary liver transplant recipients: Transplant Proc, 2017; 49; 1402-8
28. Jochmans I, Meurisse N, Neyrinck A, Hepatic ischemia/reperfusion injury associates with acute kidney injury in liver transplantation: prospective cohort study: Liver Transpl, 2017; 23; 634-44
29. Dopazo C, Charco R, Caralt M, Low total dose of anti-human T-lymphocyte globulin (ATG) guarantees a good glomerular filtration rate after liver transplant in recipients with pretransplant renal dysfunction: Can J Gastroenterol Hepatol, 2018; 2018; 1672621
30. Pascher A, De Simone P, Pratschke J, Protein kinase C inhibitor sotrastaurin in de novo liver transplant recipients: A randomized phase II trial: Am J Transplant, 2015; 15; 1283-92
31. Asrani SK, Wiesner RH, Trotter JF, De novo sirolimus and reduced-dose tacrolimus versus standard-dose tacrolimus after liver transplantation: The 2000–2003 phase II prospective randomized trial: Am J Transplant, 2014; 14; 356-66
32. Knechtle SJ, Adams AB, Belatacept: Is there BENEFIT for liver transplantation too?: Am J Transplant, 2014; 14; 1717-18
33. Massoud O, Wiesner RH, The use of sirolimus should be restricted in liver transplantation: J Hepatol, 2012; 56; 288-90
34. Levitsky J, O’Leary JG, Asrani S, Protecting the kidney in liver transplant recipients: Practice-based recommendations from the American Society of Transplantation Liver and Intestine Community of Practice: Am J Transplant, 2016; 16; 2532-44
35. Kwong A, Kim WR, Lake JR, OPTN/SRTR 2018 Annual Data Report: Liver: Am J Transplant, 2020; 20(Suppl 1); 193-299
36. Thongprayoon C, Kaewput W, Thamcharoen N, Incidence and impact of acute kidney injury after liver transplantation: A meta-analysis: J Clin Med, 2019; 8; 372
37. Lee K, Jeon J, Kim JM, Perioperative risk factors of progressive chronic kidney disease following liver transplantation: Analyses of a 10-year follow-up single-center cohort: Ann Surg Treat Res, 2020; 99; 52-62
38. Fabrizi F, Dixit V, Martin P, Messa P, Chronic kidney disease after liver transplantation: Recent evidence: Int J Artif Organs, 2010; 33; 803-11
39. Lim SY, Wang R, Tan DJH, A meta-analysis of the cumulative incidence, risk factors, and clinical outcomes associated with chronic kidney disease after liver transplantation: Transpl Int, 2021; 34; 2524-33
40. Sharma P, Welch K, Eikstadt R, Renal outcomes after liver transplantation in the model for end-stage liver disease era: Liver Transplant, 2009; 15; 1142-48
41. Cullaro G, Verna EC, Lee BP, Lai JC, Chronic kidney disease in liver transplant candidates: A rising burden impacting post-liver transplant outcomes: Liver Transpl, 2020; 26; 498-506
42. Haugen CE, Holscher CM, Luo X, Assessment of trends in transplantation of liver grafts from older donors and outcomes in recipients of liver grafts from older donors, 2003–2016: JAMA Surg, 2019; 154; 441-49
Figures
 Figure 1. PRISMA diagram. eGFR – estimated glomerular filtration rate; Embase – Excerpta Medica Database; GFR – glomerular filtration rate; PRISMA – Preferred Reporting Items for Systematic reviews and Meta-Analyses.
Figure 1. PRISMA diagram. eGFR – estimated glomerular filtration rate; Embase – Excerpta Medica Database; GFR – glomerular filtration rate; PRISMA – Preferred Reporting Items for Systematic reviews and Meta-Analyses. Figure 2. Risk of bias assessments for the included studies based on (A) author disclosures and industry funding and (B) using the Cochrane Collaboration bias tool for randomized controlled trials.
Figure 2. Risk of bias assessments for the included studies based on (A) author disclosures and industry funding and (B) using the Cochrane Collaboration bias tool for randomized controlled trials. Tables
 Table 1. PECOS framework.
Table 1. PECOS framework. Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews.
Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews. Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review.
Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review. Table 4. Patient demographics and baseline characteristics for the included studies.
Table 4. Patient demographics and baseline characteristics for the included studies. Table 5. GFR/eGFR and change from baseline for the included studies.
Table 5. GFR/eGFR and change from baseline for the included studies. Table 6. CKD stage for the included studies.
Table 6. CKD stage for the included studies. Table 7. Patient deaths for the included studies.
Table 7. Patient deaths for the included studies. Table 8. Graft survival for the included studies.
Table 8. Graft survival for the included studies. Table 9. Graft rejection for the included studies.
Table 9. Graft rejection for the included studies. Table 1. PECOS framework.
Table 1. PECOS framework. Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews.
Table 2. Search terms for PubMed, Embase, and the Cochrane Database of Systematic Reviews. Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review.
Table 3. Study characteristics and treatment regimens for the 14 publications included in the systematic literature review. Table 4. Patient demographics and baseline characteristics for the included studies.
Table 4. Patient demographics and baseline characteristics for the included studies. Table 5. GFR/eGFR and change from baseline for the included studies.
Table 5. GFR/eGFR and change from baseline for the included studies. Table 6. CKD stage for the included studies.
Table 6. CKD stage for the included studies. Table 7. Patient deaths for the included studies.
Table 7. Patient deaths for the included studies. Table 8. Graft survival for the included studies.
Table 8. Graft survival for the included studies. Table 9. Graft rejection for the included studies.
Table 9. Graft rejection for the included studies. Supplementary Table 1. Immunosuppressive treatment dose and trough levels during follow-up for the included studies.
Supplementary Table 1. Immunosuppressive treatment dose and trough levels during follow-up for the included studies. Supplementary Table 2. Incidence or prevalence of diabetes for the included studies.
Supplementary Table 2. Incidence or prevalence of diabetes for the included studies. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








