11 October 2022: Original Paper
Reduced Risk of Chronic Graft-Versus-Host Disease (cGVHD) by Rabbit Anti-Thymocyte Globulin (ATG) in Patients Undergoing Matched Sibling Donor Transplantation in Hematological Malignancies
Shu Fang12ABCDEF, Nan Wang23ABCF, Lili Wang2ABCD, Jishan Du23BCF, Jingjing Yang23BCF, Yanan Wen23BCF, Yan Wei23BCF, Kun Qian12BCD, Hao Wang23ABF, Yifan Jiao23BCF, Chunji Gao12ACEG*, Liping Dou2ABDEGDOI: 10.12659/AOT.937356
Ann Transplant 2022; 27:e937356
Abstract
BACKGROUND: With the addition of anti-thymocyte globulin (ATG) to GVHD prophylaxis in patients undergoing transplantation of peripheral blood stem cells (PBSCT), the incidence of cGVHD decreases. However, the optimal dose and timing of ATG remain undetermined.
MATERIAL AND METHODS: In this historical controlled trial, data from 85 patients who had hematological malignancies and underwent matched sibling donor (MSD)-PBSCT were used to analyze the effectiveness of rabbit ATG (rATG) for prophylaxis of GVHD. Forty patients received 5 mg/kg rATG used for days -5 to -2, and 45 patients did not receive ATG.
RESULTS: All patients had successful engraftment except for 2 in the non-ATG group, who had platelet engraftment failure. The 2-year cumulative incidence of chronic GVHD (cGVHD) in the ATG group versus non-ATG group was 19.3% (95% CI, 8.4-33.6%) versus 61.4% (95% CI, 45.4-73.9%) (P<0.001), and in those with moderate to severe cGVHD it was 11.0% (95% CI, 3.4-23.6%) versus 31.8% (95% CI, 18.8-45.6%) (P=0.029), respectively. The 2-year cumulative incidence of non-relapse mortality and relapse (CIR) were 0% versus 15.5% (95% CI, 6.8-27.5%) (P=0.018), and 53.3% (95% CI, 35.6-68.1%) versus 26.7% (95% CI, 14.9-40.0%) (P=0.019), respectively. No differences were found in other survival outcomes. In the multivariate analysis, ATG was an independent protective factor for moderate to severe cGVHD (HR=0.314, 95% CI, 0.103-0.958, P=0.042), and was an independent poor risk factor for CIR (HR=2.337, 95% CI, 1.133-4.822, P=0.022).
CONCLUSIONS: ATG in our strategy was effective for prophylaxis of cGVHD, whereas the relapse rate was increased in patients with rATG.
Keywords: Graft vs Host Disease, Peripheral Blood Stem Cell Transplantation, Recurrence, Thymoglobulin, Antilymphocyte Serum, Hematologic Neoplasms, Hematopoietic Stem Cell Transplantation, Humans, Neoplasm Recurrence, Local, Siblings, Transplantation Conditioning
Background
Hematopoietic stem cell transplantation (HSCT) is the only way to cure patients who have malignant hematological diseases. However, graft-versus-host disease (GVHD), as the main complication after HSCT, impairs life quality and longevity [1–3]. Despite the use of calcineurin inhibitor combined with methotrexate (MTX) as GVHD prophylaxis, 30–50% of patients develop acute GVHD (aGVHD) [4], and 30–70% still have chronic GVHD (cGVHD) [1]. Another effective way to reduce GVHD is the depletion of T cells. Studies have shown that in vivo removal of T cells by ATG can reduce GVHD and prolong the survival of patients receiving HSCT from unrelated donors or haploidentical donors (HID-HSCT) without increasing relapse rates [5,6]. With the addition of anti-thymocyte globulin (ATG) to GVHD prophylaxis, the incidence of cGVHD also decreased [7–9].
Infusion of donor graft derived from peripheral blood stem cells increases the incidence of cGVHD [10,11]. Adding ATG to GVHD prophylaxis decreases GVHD in PBSCT patients [12–14]. A similar result was found in a study at our center [15]. Patients undergoing HID-transplantation from peripheral blood stem cells (PBSCT) were given 10 mg/kg rabbit ATG (rATG), divided across 4 days (from −5 to −2). We found a decreased incidence of severe cGVHD in these patients compared with patients undergoing PBSCT from matched sibling donor (MSD) (5.8% vs 21.2%,
Material and Methods
PATIENTS:
Patients with myelodysplastic syndrome (MDS)/acute myeloid leukemia (AML)/acute lymphocytic leukemia (ALL) who received MSD-PBSCT in the Hematology Department of the General Hospital of the Chinese people’s Liberation Army (PLA) from November 20, 2014, to December 31, 2020, met the conditions of this study. Patients’ ages were 12 to 65 years. The exclusion criteria were: (1) uncontrolled infections; (2) severe pulmonary, cardiac, hepatic or renal diseases; and (3) AML patients with t (15;17). Patients receiving a total of 5 mg/kg ATG on days −5 to −2 for GVHD prophylaxis were enrolled between January 2019 and December 2020. This trial was registered at
All patients received cyclosporine, mycophenolate, and short-term methotrexate for GVHD prophylaxis. Forty patients were treated with the addition of rATG and were assigned to the ATG group. Forty-five patients without rATG were assigned to the non-ATG group. About 92.5% (37/40) of patients in the ATG group and 93.3% (42/45) of patients in the non-ATG group received a busulfan (Bu)/cyclophosphamide (Cy) conditioning regimen. Other patients received total body irradiation (TBI)/Cy, fludarabine (Flu) combined with cytarabine, granulocyte colony-stimulating factor (G-CSF), and Bu (FLAG/Bu), or Bu/Flu conditioning regimens (Table 1).
HLA MATCHING:
HLA loci, including HLA-A, HLA-B, HLA-C, HLA-DRB1, and HLA-DQB1, were detected in all patients. All patients and donors matched at these loci.
GVHD PROPHYLAXIS AND THERAPY:
Forty patients received 1 mg/kg rATG on days −5 to −3, and 2 mg/kg on day −2 prior to transplantation. All patients received cyclosporine A (CsA), methotrexate (MTX), and mycophenolate mofetil (MMF) as GVHD prophylaxis. CsA was injected intravenously from day −10 until oral refeeding at a dose of 3 mg/kg, and the target concentration was 150 to 200 ng/mL. After 6 months, it was tapered by 25% every 2 weeks except for patients with relapse. In patients who relapsed within 60 days, CsA was rapidly tapered within 2 weeks. In those who relapsed after 60 days, CsA was immediately discontinued. If patients were intolerant to CsA, they used tacrolimus instead. The usage of MTX was 15 mg/m2 on day +1 and 10 mg/m2 on days +3, +6, and +11. MMF was administered orally from day −10 to engraftment, at a dose of 250 mg, twice daily [16]. Patients with aGVHD received methylprednisolone (MP). The MP was used at a 2 mg/kg/day dose divided into 2 doses for 7 consecutive days. Then, it was tapered over 8 weeks.
ENDPOINTS AND DEFINITIONS:
The primary endpoint was the incidence of cGVHD. Secondary endpoints were cumulative incidence of aGVHD, NRM, relapse (CIR), GVHD-free and relapse-free survival (GRFS), OS, and disease-free survival (DFS). In this study, neutrophils were successfully engrafted in all patients and they were eligible for analysis of aGVHD. Data from patients who were still alive at 100 days were eligible for analysis of cGVHD. In this study, all patients, except 1 who died within 100 days in the non-ATG group, were eligible for analysis of cGVHD.
The definition of neutrophil and platelet engraftment after transplantation was as described in the previous study [16]. Disease risk index was assessed as previously described [17]. Acute GVHD and cGVHD were diagnosed and graded based on the established criteria [18–21]. OS was defined as the time from transplantation to death or last follow-up. DFS was defined as the time from transplantation to relapse, death, or last follow-up. Relapse was defined as reappearance of peripheral blood blasts or >5% blasts detected in bone marrow or extramedullary infiltration. Non-relapse mortality (NRM) was defined as death without disease relapse or progression. GRFS was defined as the absence of grade 3 to 4 aGVHD, moderate to severe cGVHD, relapse, or death.
STATISTICAL ANALYSIS:
SPSS 24.0 and R 4.0.3 software was used to do statistical analysis. Continuous variables were exhibited as median (range). The Mann-Whitney U test was used to compare these variables. The chi-square test was used to compare categorical variables. For the expected count of an event <5 or a total number of patients <40, Fisher’s exact test was used. The competing risk model was used to calculate the cumulative incidence of GVHD, relapse, and NRM between 2 groups. Gray’s test was used to compare the
Results
CLINICAL CHARACTERISTICS OF PATIENTS:
We included 85 patients in this study. Forty patients receiving ATG treatment were assigned to the ATG group, and 45 were in the non-ATG group. The proportion of patients with ALL in ATG group was higher than that in non-ATG group (42.5% vs 17.8%, P=0.013, Table 1). Salvage HSCT was performed in 2 patients in the ATG group and 4 patients in the non-ATG group who were refractory or relapsed (Table 1). The counts of mononuclear cells (MNCs) and CD34-positive cells in the ATG group were higher than those in the non-ATG group (MNCs, P<0.001, CD34+ cells, P=0.001). The distribution of other baseline characteristics was similar in both groups (Table 1).
ENGRAFTMENT:
All patients had successful neutrophil and platelet engraftment, except for 2 in the non-ATG group who had platelet engraftment failure. One of these 2 patients died at day +96 because of aGVHD, and the other failed to have platelet engraftment and died of relapse at day +125. The median time of neutrophil engraftment in the ATG group versus non-ATG group was 12 (range, 8–18) days versus 10 (range, 9–32) days (P=0.060, Table 2). The median time of platelet engraftment in the ATG and non-ATG groups were 12 (range, 7–29) days and 13 (range, 9–75) days, respectively (P=0.084, Table 2).
MAINTENANCE THERAPY AFTER TRANSPLANTATION:
Four patients in the ATG group and 8 in the non-ATG group received prophylactic donor lymphocyte infusion (DLI). None of them developed cGVHD in the ATG group, while 3 out of 4 patients had a relapse. In the non-ATG group, 4 out of 8 patients developed cGVHD, 1 of whom died due to a relapse. The other 4 patients without cGVHD died due to relapse. Three FLT3-ITD+ patients in the ATG group and 5 in the non-ATG group received sorafenib after transplantation. None of them developed cGVHD in the ATG group, but 1 of them eventually relapsed. In the non-ATG group, 3 of them developed cGVHD and 1 of them died of relapse. The remaining 2 patients died of relapse. For Ph+ ALL, 6 patients in the ATG group and 3 in the non-ATG group received tyrosine kinase inhibitor (TKI) after transplantation. None of them developed cGVHD in the ATG group, but 3 of them had a relapse. One out of 3 patients in the non-ATG group developed cGVHD, and none of them relapsed.
ACUTE AND CHRONIC GVHD:
There were no significant differences observed in the cumulative incidence of grades 2–4 aGVHD (P=0.231, Figure 1A) and grades 3–4 aGVHD (P=0.885, Figure 1B) between the ATG group and non-ATG group. The median time to onset was 26.5 days (range, 13.0–78.0 days) in the ATG group and 47 days (range, 16–98 days) in the non-ATG group. The 100-day cumulative incidence of grades 2–4 aGVHD in the ATG group versus non-ATG group was 37.5% (95% CI, 22.9–52.1%) and 28.9% (95% CI, 16.6–42.4%), respectively. The 100-day cumulative incidence of grades 3–4 aGVHD and grades 3–4 aGVHD was 5% (95% CI, 0.9%–14.8%) and 4% (95% CI, 0.8–13.3%), respectively. One patient died of severe aGVHD on day +96 in the non-ATG group. In the univariate and multivariate analysis, there were no risk factors associated with grades 2–4 aGVHD (Tables 3, 4).
In the ATG group, 17.5% (7/40) of patients had cGVHD, 4 of whom had preceding aGVHD. The median time to develop cGVHD was 297 days (range, 162–559 days). One patient with cGVHD died of relapse. In the non-ATG group, 60% (27/45) of patients had cGVHD. Among them, 11 patients had preceding aGVHD. The median time to develop cGVHD was 316 days (range, 116–900 days). Six out of 27 patients with cGVHD died because of pneumonia, and another 4 died of relapse. The two-year cumulative incidence of cGVHD in the ATG group was lower than that in the non-ATG group (19.3% [95% CI, 8.4–33.6%] versus 61.4% [95% CI, 45.4–73.9%], P<0.001, Figure 1C). The two-year cumulative incidence of moderate to severe cGVHD was also lower in the ATG group than that in the non-ATG group (11.0% [95% CI, 3.4–23.6%] versus 31.8% [95% CI, 18.8–45.6%], P=0.029, Figure 1D). In the univariate analysis, absence of ATG was a poor risk factor for overall cGVHD and moderate to severe cGVHD (Tables 3, 5, Figure 1C, 1D). In the multivariate analysis, ATG was an independent protective factor for occurrence of overall cGVHD and moderate to severe cGVHD (ATG vs non-ATG, overall cGVHD, HR=0.251, 95% CI, 0.108–0.582, P=0.001; moderate to severe cGVHD, HR=0.314, 95% CI, 0.103–0.958, P=0.042. Tables 3–5).
VIRUS INFECTION, RELAPSE AND NRM:
In the ATG group, 8 patients experienced cytomegalovirus (CMV) reactivation. Fifteen patients experienced Epstein-Barr virus (EBV) reactivation. In the non-ATG group, 8 and 2 patients experienced CMV and EBV reactivation, respectively (Table 2). The percentage of patients with EBV reactivation was 37.5% in the ATG group and 4.4% in the non-ATG group (P<0.001, Table 2). No patients had post-transplantation lymphoproliferative disorder (PTLD) in either group.
Twenty patients relapsed after transplantation in the ATG group. By the end of the follow-up, 11 patients had died of relapse. The two-year CIR was 53.3% (95% CI, 35.6–68.1%). In the non-ATG group, 12 patients relapsed and died. The two-year CIR was 26.7% (95% CI, 14.9–40.0%). In the univariate analysis, use of ATG was an inferior factor associated with CIR (Table 5, Figure 2A). In the multivariate analysis, ATG treatment was an independent poor risk factor for CIR (HR=2.337, 95% CI, 1.133–4.822, P=0.022, Table 4).
In the ATG group, no patients died because of NRM. In the non-ATG group, 7 patients died because of NRM: 1 of them died due to aGVHD and the other 6 died because of pneumonia and eventual respiratory failure. The two-year NRM was 15.5% (95% CI, 6.8%–27.5%). The rate of NRM was lower in the ATG group than that in the non-ATG group (P=0.018, Figure 2B).
SURVIVAL:
In the ATG group, 50% (20/40) of patients survived with CR status. About 22.5% (9/40) of patients relapsed and were still alive at the end of follow-up. The median follow-up was 811 days (range, 458–1137 days). In the non-ATG group, 57.8% (26/45) of patients survived with CR status. The median follow-up was 1906.5 days (range, 1290.0–2686.0 days). The two-year OS was 71.9% (95% CI, 54.9–83.4%) in the ATG group and 62.2% (95% CI, 46.5–74.6%) in the non-ATG group (P=0.315, Figure 2C). The two-year DFS was 46.7% (95% CI, 29.7–62.1%) versus 62.2% (95% CI, 46.5%–74.6%) in the ATG group versus non-ATG group (P=0.284, Figure 2D). The two-year GRFS was 76.5% (95% CI, 67.1–83.6%) versus 66.9% (95% CI, 55.7–76.0%) in the ATG group versus non-ATG group (P=0.306, Figure 2E, Supplementary Table 1). In the univariate and multivariate analysis, no risk factors were found to be associated with OS and DFS (Supplementary Table 2, Table 4). In the multivariate analysis, high count of CD34+ cells were related to GRFS (HR=1.785, 95% CI, 1.067–2.984, P=0.027, Table 4).
Discussion
GVHD is the leading cause affecting long-term survival after HSCT [22,23]. The current regimens for GVHD prophylaxis are CsA and MTX. However, the incidence of grades 2–4 aGVHD remains 19–40% in patients with MSD-HSCT, and that of cGVHD is 40–60% [16,24,25]. A mainstream approach to the removal of T cells in vivo and to reducing GVHD is post-transplant cyclophosphamide (PTCy) [26,27]. Another effective strategy for T cell depletion in vivo is ATG [28]. ATG effectively targets alloreactive T cells from the graft, leading to T cell depletion and decreased incidence of GVHD [29]. PTCy appears to be more effective in GVHD prophylaxis compared with ATG [26,27]. However, there is no difference in OS between the 2 regimens [26,27]. These may be due to the relatively higher incidences of graft failure and relapse [30,31]. Due to the absence of prospective randomized trials directly comparing the efficacy of these 2 approaches, it remains unclear which regimen is better. In our study, we used a strategy for GVHD prophylaxis with 5 mg/kg rATG divided over 4 days in 40 patients. We found that the 2-year cumulative incidence of cGVHD in ATG group was 23.2%, lower than that of the non-ATG group.
A previous study used 10 mg/kg ATG (ATG-Fresenius) as GVHD prophylaxis before HLA-identical sibling transplantation in acute leukemia patients [32]. The 2-year incidence of cGVHD was lower in the ATG group compared with non-ATG group. No differences were found in the rates of aGVHD, relapse, 2-year relapse-free survival, and OS between groups [32]. Another study revealed that 4.5 mg/kg ATG in MSD-HSCT patients could reduce the risk of overall cGVHD, extensive cGVHD, and grades 2–4 aGVHD but not increase relapse and not improve NRM, OS, or LFS [33]. Other studies investigated 5 mg/kg rATG divided over 3 days [25] or 2 days [16] for GVHD prophylaxis in patients receiving MSD-HSCT, and found a lower incidence of overall cGVHD and extensive cGVHD and improved OS in the ATG group. Our results showing the benefits of rATG in reducing the risk of cGVHD were similar to previous studies. Our study showed that rATG reduced the risk of total cGVHD and moderate to severe cGVHD, but not grades 2–4 aGVHD.
However, our study showed higher rates of relapse and EBV infection in the ATG group than those in the non-ATG group. The use of rATG before HSCT leads to depletion of T cells [28], immunosuppression, infection, and relapse [34]. Previous studies found that the relapse rate increased in patients with high-dose ATG [35] but not in patients receiving low-dose ATG [32,33]. Nevertheless, a recent prospective, single-center, randomized study showed that the relapse rate was increased in patients with a 2.5 mg/kg ATG to prevent GVHD [36], especially in cytogenetic high-risk patients. In our study, we found that the percentage of cytogenetic high-risk patients was higher in the ATG group than in the non-ATG group, which might lead to a higher relapse rate in the ATG group. Another reason might be the lower prophylactic DLI in the ATG group. Although we found that in the ATG group the risk of EBV infection was increased, no patients developed PTLD. These might be due to adequate and timely antiviral therapy.
In the ATG group, we also found a lower rate of NRM. These might be due to the lower incidence of cGVHD in the ATG group. We discovered that cGVHD occurred in 6 out of 7 patients who died of NRM in the non-ATG group. These patients eventually died of pneumonia infection and respiratory failure after long-term immunosuppressive regimen treatment. These might be the reason for the higher NRM in patients without ATG treatment.
This study also has some limitations. First, it was a singer-center study and patient selection may have been biased. Second, it was a non-randomized, non-concurrent control study, the distribution of baseline characteristics of patients between the 2 groups may have been uneven, and many confounding factors may have affected the results. In addition, the sample size is not very large, so the data acquired in our study may be insufficient to prove the reliability of the conclusions. Multicenter, large-sample, prospective, randomized, controlled studies are still needed to verify these results.
Conclusions
In summary, our study shows that 5 mg/kg rATG divided over 4 days can reduce the risk of cGVHD, whereas the relapse rate was higher in patients with rATG. The strategy of rATG in our study need to be cautiously used in clinical practice. More studies are needed to investigate the optimal dose and timing of rATG in transplant patients.
Figures
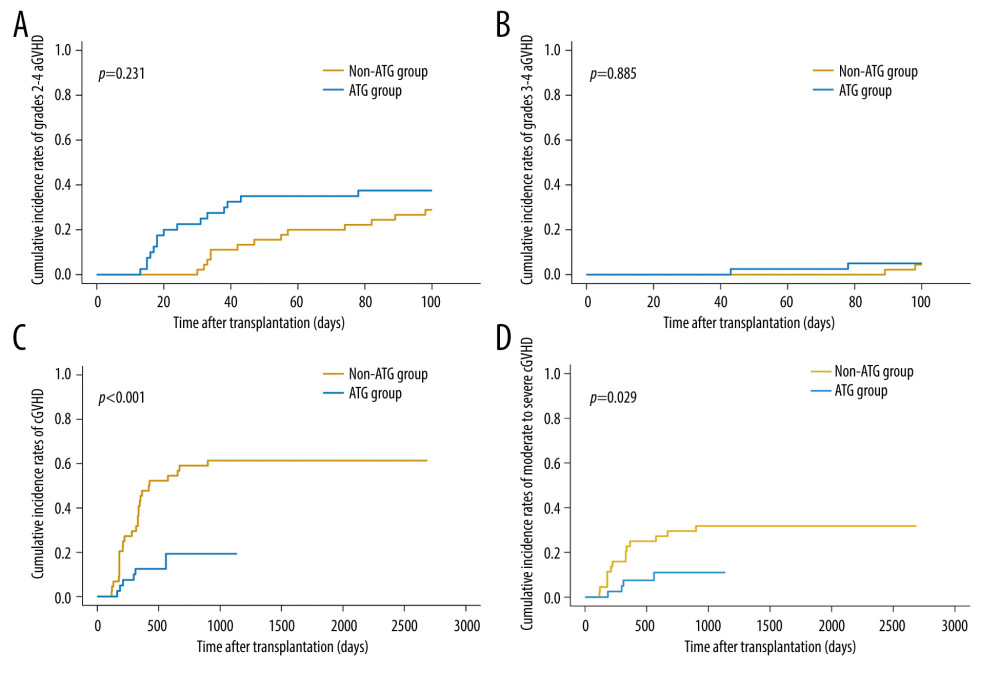 Figure 1. (A–D) Comparison of cumulative incidence of aGVHD and cGVHD between ATG group and non-ATG group (R version 4.0.3). aGVHD – acute graft-versus-host disease; cGVHD – chronic graft-versus-host disease.
Figure 1. (A–D) Comparison of cumulative incidence of aGVHD and cGVHD between ATG group and non-ATG group (R version 4.0.3). aGVHD – acute graft-versus-host disease; cGVHD – chronic graft-versus-host disease. 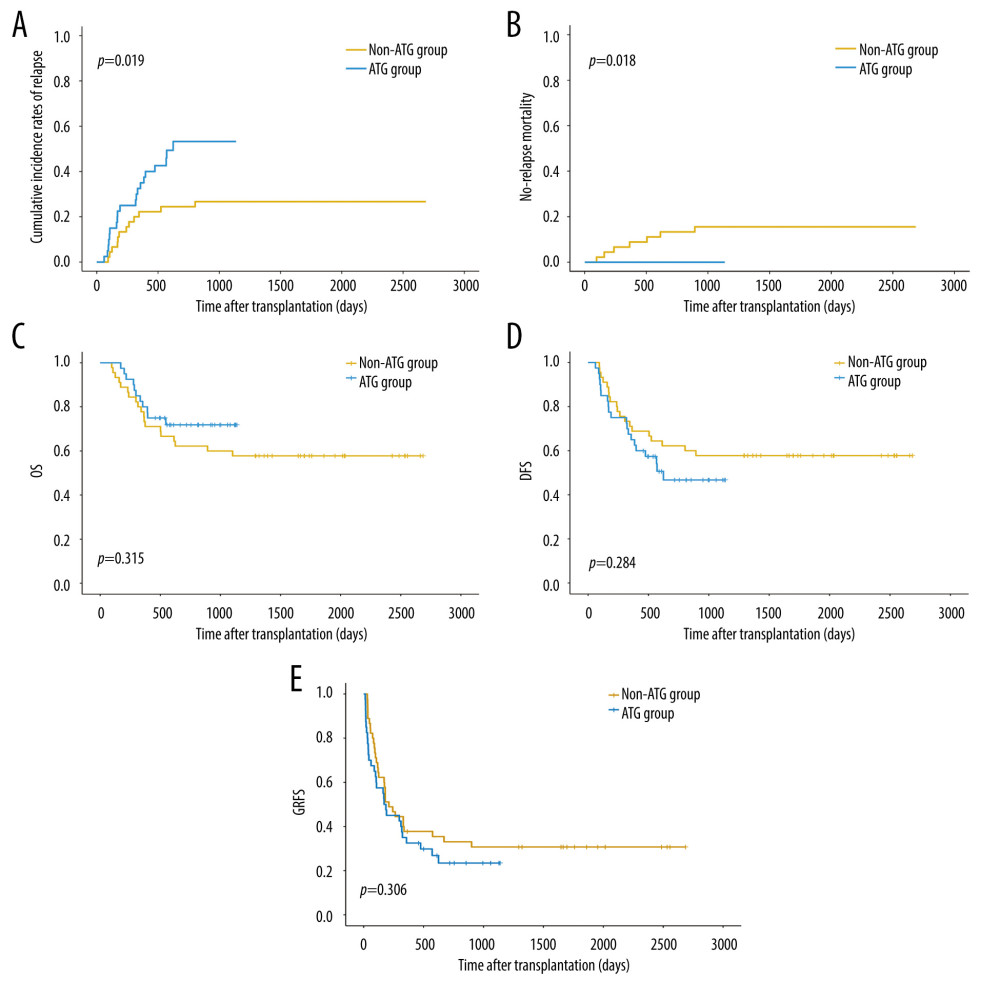 Figure 2. (A–E) Comparison of cumulative incidence of relapse, non-relapse mortality, OS, DFS, and GRFS (R version 4.0.3). OS – overall survival; DFS – disease-free survival; GRFS – GVHD-free relapse-free survival; GVHD – graft-versus-host disease.
Figure 2. (A–E) Comparison of cumulative incidence of relapse, non-relapse mortality, OS, DFS, and GRFS (R version 4.0.3). OS – overall survival; DFS – disease-free survival; GRFS – GVHD-free relapse-free survival; GVHD – graft-versus-host disease. Tables
Table 1. Clinical characteristics of 85 patients.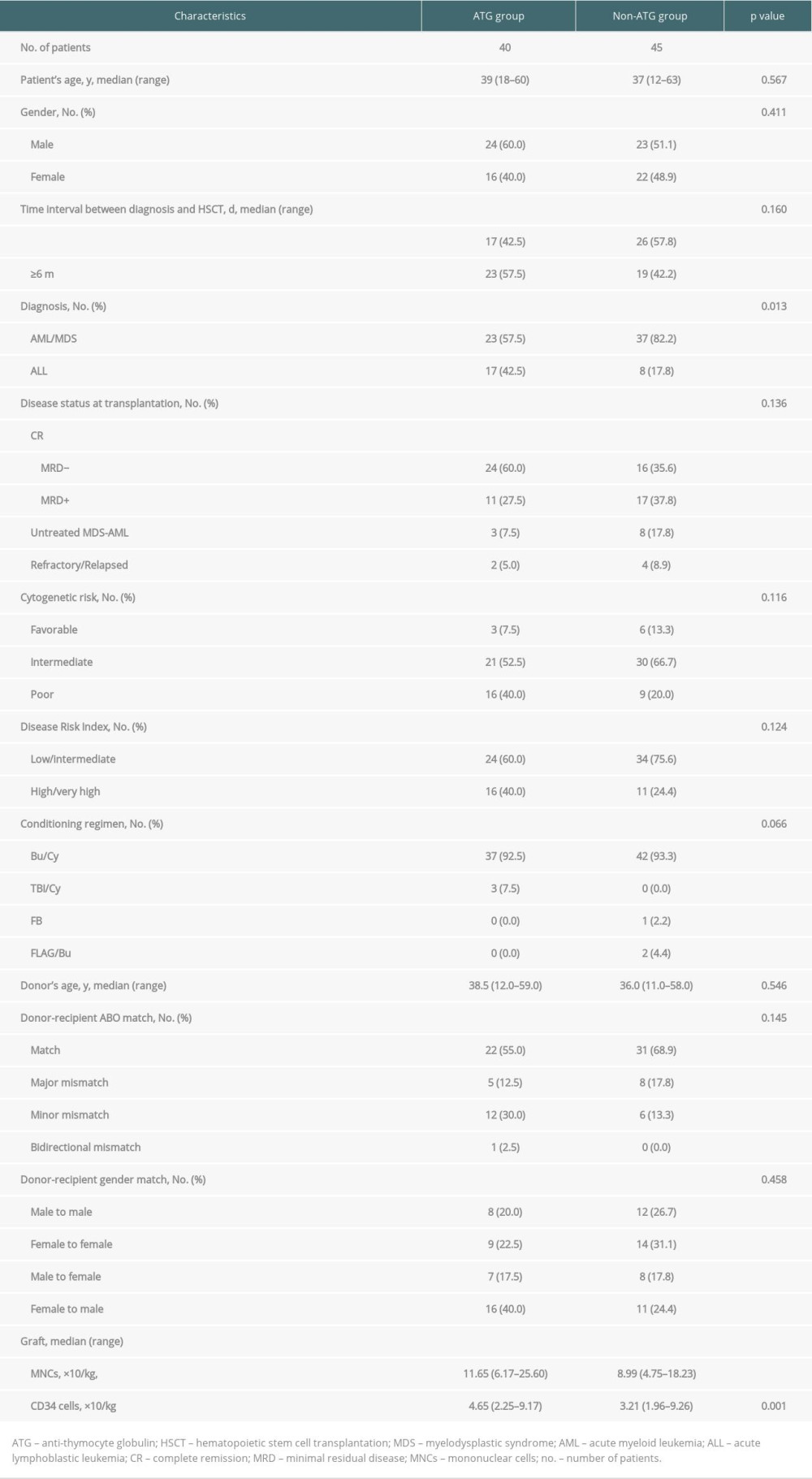 Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT.
Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT.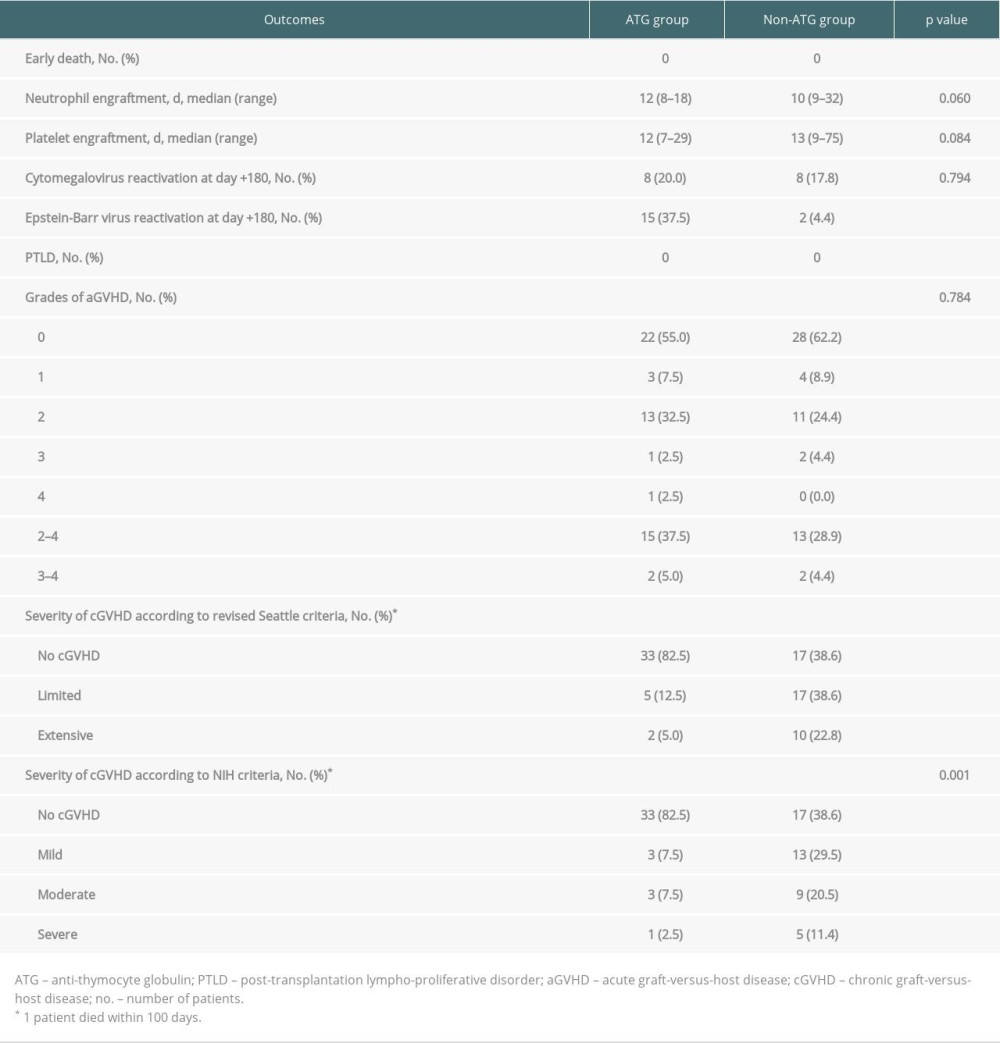 Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients.
Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients. Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients.
Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients.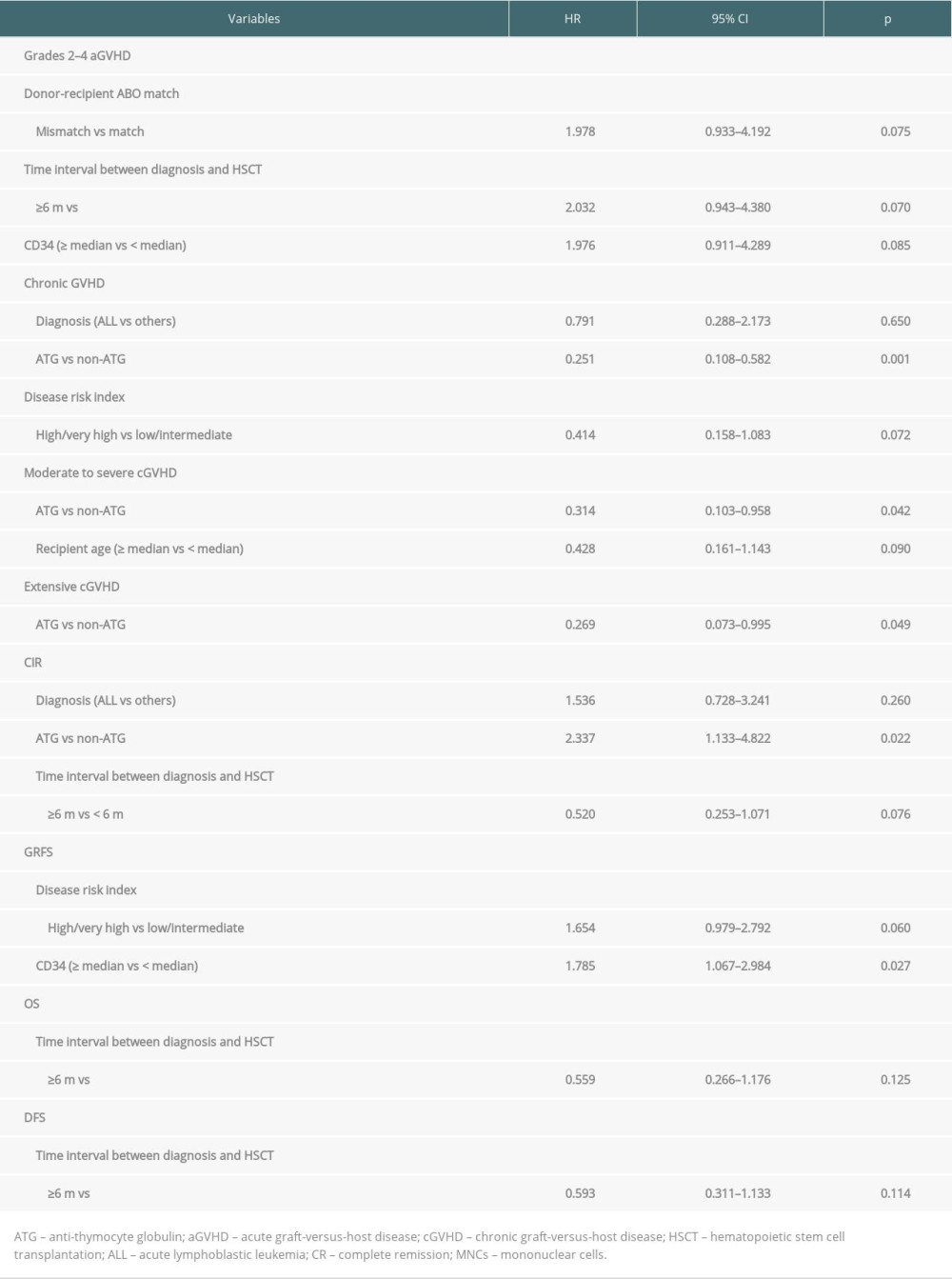 Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients.
Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients.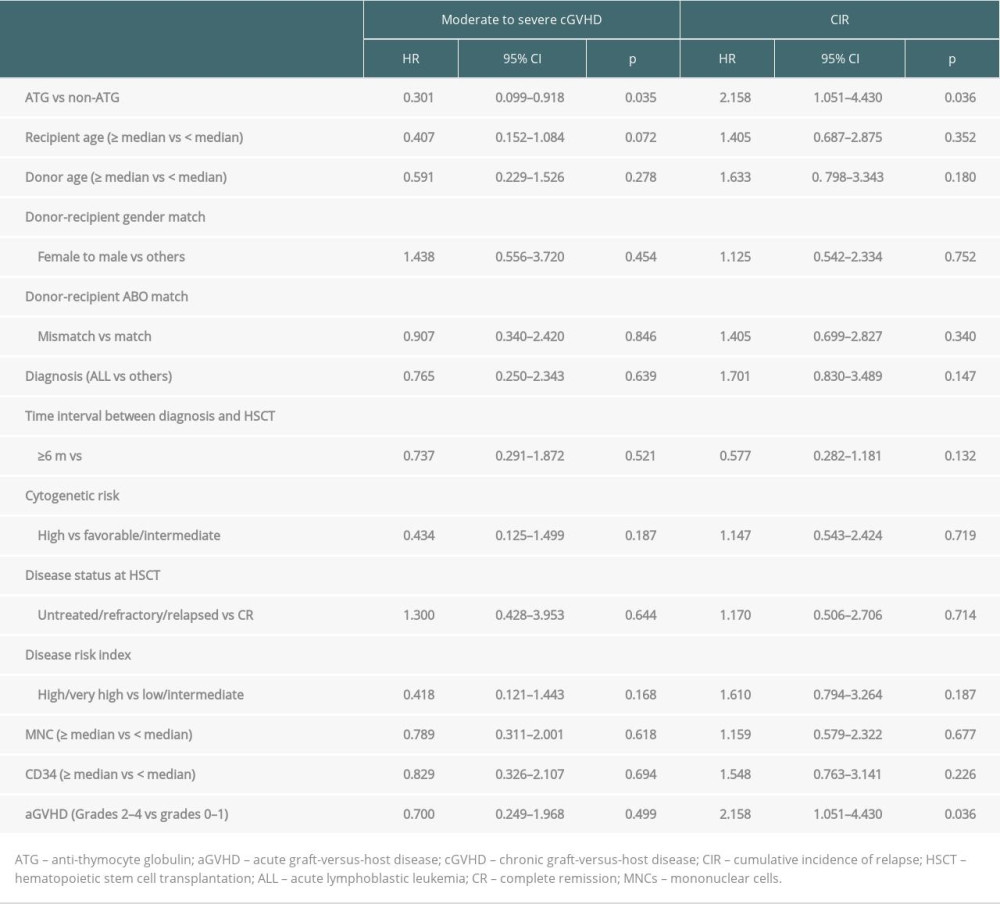 Supplementary Table 1. Univariate analysis of risk factors for GRFS in all patients.
Supplementary Table 1. Univariate analysis of risk factors for GRFS in all patients.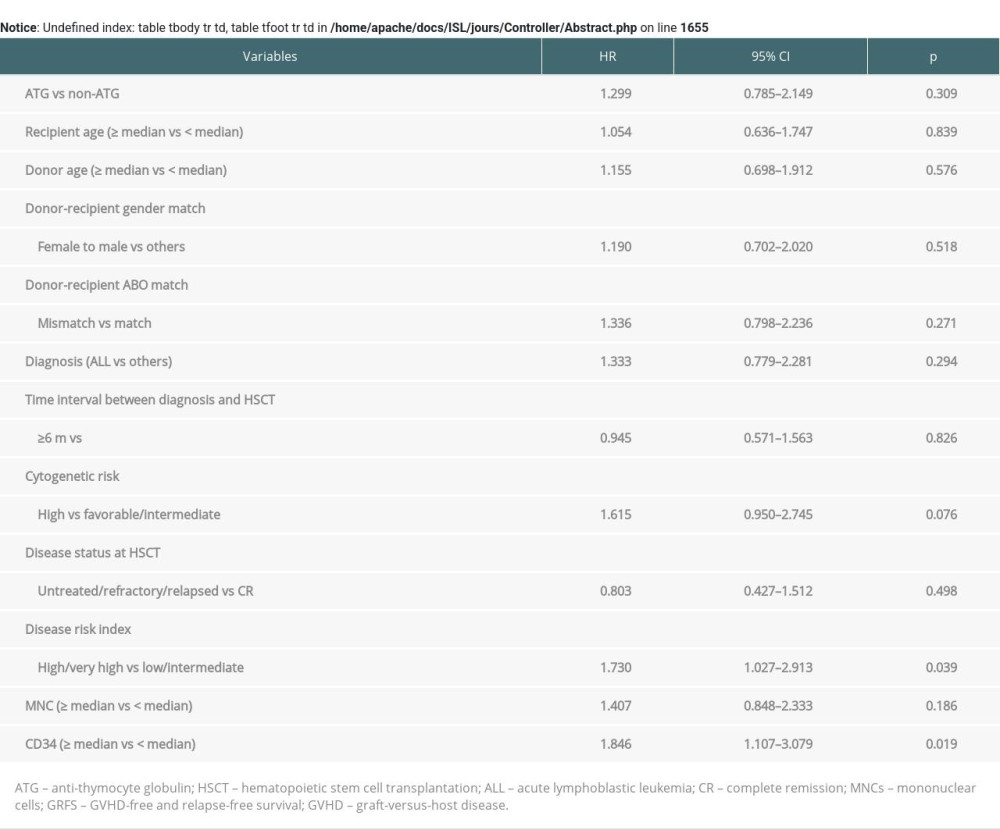 Supplementary Table 2. Univariate analysis of risk factors for OS and DFS in all patients.
Supplementary Table 2. Univariate analysis of risk factors for OS and DFS in all patients.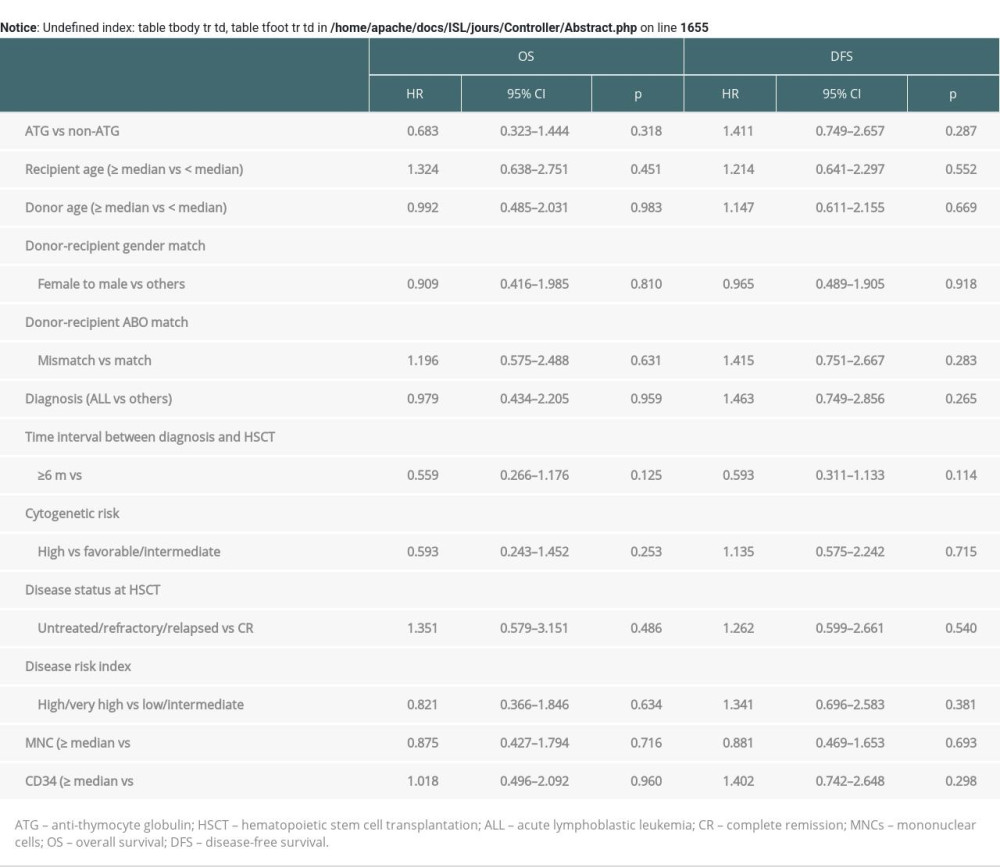
References
1. Zeiser R, Blazar BR, Pathophysiology of chronic graft-versus-host disease and therapeutic targets: N Engl J Med, 2017; 377(26); 2565-79
2. Wingard JR, Majhail NS, Brazauskas R, Long-term survival and late deaths after allogeneic hematopoietic cell transplantation: J cClin Oncol, 2011; 29(16); 2230-39
3. Martin PJ, Counts GW, Appelbaum FR, Life expectancy in patients surviving more than 5 years after hematopoietic cell transplantation: J Clin Oncol, 2010; 28(6); 1011-16
4. Zeiser R, Blazar BR, Acute graft-versus-host disease – biologic process, prevention, and therapy: N Engl J Med, 2017; 377(22); 2167-79
5. Walker I, Panzarella T, Couban S, Addition of anti-thymocyte globulin to standard graft-versus-host disease prophylaxis versus standard treatment alone in patients with haematological malignancies undergoing transplantation from unrelated donors: Final analysis of a randomised, open-label, multicentre, phase 3 trial: Lancet Haematol, 2020; 7(2); e100-e11
6. El-Cheikh J, Devillier R, Dulery R, Impact of adding antithymocyte globulin to posttransplantation cyclophosphamide in haploidentical stem-cell transplantation: Clin Lymphoma Myeloma Leuk, 2020; 20(9); 617-23
7. Martinez-Cibrian N, Zeiser R, Perez-Simon JA, Graft-versus-host disease prophylaxis: Pathophysiology-based review on current approaches and future directions: Blood Rev, 2021; 48; 100792
8. Bacigalupo A, Lamparelli T, Bruzzi P, Antithymocyte globulin for graft-versus-host disease prophylaxis in transplants from unrelated donors: 2 randomized studies from Gruppo Italiano Trapianti Midollo Osseo (GITMO): Blood, 2001; 98(10); 2942-47
9. Bacigalupo A, Lamparelli T, Barisione G, Thymoglobulin prevents chronic graft-versus-host disease, chronic lung dysfunction, and late transplant-related mortality: long-term follow-up of a randomized trial in patients undergoing unrelated donor transplantation: Biol Blood Marrow Transplant, 2006; 12(5); 560-65
10. Arai S, Arora M, Wang T, Increasing incidence of chronic graft-versus-host disease in allogeneic transplantation: A report from the Center for International Blood and Marrow Transplant Research: Biol Blood Marrow Transplant, 2015; 21(2); 266-74
11. Friedrichs B, Tichelli A, Bacigalupo A, Long-term outcome and late effects in patients transplanted with mobilised blood or bone marrow: a randomised trial: Lancet Oncol, 2010; 11(4); 331-38
12. Baron F, Mohty M, Blaise D, Anti-thymocyte globulin as graft-versus-host disease prevention in the setting of allogeneic peripheral blood stem cell transplantation: a review from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation: Haematologica, 2017; 102(2); 224-34
13. Bonifazi F, Rubio MT, Bacigalupo A, Rabbit ATG/ATLG in preventing graft-versus-host disease after allogeneic stem cell transplantation: Consensus-based recommendations by an international expert panel: Bone Marrow Transplant, 2020; 55(6); 1093-102
14. Deeg HJ, Storer BE, Boeckh M, Reduced incidence of acute and chronic graft-versus-host disease with the addition of thymoglobulin to a targeted busulfan/cyclophosphamide regimen: Biol Blood Marrow Transplant, 2006; 12(5); 573-84
15. Li HH, Li F, Gao CJ, Similar incidence of severe acute GVHD and less severe chronic GVHD in PBSCT from unmanipulated, haploidentical donors compared with that from matched sibling donors for patients with haematological malignancies: Br J Haematol, 2017; 176(1); 92-100
16. Dou L, Hou C, Ma C, Reduced risk of chronic GVHD by low-dose rATG in adult matched sibling donor peripheral blood stem cell transplantation for hematologic malignancies: Ann Hematol, 2020; 99(1); 167-79
17. Armand P, Kim HT, Logan BR, Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation: Blood, 2014; 123(23); 3664-71
18. Lee SE, Cho BS, Kim JH, Risk and prognostic factors for acute GVHD based on NIH consensus criteria: Bone Marrow Transplant, 2013; 48(4); 587-92
19. Dignan FL, Amrolia P, Clark A, Diagnosis and management of chronic graft-versus-host disease: Br J Haematol, 2012; 158(1); 46-61
20. Lee SJ, Wolff D, Kitko C, Measuring therapeutic response in chronic graft-versus-host disease. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: IV. The 2014 Response Criteria Working Group report: Biol Blood Marrow Transplant, 2015; 21(6); 984-99
21. Harris AC, Young R, Devine S, International, multicenter standardization of acute graft-versus-host disease clinical data collection: A report from the Mount Sinai Acute GVHD International Consortium: Biol Blood Marrow Transplant, 2016; 22(1); 4-10
22. Bonifazi F, Solano C, Wolschke C, Acute GVHD prophylaxis plus ATLG after myeloablative allogeneic haemopoietic peripheral blood stem-cell transplantation from HLA-identical siblings in patients with acute myeloid leukaemia in remission: final results of quality of life and long-term outcome analysis of a phase 3 randomised study: Lancet Haematol, 2019; 6(2); e89-e99
23. Socié G, Schmoor C, Bethge WA, Chronic graft-versus-host disease: Long-term results from a randomized trial on graft-versus-host disease prophylaxis with or without anti-T-cell globulin ATG-Fresenius: Blood, 2011; 117(23); 6375-82
24. Lazaryan A, Weisdorf DJ, DeFor T, Risk factors for acute and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation with umbilical cord blood and matched sibling donors: Biol Blood Marrow Transplant, 2016; 22(1); 134-40
25. Song ZY, Ren HY, Dong YJ, Impact of low-dose rATG prior to matched sibling donor hematopoietic stem cell transplantation for hematologic malignancies: Reduced risk of chronic graft-versus-host disease and improved survival outcomes: Cancer Manag Res, 2020; 12; 12287-300
26. Bailén R, Kwon M, Pascual-Cascón MJ, Post-transplant cyclophosphamide for GVHD prophylaxis compared to ATG-based prophylaxis in unrelated donor transplantation: Ann Hematol, 2021; 100(2); 541-53
27. Battipaglia G, Labopin M, Hamladji RM, Post-transplantation cyclophosphamide versus antithymocyte globulin in patients with acute myeloid leukemia undergoing allogeneic stem cell transplantation from HLA-identical sibling donors: A retrospective analysis from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation: Cancer, 2021; 127(2); 209-18
28. Zhang P, Curley CI, Mudie K, Effect of plasmapheresis on ATG (Thymoglobulin) clearance prior to adoptive T cell transfer: Bone Marrow Transplant, 2019; 54(12); 2110-16
29. Nishihori T, Al-Kadhimi Z, Hamadani M, Kharfan-Dabaja MA, Antithymocyte globulin in allogeneic hematopoietic cell transplantation: Benefits and limitations: Immunotherapy, 2016; 8(4); 435-47
30. Luznik L, O’Donnell PV, Symons HJ, HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide: Biol Blood Marrow Transplant, 2008; 14(6); 641-50
31. Nagler A, Labopin M, Dholaria B, Graft-versus-host disease prophylaxis with post-transplantation cyclophosphamide versus cyclosporine A and methotrexate in matched sibling donor transplantation: Transplant Cell Ther, 2022; 28(2); 86e1-e8
32. Kröger N, Solano C, Wolschke C, Antilymphocyte globulin for prevention of chronic graft-versus-host disease: N Engl J Med, 2016; 374(1); 43-53
33. Chang YJ, Wu DP, Lai YR, Antithymocyte globulin for matched sibling donor transplantation in patients with hematologic malignancies: A multicenter, open-label, randomized controlled study: J Clin Oncol, 2020; 38(29); 3367-76
34. Theurich S, Fischmann H, Shimabukuro-Vornhagen A, Polyclonal anti-thymocyte globulins for the prophylaxis of graft-versus-host disease after allogeneic stem cell or bone marrow transplantation in adults: Cochrane Database Syst Rev, 2012(9); CD009159
35. Devillier R, Labopin M, Chevallier P, Impact of antithymocyte globulin doses in reduced intensity conditioning before allogeneic transplantation from matched sibling donor for patients with acute myeloid leukemia: A report from the acute leukemia working party of European group of Bone Marrow Transplantation: Bone Marrow Transpl, 2018; 53(4); 431-37
36. Cho BS, Min GJ, Park SS, Low-dose thymoglobulin for prevention of chronic graft-versus-host disease in transplantation from an HLA-matched sibling donor: Am J Hematol, 2021; 96(11); 1441-49
Figures
 Figure 1. (A–D) Comparison of cumulative incidence of aGVHD and cGVHD between ATG group and non-ATG group (R version 4.0.3). aGVHD – acute graft-versus-host disease; cGVHD – chronic graft-versus-host disease.
Figure 1. (A–D) Comparison of cumulative incidence of aGVHD and cGVHD between ATG group and non-ATG group (R version 4.0.3). aGVHD – acute graft-versus-host disease; cGVHD – chronic graft-versus-host disease. Figure 2. (A–E) Comparison of cumulative incidence of relapse, non-relapse mortality, OS, DFS, and GRFS (R version 4.0.3). OS – overall survival; DFS – disease-free survival; GRFS – GVHD-free relapse-free survival; GVHD – graft-versus-host disease.
Figure 2. (A–E) Comparison of cumulative incidence of relapse, non-relapse mortality, OS, DFS, and GRFS (R version 4.0.3). OS – overall survival; DFS – disease-free survival; GRFS – GVHD-free relapse-free survival; GVHD – graft-versus-host disease. Tables
 Table 1. Clinical characteristics of 85 patients.
Table 1. Clinical characteristics of 85 patients. Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT.
Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT. Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients.
Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients. Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients.
Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients. Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients.
Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients. Table 1. Clinical characteristics of 85 patients.
Table 1. Clinical characteristics of 85 patients. Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT.
Table 2. Engraftment, infection, acute and chronic GVHD, and other complications after MSD-PBSCT. Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients.
Table 3. Univariate analysis of risk factors for grades 2–4 aGVHD and cGVHD in all patients. Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients.
Table 4. Multivariate analysis of risk factors for transplant outcomes in all patients. Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients.
Table 5. Univariate analysis of risk factors for moderate to severe cGVHD and CIR in all patients. Supplementary Table 1. Univariate analysis of risk factors for GRFS in all patients.
Supplementary Table 1. Univariate analysis of risk factors for GRFS in all patients. Supplementary Table 2. Univariate analysis of risk factors for OS and DFS in all patients.
Supplementary Table 2. Univariate analysis of risk factors for OS and DFS in all patients. In Press
Original article
Diagnostic Utility of FAR1 Methylation Levels in Hepatocellular Carcinoma Patients Undergoing Liver Transpl...Ann Transplant In Press; DOI: 10.12659/AOT.951568
Original article
Inferior Long-Term Outcome of Fatty Liver Allografts After Orthotopic Liver TransplantationAnn Transplant In Press; DOI: 10.12659/AOT.950589
Database Analysis
Identification and Validation of Liver Transplantation-Induced Acute Lung Injury Biomarkers Using a Bioinfo...Ann Transplant In Press; DOI: 10.12659/AOT.950289
Original article
Survival and Recurrence in Liver Transplant Patients With Intrahepatic Cholangiocarcinoma and Hepatocellula...Ann Transplant In Press; DOI: 10.12659/AOT.950997
Most Viewed Current Articles
24 Aug 2021 : Review article 18,372
Normothermic Machine Perfusion (NMP) of the Liver – Current Status and Future PerspectivesDOI :10.12659/AOT.931664
Ann Transplant 2021; 26:e931664
05 Apr 2022 : Original article 14,731
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
22 Nov 2022 : Original article 14,244
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
29 Dec 2021 : Original article 13,752
Efficacy and Safety of Tacrolimus-Based Maintenance Regimens in De Novo Kidney Transplant Recipients: A Sys...DOI :10.12659/AOT.933588
Ann Transplant 2021; 26:e933588








